Part:BBa_K3904005
Bile-regulated and cold-inducible VapXD kill-switch
To prevent our genetically modified probiotic bacteria from escaping the intestine to the environment, they were provided with the disabling mechanism, which could be activated after exposure to some environmental factors. This goal was obtained by implementation of the newly described type II toxin-antitoxin (TA) system VapXD from non-typeable Haemophilus influenzae [1].
Introduction
Vilnius-Lithuania iGEM 2021 project AmeByelooks at amebiasis holistically and comprehensively, therefore target E. histolytica from several angles: prevention and diagnostics. Our team's preventive solution consists of probiotics engineered to produce naringenin - an antiprotozoal compound. Two strains of genetically modified microorganisms were chosen as the main chassis - world-renowned Lactobacillus casei BL23 (Lactobacillus paracasei) and Escherichia coli Nissle 1917. Furthermore, the team made specific gene deletions to enhance naringenin production and adapted a novel toxin-antitoxin system to prevent GMO spreads into the environment. The diagnostic part includes a rapid, point of care, user-friendly diagnostic test identifying extraintestinal amebiasis. The main components of this test are aptamers specific to the E. histolytica secreted proteins. These single-stranded DNA sequences fold into tertiary structures for particular fit with target proteins.
Contents
Usage and Biology
Bile-regulated and cold-inducible VapXD kill-switch is a composite part that was designed to ensure two-factor control over GMO probiotics spread from the human intestine to the environment. If probiotic bacteria are present in the intestine where bile-salts occur, and temperature is around 37 °C, bile-regulated promoter induces anti-toxin production, as cold-inducible promoter activity is inhibited. However, if bacteria escape, a bile-inducible promoter is inhibited, as cold-regulated promoter causes greater toxin production and lowers bacteria’s chance of survival.
Part characterization
First of all, this part was characterized in a plasmid construct. Series of plate reader measurements of OD600 were performed while changing conditions in LB media (see the 1 Table).
Table 1. VapXD kill-switch characterization conditions
| Temperature | Bile salts |
|---|---|
| 37 °C | + |
| 37 °C | - |
| 24 °C | + |
| 24 °C | - |
What is more, 10 times serial dilutions of bacteria were performed with the start point of OD600=0.2. As controls for this experiment, bacteria that either produce only anti-toxin or toxin were used. In analogy, the same measurements were performed with bacteria containing such construct in their genome.
Results
VapXD kill-switch accuracy was firstly evaluated with a serial dilution spotting method. Sets of agar plates with and without bile salts were incubated in different temperatures of 37 °C, 30 °C, and room temperature (24 °C). However, the distinction between plates with and without bile salts supplementation was only seen at room temperature (figures 1 and 2).
Fig. 1 Serial dilution spotting assay on M9 medium without bile salts
Fig. 2 Serial dilution spotting assay on M9 medium with bile salts
VapXD system without promoter before toxin – K, bile-regulated and cold-inducible VapXD kill-switch marked as 1, Bile-regulated and p-slpA VapXD kill-switch - 2, bile-regulated and J23101 VapXD kill-switch - 3, bile-regulated and J23106 VapXD kill-switch - 4, bile-regulated and J23101 VapXD kill-switch - 5
Results showed that bile-regulated and cold-inducible VapXD kill-switch and bile-regulated and p-slpA VapXD kill-switch work on agar plates at room temperature, as with lower OD600 values they inhibited cell growth.
