Signalling
Part:BBa_F2620:Transfer Function
Designed by: Barry Canton [bcanton@mit.edu] and Anna Labno [labnoa@mit.edu] Group: iGEM04_MIT (2004-08-09)
|
|
The transfer function describes the equilibrium relationship between input (exogenous 3OC6HSL concentration) and output (PoPS) signals. We used the fluorescent reporter device, BBa_E0240 to indirectly measure the output from BBa_F2620 following induction with different exogenous concentrations of 3OC6HSL. Please see the part experience page for a general description of our characterization method.
Data
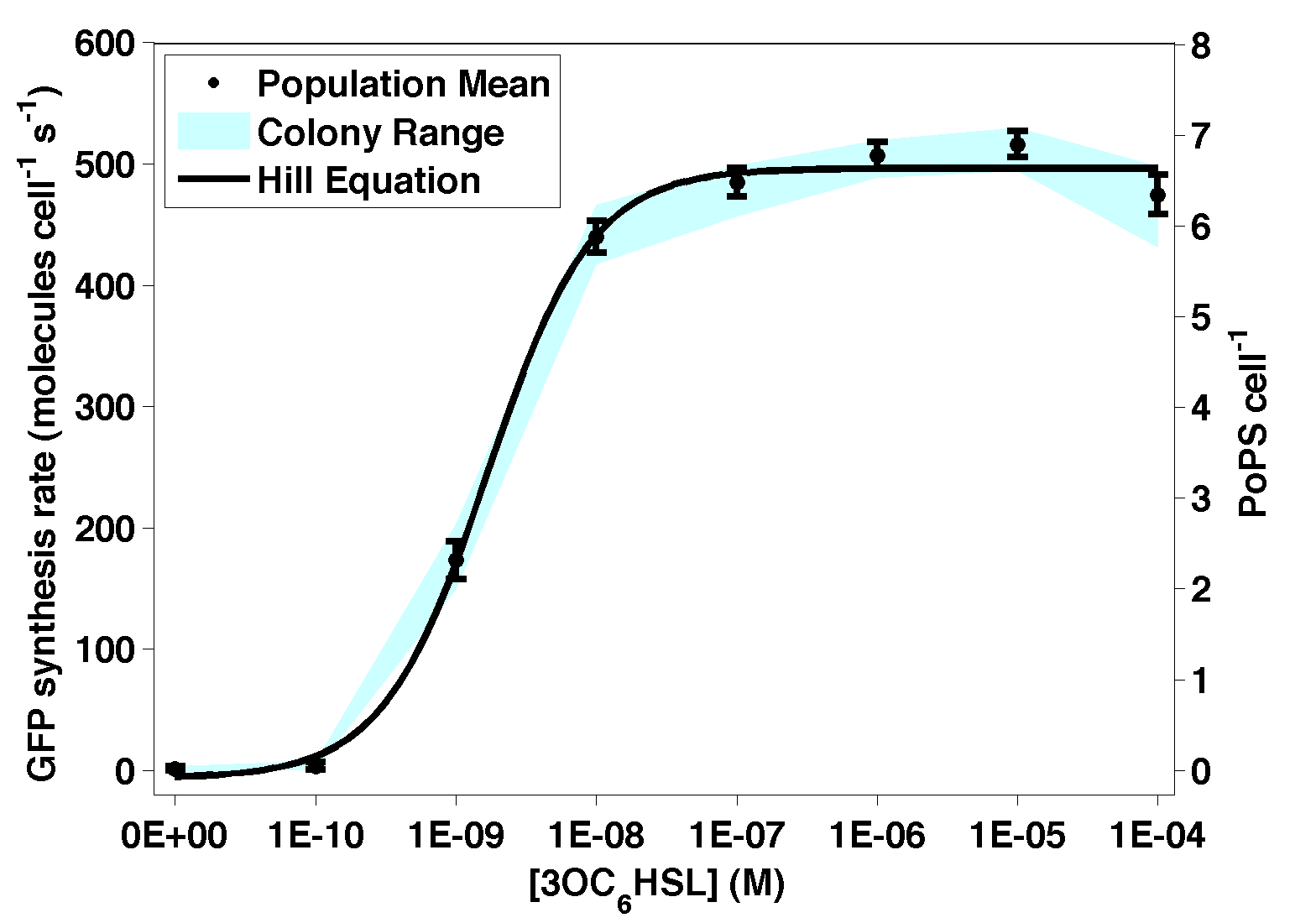
Figure 1 - Transfer function of BBa_F2620. The data points represent the mean of 6 or 9 individual measurements. The corresponding error bars represent the 95% confidence interval in the mean of the independent measurements. The blue shaded region represents the range bounded by the lowest and highest outputs among the independent measurements at a given input level. The solid black curve was calculated by fitting a simple Hill function to the experimental measurements.

