Part:BBa_K257002
ClyA_Fusion_C-Term
ClyA (or HlyE) is an alpha-PFT for Pore Forming Toxins.
PFTs are potent virulence factors class starting in a soluble form to an outer membrane-integrated pore. They exhibit their toxic effect either by membrane permeability barrier destruction or by toxic components delivery through the pores which forming by several assembly 8 or 13 ClyA subunits.
- ClyA can be used to co-localize fully functional heterologous proteins directly in bacterial OMVs
- We can fuse GFP to the C or N term of Cly A, to track OMVs easily.
- ClyA is capable of co-localizing a variety of structurally diverse fusion partners to the surface of E. coli and their released vesicles, but only when the periplasmic disulfide bond-forming machinery was present ,it’s makes OMVs an ideal structure to transport hydrophobic compounds like membrane proteins into the host.
- Cly A confers vesicle binding to and invasion of host cells.
- ClyA was significantly enriched in OMVs relative to other lumenal and membrane bound OMV proteins.
Team XMU-China 2022
Usage and design
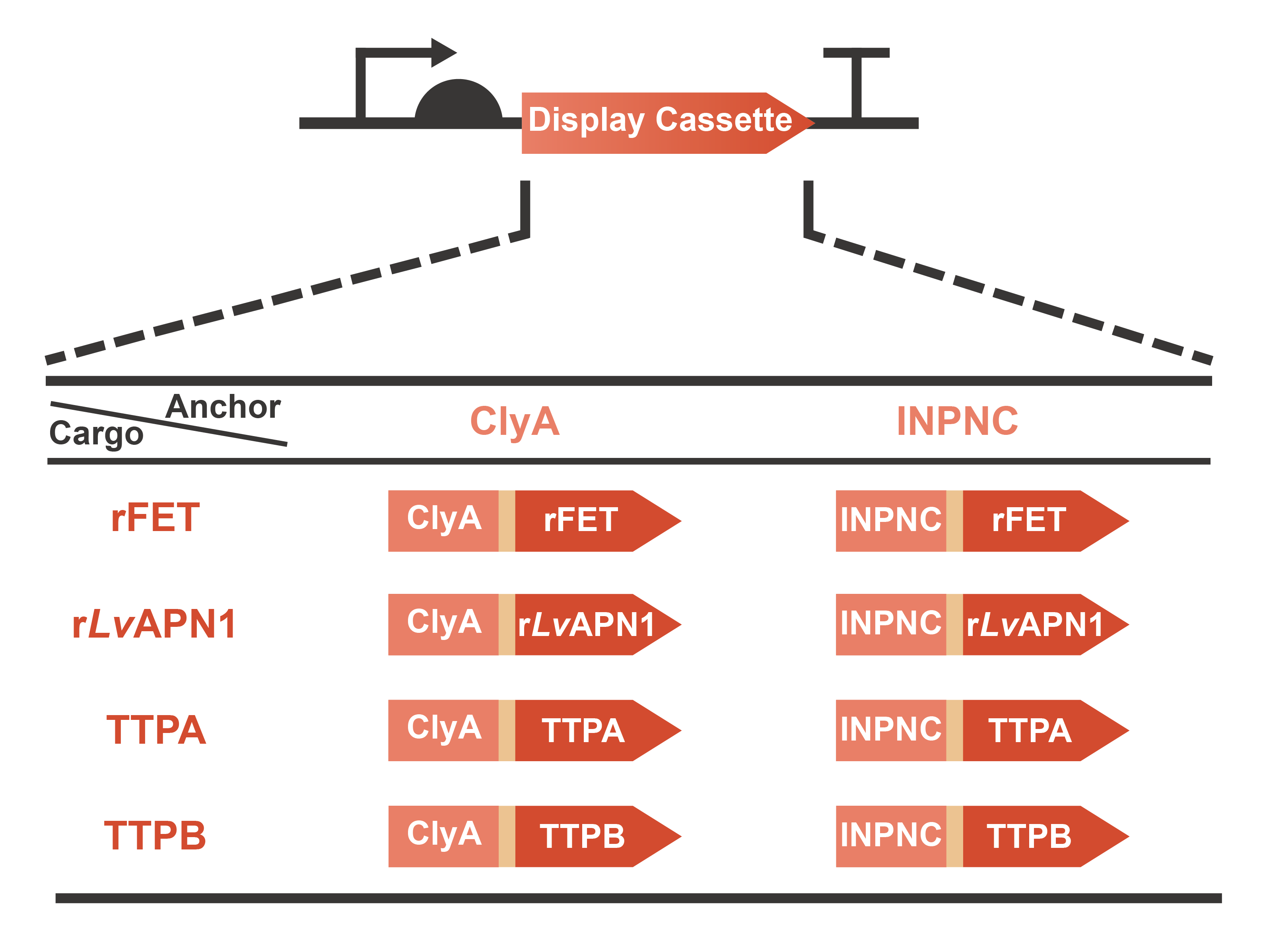
Engineering OMVs for treating and preventing AHPND caused by the pathogen V. parahaemolyticus are a significant part of OMEGA project (Operable Magic to Efficiently Getting over AHPND). Based on the efforts of our previous projects in 2020 (AnTea-Glyphosate) and 2021 (SALAGE), we further developed the surface display system on the OMVs released by the engineered bacteria. The usage of cargo proteins was no more limited to enzymes that are usually utilized to catalyze series bio-chemical reactions, since some receptors or ligands involved in complex protein-protein interaction (PPI) were selected as the cargo candidates. This year, we chose two classic anchor proteins, ClyA and INPNC, to construct the display cassette with various cargo proteins including rFET (receptor), rLvAPN1 (receptor), TTPA (ligand) and TTPB (ligand) (Fig. 1). On one hand, with the receptors displayed, OMVs will gain the function of neutralizing toxins