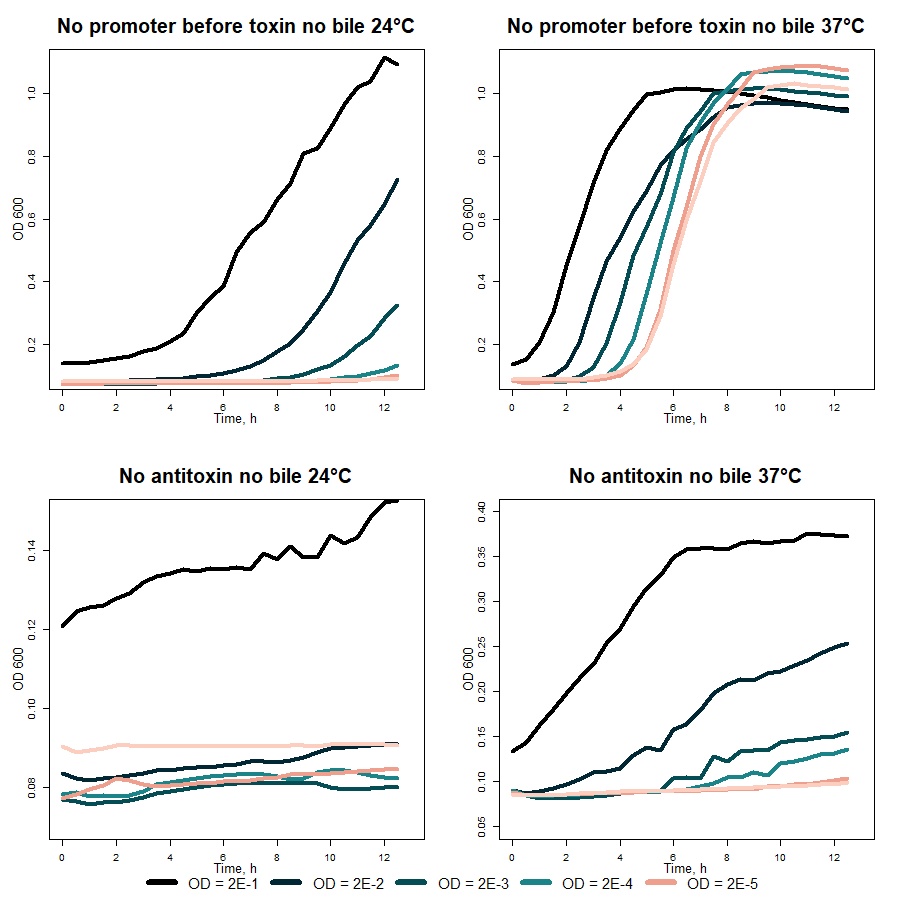
Further VapXD kill-switch performance was tested in liquid medium, as OD600 measurements were performed. First of all, VapD toxin (BBa_K3904000) activity was characterized while regulating its production with cold-induced promoter (BBa_K3904003). Graphs at the top of figure 3 illustrate bacteria growth without toxin and graphs at the bottom with the toxin in different temperatures. While comparing obtained data in 37 and 24 °C, temperature change can be seen as inducing greater toxin production and cell death. On the other hand, VapX activity is not fully accurate due to the leakage of the promoter during the temperature change from 37 and 24 °C.