Figure 2 - Growth curves of cultures of MG1655 bearing BBa_T9002 from the transfer function experiment under zero input and an input of 1E-4 M 3OC6HSL. The data points represent the mean of three identical samples; the error bars represent the 95% confidence interval in the estimate of the mean. The growth curves approximate straight lines on a log-ordinate axis, suggesting that exponential growth continues for the course of the experiment.
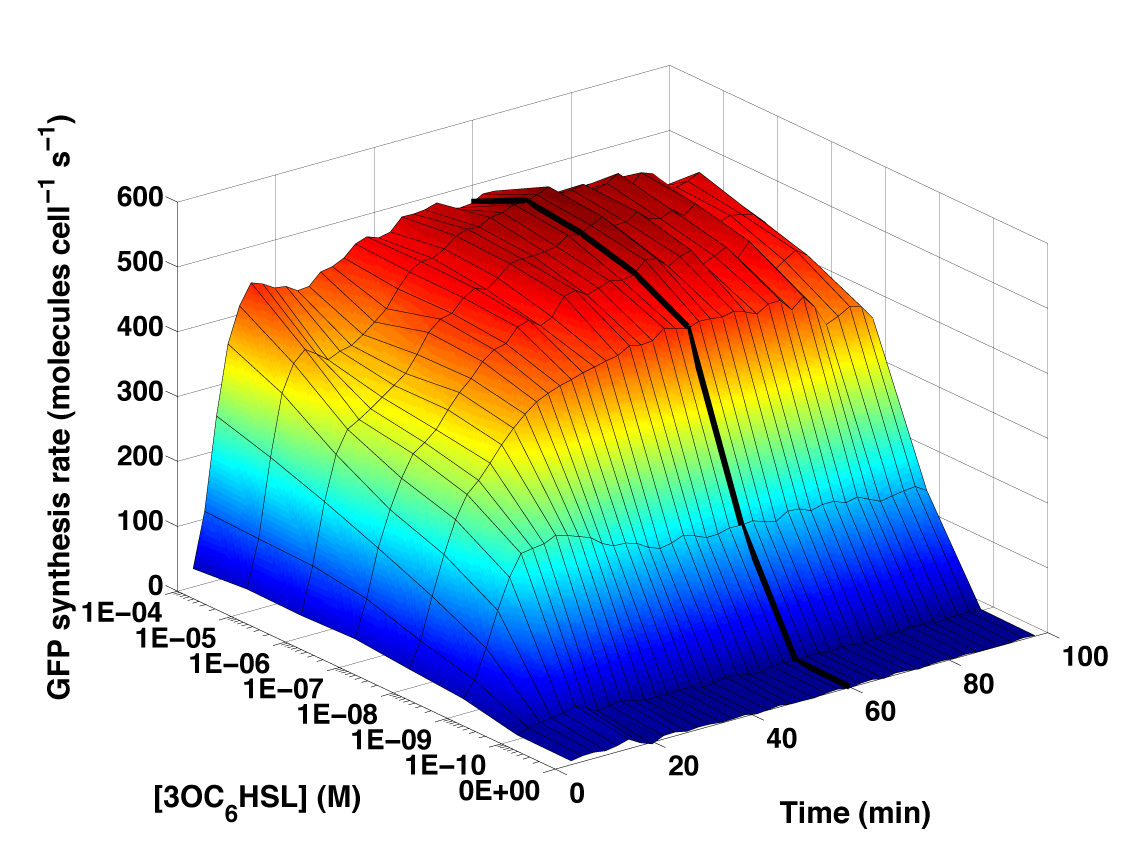
Figure 3 - Time and dose response measurements of GFP synthesis rate per cell following addition of 3OC6HSL. The surface shown is the average of six or nine independent measurements at each datapoint. The heavy black line is the 60 min time-slice that defines the transfer function shown in Figure 1.