secreted by V. parahaemolyticus. On the other hand, with the assistance of ligands displayed on the surface, OMVs will become a special vector to deliver antimicrobials for the specific pathogen. In summary, we have taken a step closer to the collections of extracellular functional elements (EFE), combining the OMVs, secretion systems and surface display systems which we have been dedicated to since 2020. Learn more information from our Design page.
Fig. 1 Graphic description of the expression gene circuits for display cassette designed in OMEGA project.
The isolation and purification of OMVs can be achieved in many ways. Nathan et al. obtained the purified OMVs via immobilized metal affinity chromatography (IMAC) using a His-tag mutant (1).In order to purify OMVs, we added a his-tag (6×His) at the C-terminal of ClyA. We used both BBa_I0500 and BBa_B0034 to construct the expression system and obtained the composite part BBa_K4195135, which are assembled into the vector pSB1C3 by standard BioBrick assembly. The constructed plasmids were transformed into E. coli BL21(DE3), then the positive transformants were selected by chloramphenicol and confirmed by colony PCR and sequencing.
Characterization
1.Identification
After transforming the plasmid into E. coli BL21(DE3), colony PCR was used to verify that the plasmid was correct. Target bands (2662 bp) can be observed at the position between 3000 bp and 2000 bp (Fig. 2).

Fig. 2 DNA gel electrophoresis of the colony PCR products of BBa_K4195135_pSB1C3.
2.Immunofluorescence (IF)
The culture was incubated overnight. Then the FITC-labeled anti-His-Tag antibody was used to target the His-tag (6×His) displayed via ClyA, followed by measuring the fluorescence intensity (λEx= 492 nm, λEm = 528 nm) to OD600 of the culture.
The results showed that the ratio of fluorescence intensity to OD600 of positive control (bacteria harboring BBa_K4195135) is higher than that of negative control (bacteria harboring BBa_K4195119) (Fig. 3), which indicated that these two surface display systems can work well.