Fig. 3 VapD toxin (BBa_K3904000) characterization
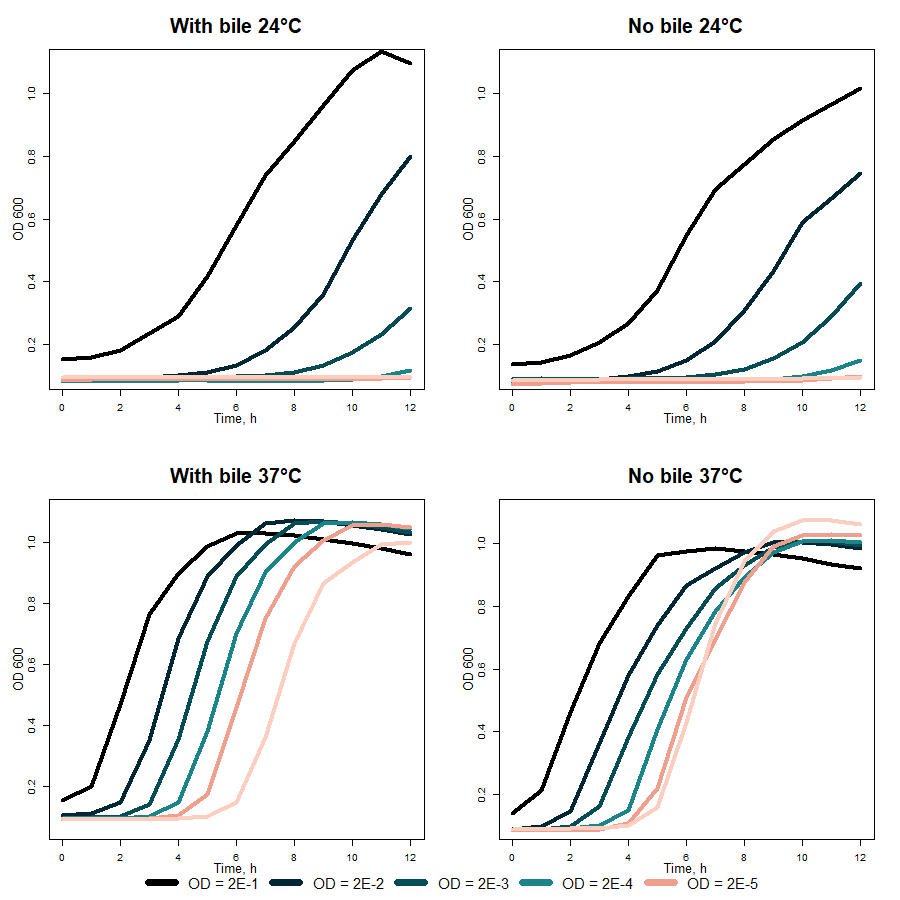
What is more, VapXD with the bile-induced promoter before antitoxin and with the cold-induced promoter before toxin was characterized. Graphs at the top of 4 figure demonstrate bacteria growth with and without bile salts supplementation in media at 24 °C, as graphs in the bottom at 37 °C. It can be seen that OD600 in the presence of bile salts and 37 °C bacteria grow more exponentially than without bile salts and in 24 °C. In the ideal case, no antitoxin should be produced without bile salts and 24 °C, and toxin synthesis