Part:BBa_K3182104
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BamHI site found at 580
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
Introduction
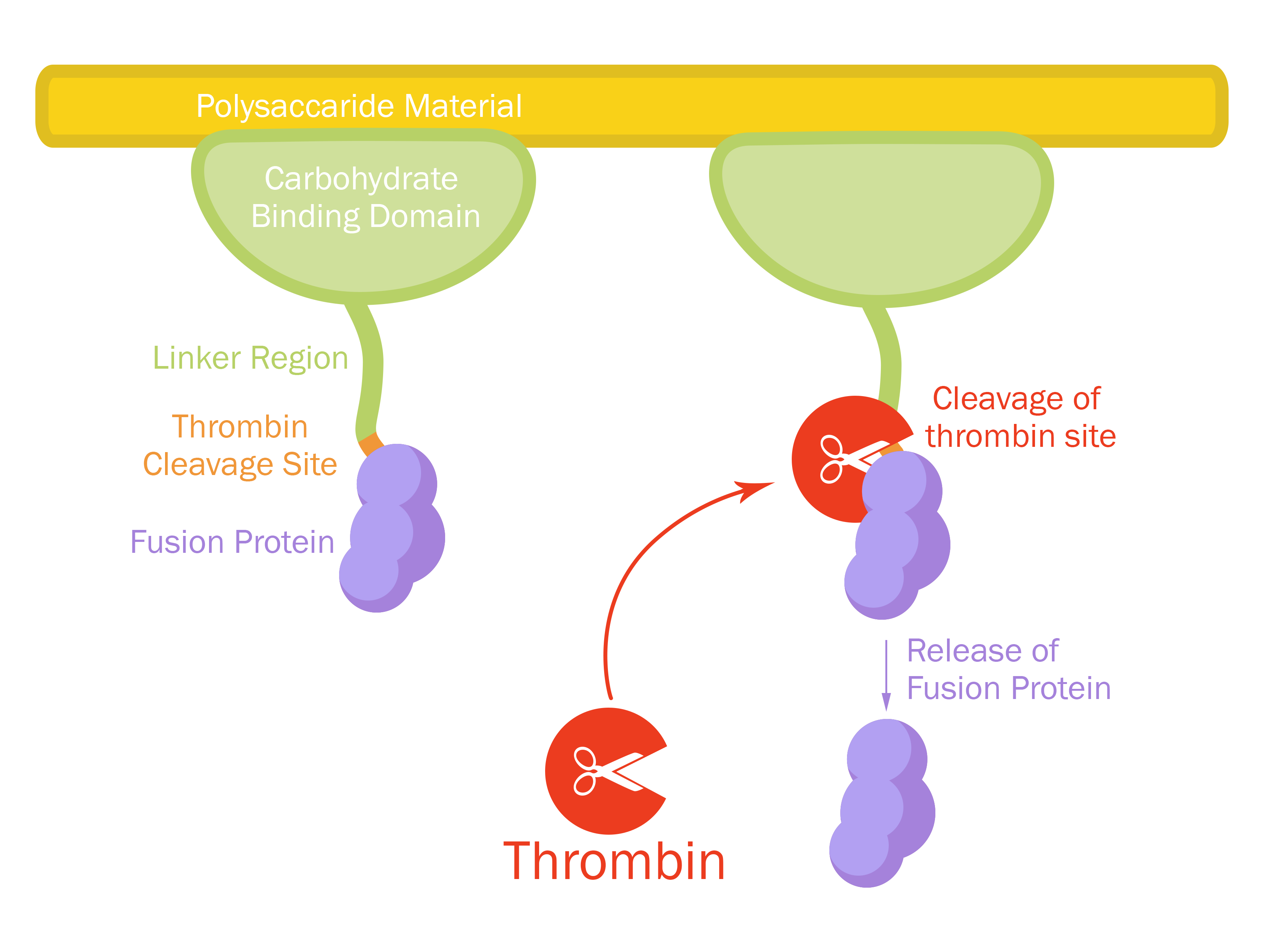
This part consists of a carbohydrate binding domain (CBD) from Clostridium thermocellum (C. thermocellum) cellulose scaffolding protein (CipA). This binding domain is a central part of Clostridium thermocellum's cellusome and has a strong affinity for cellulose. The CBD was fused to another protein using a flexible GS-linker (-GGGGSGGGGS-) in order to attach this complex to a polysaccaride material. A thrombin cleavage site (-LVPRGS-) was added to the end of the linker and its breakage will leave a glycine and serine attached to the N-terminal of the fusion protein. The main mechanism of iGEM19 Linköping's project can be seen in Figure 1.