| Data | Notes | Date Uploaded |
|---|---|---|
| File:BBa F2620-Variability-3-P2.txt | Raw data for colonies 1-3 | 12/11/2006 |
| File:BBa F2620-Variability-4-P1.txt | Raw data for colonies 4-6 | 1/4/2007 |
| File:BBa F2620-Variability-5-P1.txt | Raw data for colonies 7-9 | 1/5/2007 |
Protocol
- Three 5 ml cultures of [http://openwetware.org/wiki/Endy:M9_media/supplemented supplemented M9 medium] and antibiotic (kanamycin, 20 µg/ml) were inoculated with single colonies (~2mm ø) from a freshly streaked plate of MG1655 containing BBa_T9002. One 5 ml culture was inoculated with a single colony from a freshly streaked plate of MG1655 containing a BBa_T9002 mutant (T9002m) lacking a GFP expression device described in the stability section.
- Cultures were grown in 17 mm test tubes for 15 hrs at 37°C with shaking at 70 rpm.
- Cultures were diluted 1:1000 into 5.5 ml of fresh medium and grown to an OD600 of 0.15 under the same conditions as before. This growth took on average 4.5 hrs.
- Twenty-four 200 µl aliquots of each of the cultures were transferred into a flat-bottomed 96 well plate (Cellstar Uclear bottom, cat. # T-3026-16, Greiner).
- 2 µl of the stock concentrations of the cognate AHL, 3-oxohexanoyl-homoserine lactone (3OC6HSL), was added to each well to yield 8 different final concentrations (0, 1E-10, 1E-9, 1E-8, 1E-7, 1E-6, 1E-5 and 1E-4 M). Three replicate wells were measured for each concentration of 3OC6HSL. Three wells were each filled with 200 µl of medium to measure the absorbance background. Three further wells were each filled with 200 µl of the BBa_T9002 mutant culture to measure fluorescent background.
- The plate was incubated in a [http://openwetware.org/wiki/Endy:Victor3_plate_reader Wallac Victor3 multi-well fluorimeter] (Perkin Elmer) at 37°C and assayed with an automatically repeating protocol of absorbance measurements (600 nm absorbance filter, 0.1 second counting time through 5 mm of fluid), fluorescence measurements (488 nm excitation filter, 525 nm emission filter, 0.5 seconds, CW lamp energy 12901 units), and shaking (1 mm, linear, normal speed, 5 seconds). Time between repeated measurements was 2 min and 21 s. Approximately 6 min elapsed between beginning addition of 3OC6HSL to the wells and the first plate reader measurement. 3OC6HSL was added in order of increasing concentration to minimize GFP synthesis during plate loading. Cells appear to grow exponentially for the duration of the plate reader measurement protocol (see Figure 2 for representative growth curves).
- We repeated steps 1 through 6 on three separate days to obtain data for nine colonies from a single plate.
- We processed the data to compute the PoPS output from BBa_F2620 as described on the Data analysis page. The data for each colony tested was averaged across the three replicate wells. The mean for each colony was then averaged to obtain a population mean. The time and dose dependent input-output surface is shown above in Figure 3. Following an initial transient response, device output reached an approximate steady state.
- The snapshot transfer function in Figure 1 is the 60 min time-slice from the surface shown in Figure 3 (highlighted as a heavy black line). Error bars in Figure 1 representing the 95% confidence interval in the population for the nine independent samples. The cyan shaded region represents the range of the nine independent samples.
- To estimate parameters that characterize the measured transfer function, we used least squares estimation to fit a simple model to the data. A Hill equation derived from simple biochemical equations describes the data well (R2=0.99). In the equation (shown below), Pout is the PoPS per cell output of BBa_F2620, Pmax is the maximum output level, K is the switch point, and n is the hill coefficient describing the steepness of the transition from low output to high output.
- To gain further information about the transition region of the transfer function, measurements were subsequently taken at two intermediate 3OC6HSL concentrations (3.3E-09 M and 3.3E-08 M) using the same protocol defined above. Measurements were simultaneously taken at a subset of the original concentrations to ensure the new data was consistent with the earlier data. The new data was processed simultaneously with the original data, with the exception that only six independent colonies were measured for the intermediate 3OC6HSL concentrations.
<math> P_{out} = \frac{P_{max}[3OC_6HSL]^n}{K^n+[3OC_6HSL]^n} </math>

 1 Registry Star
1 Registry Star