should be induced. However, the results indicate that in such conditions, bacteria growth is only slightly repressed.
Fig. 4 Cold-shock promoter (BBa_K3904003) activity comparison in different temperatures and with/without bile salts
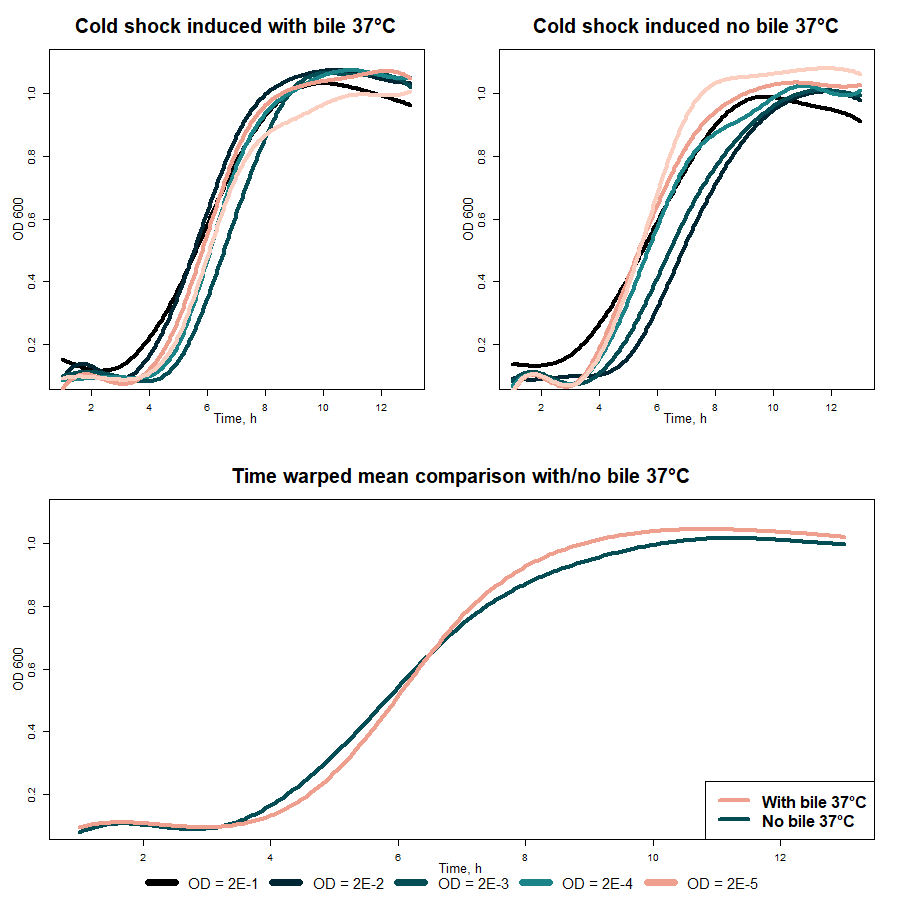
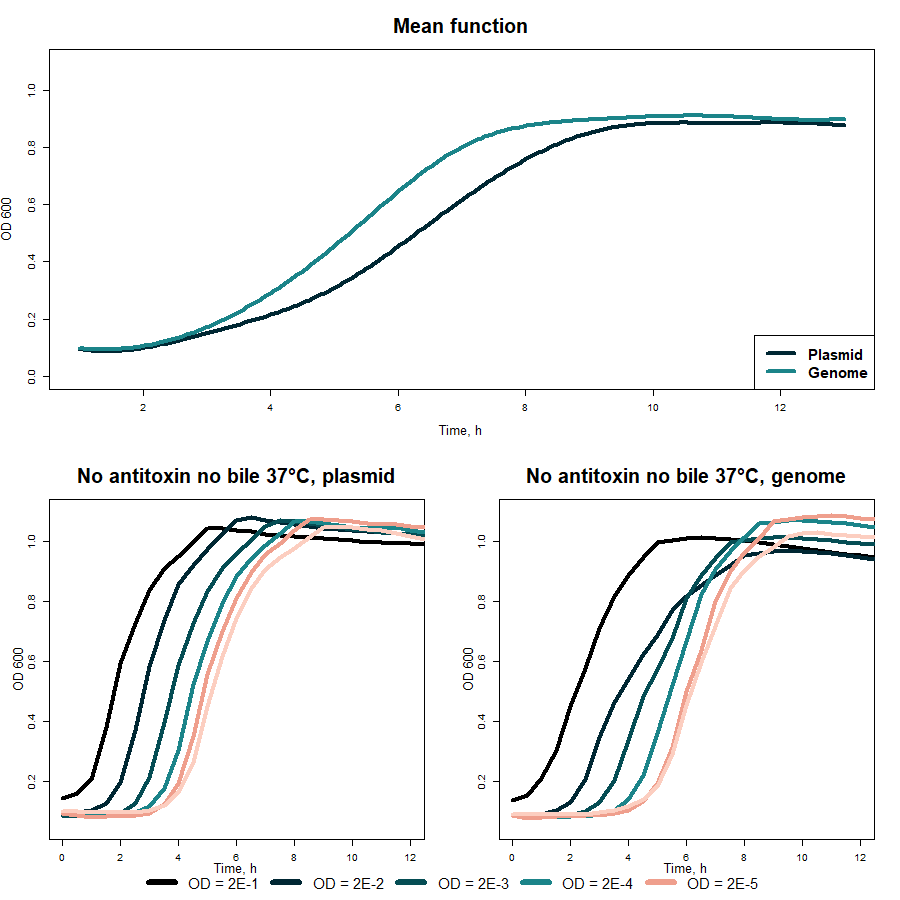
When results in 37 °C obtained with different OD600 values were averaged (fig. 5 “Mean comparison with/no bile 37 °C), the difference between measurements with and without bile salts appeared to be mathematically insignificant.
Fig. 5 Cold-shock promoter (BBa_K3904003) comparison with and no bile salts in 37 °C
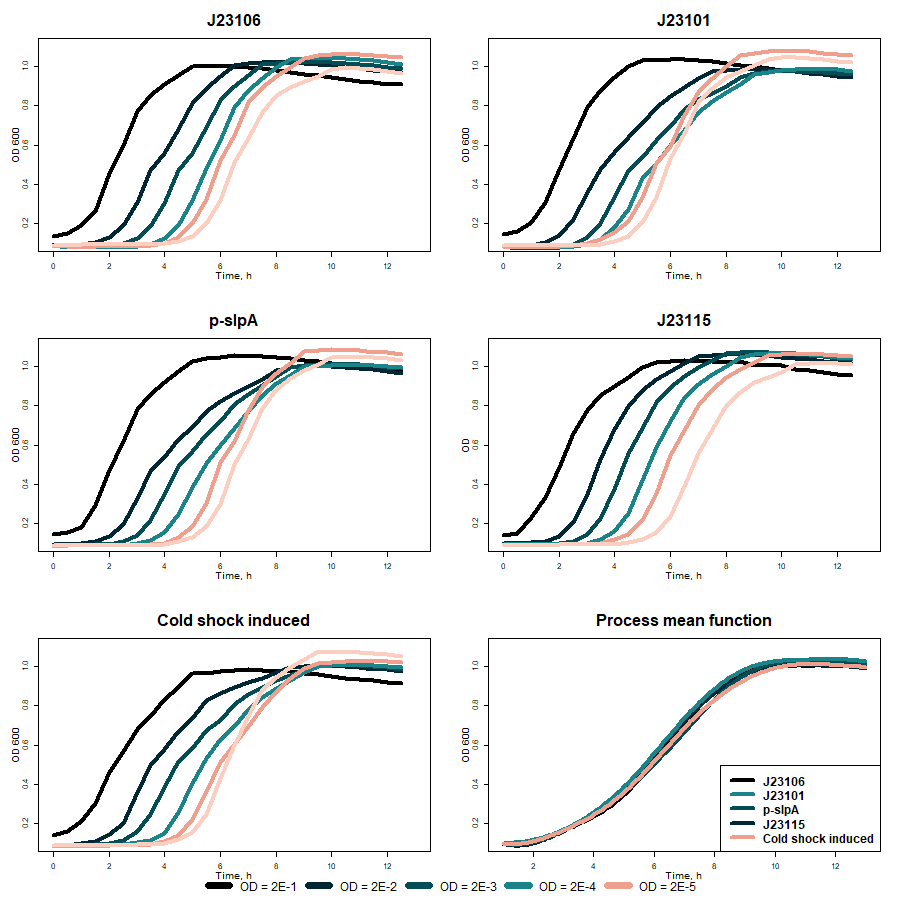