Protease site and use
The thrombin site was added to enable the ability to release the fusion protein down into skin wounds. Thanks to our integrated human practice we learned that infections span much deeper into wounds that we thought. Simply attaching the CBD-fusion protein to a carbohydrate material would not enable the fusion protein to reach far into the wound. The thrombin site was also chosen because of thrombin's endogenous existence in humans.
Assembly compabilities
An internal BamHI recognition sequence (RS) has been added to enable interchangeable fusion proteins to the CBD. BamHI was chosen because its RS codes for glycine and serine, fitting it to the end of the thrombin site. It is also a cost-effective enzyme and is unaffected by methylated DNA. BamHI is a part of the RFC21 standard.
CBDcipA crystal structure
Important molecular faces
CBDcipA is composed of a nine-stranded beta sandwich with a jelly roll topology and binds a calcium ion, which can be seen in Figure 2. It further contains conserved residues exposed on the surface which map into two clear surfaces on each side of the molecule. One of the faces mainly contains planar strips of aromatic and polar residues which may be the carbohydrate binding part. Further aspects are unknown and unique to this CBD such as the other conserved residues which are contained in a groove.
Carbohydrate binding domain specificity
Since the CBD is from the cellusome of C. thermocellum some research labeled it a cellulose binding domain. However, iGEM19 Linköping noticed that this domain could also bind to different sources of polysaccaride materials. This serves as a domain for iGEM19 Linköpings modular bandage, where the polysaccaride material can be exchanged for other/similar materials and not exclusively cellulose.
The choice of carbohydrate binding domain
iGEM Linköping 2019 chose CBDcipA due to the fact that many other iGEM teams had explored the possibilities of this domain. Our basic design was influenced by [http://2014.igem.org/Team:Imperial iGEM14 Imperial], [http://2015.igem.org/Team:edinburgh iGEM15 Edinburgh] and [http://2018.igem.org/Team:ecuador iGEM18 Ecuador]. Purification and where to place the fusion protein (N- or C-terminal) was determined by studying the former projects. CBDcipA also originates from a thermophilic bacteria which further increases the domain's applications.
Expression system
The part has a strong expression with a T7-RNA-polymerase promotor (BBa_I719005), seen in Figure 3, as well as a 5'-UTR (BBa_K1758100) region which has been shown to further increase expression in Escherichia coli (E. coli) (BBa_K1758106), ([http://www.ncbi.nlm.nih.gov/pubmed/2676996 Olins et al. 1989]), ([http://www.ncbi.nlm.nih.gov/pubmed/23927491 Takahashi et al. 2013]).

Antimicrobial Agent - CHAP(K)
Bacteriophage endolysins digest the peptidoglycan wall of bacteria. The LysK is an endolysin targeting gram positive bacteria. It has two catalytical domains known as CHAP (cysteine-and histidine dependent amidohydrolase/peptidase) and a central amidase-2 domain together with a cell binding domain. The CHAP domain (more specifically CHAP C1-C162), which is the truncated part expressed in this year’s LiU iGEM team, still has lytic activity against staphylococcus. The C1 to C162 amino acids are required for full endopeptidase activity. The purpose of the truncation is to increase the antimicrobial effect of CHAP, decrease its risk of immunological response and increase protein yield.
The enzyme is designed to battle the Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, Enterobacter spp. family of pathogens (ESKAPE). ESKAPE is a family(s) of bacteria which has multiple substrains that has evolved resistance to the most commonly used antibiotics.
Usage and Biology
The desired function of this construct is that the CBD will allow a more efficient expression of antimicrobial peptides by being a hydrophilic anchor to the bactericidal peptide or enzyme. The CBD can be used for easy purification of the fusion protein and via thrombin cleavage of the linker, yield the peptide or enzyme only. In iGEM19 Linköping's project the thought was to create an antimicrobial bandage by having a polysaccharide bandage with CBD-antimicrobial agents bound to it. When applying the bandage to a patient, the patient's blood (containing thrombin) will release the peptide or enzyme into the wound (thrombin sprays used in hospitals can be used as well) leading to the activation of the peptides/enzymes and enabling them to reach the bacteria, thus eliminating the infecting pathogens.