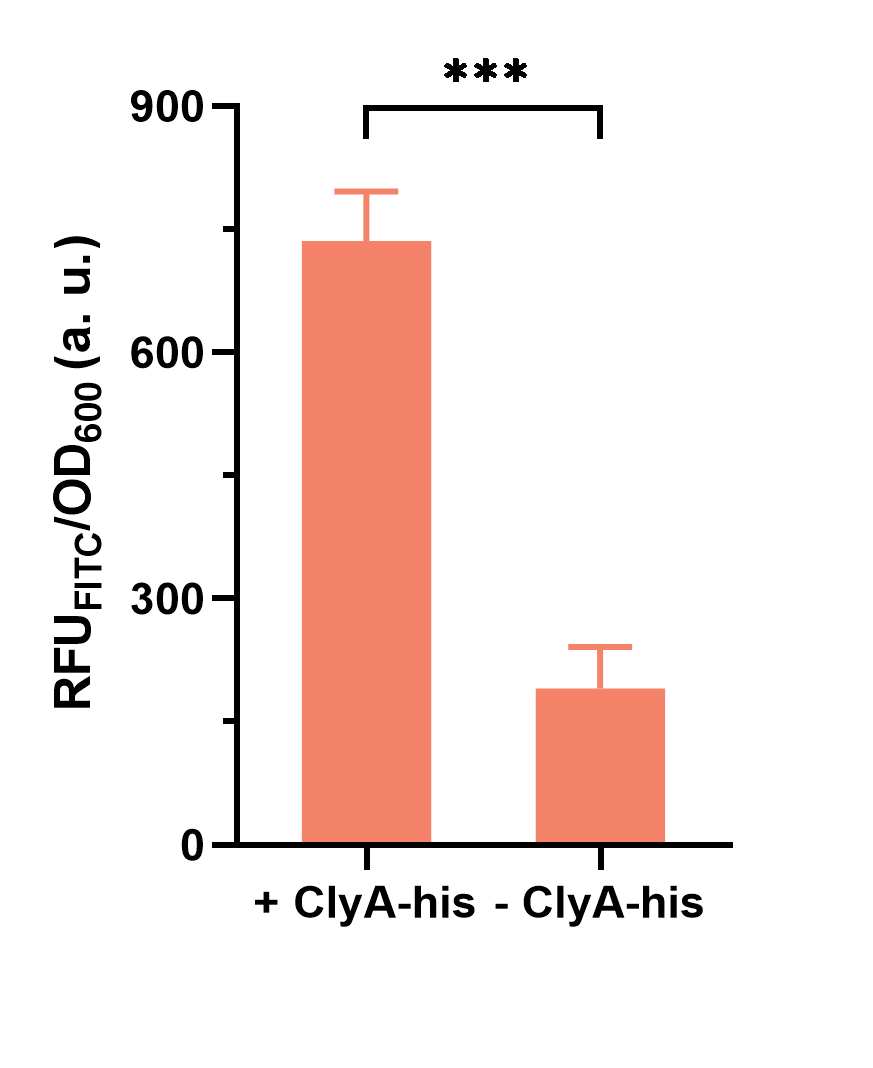
Fig. 3 Fluorescence intensity/OD600 of E. coli whether express the anchor protein or not (p=0.0003).
Reference
1. N. J. Alves, K. B. Turner, K. A. DiVito, M. A. Daniele, S. A. Walper, Affinity purification of bacterial outer membrane vesicles (OMVs) utilizing a His-tag mutant. Res. Microbiol. 168, 139-146 (2017).
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BglII site found at 85
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
References
- The structure of a cytolytic a-helical toxin pore reveals its assembly mechanism, M.Mueller & N.Ban. 2009 - [http://www.ncbi.nlm.nih.gov/pubmed/19421192 | 19421192 ]
- S.N. Wai, B.Lindmark, T.Soderblom, A.Takade, M.Westermark, J.Oscarsson, et al. Vesicle-mediated export and assembly of pore-forming oligomers of the enterobacterial ClyA cytotoxin, 2003, Cell, 115, 25–35. [http://www.ncbi.nlm.nih.gov/pubmed/14532000 14532000]
- F.J del Castillo, F. Moreno. and I.del Castillo. Secretion of the Escherichia coli K-12 SheA hemolysin is independent of its cytolytic activity, 2001, FEMS Microbiol.Lett. 204, 281–285. [http://www.ncbi.nlm.nih.gov/pubmed/11731136 11731136]
- J.E.Galen, L.Zhao, M.Chinchilla, J.Y.Wang,M.F.Pasetti, J.Green and M.M. Levine. Adaptation of the endogenous Salmonella enterica serovar Typhi clyA-encoded hemolysin for antigen export enhances the immunogenicity of anthrax protective antigen domain 4 expressed by the attenuated live-vector vaccine strain CVD 908-htrA, 2004, Infect. Immun. 72, 7096–7106.[http://www.ncbi.nlm.nih.gov/pubmed/15557633 15557633]
- J.Y. Kim, A.M. Doody, D. J. Chen, G.H. Cremona, M.L. Shuler, D.Putnam,and M.P. DeLisa.Engineered. Bacterial Outer Membrane Vesicles with Enhanced Functionality, 2008, J. Mol. Biol. 380, 51–66. [http://www.ncbi.nlm.nih.gov/pubmed/18511069 18511069]
- N.C.Kesty, M.J.Kuehn. Incorporation of heterologous outer membrane and periplasmic proteins into Escherichia coli outer membrane vesicles, 2004, J Biol Chem. 279(3):2069-76.[http://www.ncbi.nlm.nih.gov/pubmed/14578354 14578354]
| None |