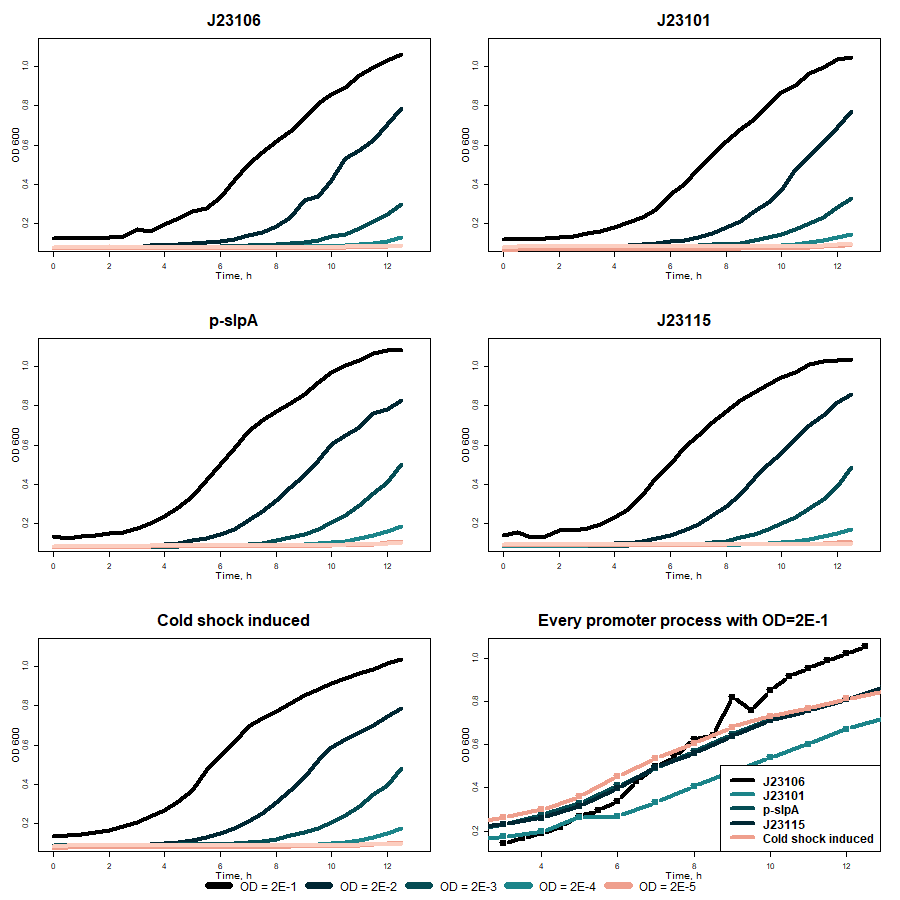
The activity of different promoters before VapX toxin was compared without bile salts supplementation in media at 37 °C. From promoters’ strength evaluation measurements (fig. 6), it was seen that the p-slpA promoter (BBa_K3904712) is the strongest in our inventor. VapXD assessment also showed that under this promoter, toxin production is more significant than under other promoters. The sloping graph rise illustrates this because more toxin is produced, and bacteria growth is inhibited. However, after the results were averaged, no significant difference between different promoters was seen.