Expression, purification and protease treatment
The antimicrobial agents which iGEM19 Linköping used were all expressed the same way. Early expression experiments showed great promise but with low yields. The early experiments were done with standard E. coli BL21 (DE3) expression, cultures were grown to an optical density at 600 nm (OD600) of 0.4-1.0 in varying volumes and induced with 0.5 mM IPTG, and let to express the agents for 16 hours in 18 °C. Harvest of the cells was done at 3500 rpm for 30 minutes.
Solubilization and purification
After harvest the cells were re-suspended in phosphate buffered saline (PBS, 137 mM NaCl, 2.7 mM KCl, 10 mM Na2HPO4, 1.8 mM KH2PO4) buffer or carbohydrate binding module buffer (CBM-buffer, Tris–HCl pH 7.0, 20 mM NaCl, 5 mM CaCl2) and sonicated for 6 minutes, 30 % amplitude and 30 seconds on, 30 seconds off. The sonicated bacteria was then centrifuged at 12000 g for 15 minutes. The soluble fraction was then purified by attaching the CBD-fusion to cellulose. This showed very faint bands on SDS-PAGE analysis. Because of the low yield from the soluble fraction, a detergent (Triton X100, 1 %) was used to re-suspend the lipid-soluble proteins (insoluble fraction).
The Triton-X100 1 % fraction was later bound to microcristallin cellulose by Sigma Aldrich. A culture of 1 L E. coli BL21 (DE3) treated as above and lysed in 60 mL CBM-buffer. Half of that amount was then incubated with 5 g of microcristallin cellulose for 2 hours in 4 °C. After binding the CBD-fusion protein to the cellulose, washing was done with either 70 % ethanol, PBS- or CBM-buffer. A volume of 10 mL of each solution was added and incubated with the cellulose for 30 minutes. This step was repeated three times to remove unbound proteins and any residual Triton X100. After washing, elution was done with deionized water. A volume of 10 mL was sufficient to elute some CBD-fusion proteins (room temperature), while others needed higher temperatures (37 °C). This was done in 50 mL tubes under shaking. In hindsight, a column should be used to better track the elution.
Thrombin treatment
These bound fractions could also be cleaved with thrombin, where 25 units Bovine Thrombin (100 µL) by Sigma Aldrich was added. To this thrombin cleavage buffer (20 mM Tris-HCl, 150 mM NaCl and 2.5 mM CaCl2) was also added (500 µL) along with deionized water (4.4 mL). The cellulose bound fraction was discarded and the supernatant, with thrombin and the now unbound antimicrobial agent, was saved for later use. The results from this can be seen in Figure 4.
Antimicrobial activity agents in different states
All experiments below used E. coli BL21 (DE3) or Bacillus Subtilis in concentrations of 10000 CFU/mL. This was done in order to get starting cultures of 0 OD600. Using spectrometry to measure the time until the bacteria started growing, instead of showing the killing capability on high optical density cultures, mimicked early stage wounds. This also showed many other interesting things, which can be seen in the results below. One being that an equilibrium settled in lower than for the negative control, meaning even though the antimicrobial agents could not inhibit the growth fully, it was slowed and the stationary phase was reached earlier (Figure 5).
Antimicrobial activity of unbound agents

Unbound agents
As a positive control before starting our experiments with the CBD-agent-bandage, the unbound agents were tested for antimicrobial activity when they were not bound to the CBD. The results from this experiment can be seen in Figure 5. The agents were purified and treated with thrombin as stated before. The experimental setup was the same as the earlier experiment but instead of adding water to the negative control, thrombin (0.5 U, final concentration 0.6 µM) and thrombin cleavage buffer (40 µL, 20 mM Tris-HCl, 150 mM NaCl and 2.5 mM CaCl2) was added. This was because the agents had thrombin and cleavage buffer with them. This showed to not affect the bacterial growth, but rather increase it due to the added salt due to the cleavage buffer.
| None |