Fig. 6 Comparison of VapXD kill-switch with different promoters before toxin (without bile salts in 37 °C)
While comparing results with different promoters in 24 °C, no significant difference can be seen (fig. 7).
Fig. 7 Comparison of VapXD kill-switch with different promoters before toxin (without bile salts in 24 °C)
Furthermore, we successfully inserted the VapXD system construct into E. coli Nissle 1917 genome. Both of these VapXD systems constructs have been inserted into the E. coli Nissle 1917 genome with 100 % efficiency and proved by DNA sequencing.
VapXD kill-switch inserted in E.coli Nissle 1917 genome was also characterized by OD600 measurements. The results of the genomic construct were compared to the plasmid construct.
The first observation from fig. 8 was that bacteria containing construct in the genome grow more exponentially. It does not have to maintain antibiotic resistance and the burden for the cell lowers.
Fig. 8 E.coli Nissle 1917 growth comparison with genomic and plasmid VapXD constructs
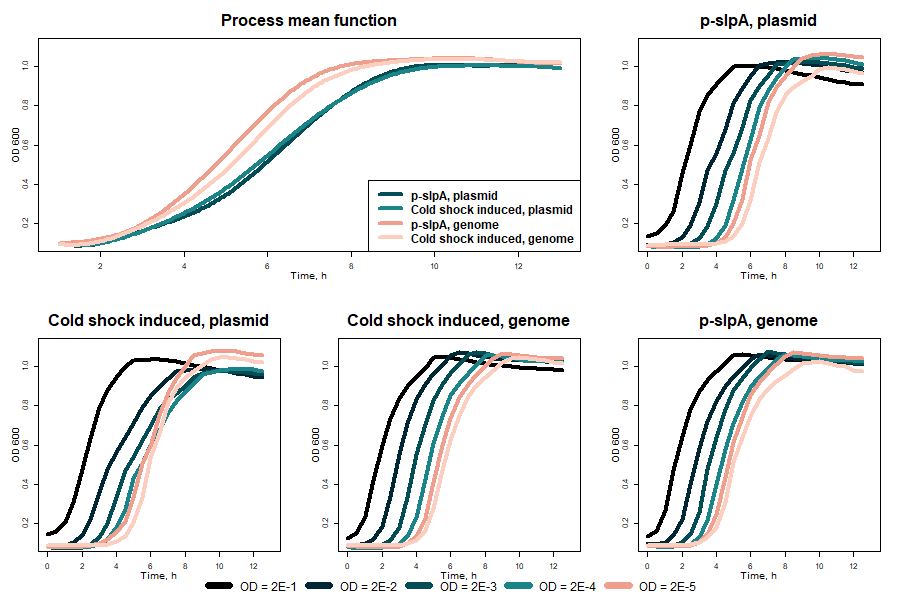
As shown in fig. 9 no significant difference in the VapXD kill-switch performance was seen while comparing results of the genomic and plasmid constructs with the same promoters.
Fig. 9 VapXD kill-switch performance with the same promoters in the genome and in the plasmid at 37 °C
While comparing results obtained during the measurements with and without bile salts (fig. 10), no significant difference was seen. This once again indicates promoter leakage and the system's inaccuracy.
Fig. 10 VapXD system performance in the environment with and without bile salts
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BglII site found at 199
Illegal BamHI site found at 397
Illegal XhoI site found at 442 - 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal AgeI site found at 179
- 1000COMPATIBLE WITH RFC[1000]
References
- Bertelsen, M. B., Senissar, M., Nielsen, M. H., Bisiak, F., Cunha, M. V., Molinaro, A. L., Daines, D. A., & Brodersen, D. E. (2021). Structural Basis For Toxin Inhibition in the VapXD Toxin-Antitoxin System. Structure, 29(2). https://doi.org/10.1016/j.str.2020.10.002
| None |