Part:BBa_K4195032
INPNC-rLvAPN1-his
Biology
INPNC
INPNC is a truncated form of ice nucleation protein (INP) consisting of N- and C-terminal domains. It is a membrane protein commonly used to display protein of interest on the cell surface (1).
rLvAPN1
LvAPN1, a protein from the aminopeptidase N family, was identified in Litopenaeus vannamei hemocytes as a receptor for VPAHPND toxin PirA and PirB, which can help the toxins pass through the cell membrane of hemocytes (2).
rLvAPN1 is a truncated form of LvAPN1 (residues 205-591) that composes of a crystal insecticidal (Cry) toxin binding region and the active site of peptidase-M1 domain, which was reported to directly bind to both PirA and PirB toxins (2). What’s more, there is no glycosylation site in rLvAPN1, which makes it easier to obtain the purified protein by using prokaryotic expression system (such as E. coli).
Usage and design
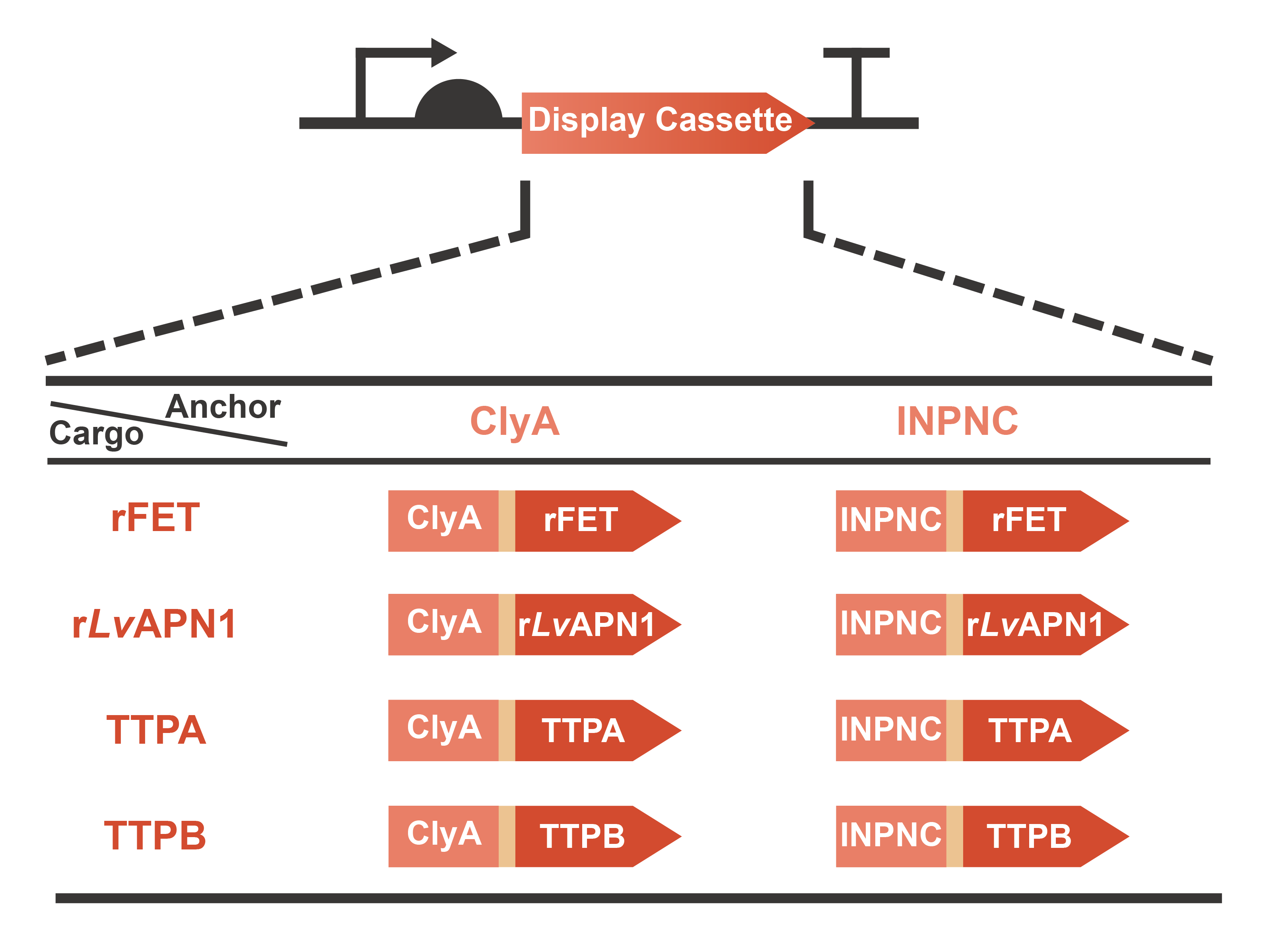
Engineering OMVs for treating and preventing AHPND caused by the pathogen V. parahaemolyticus are a significant part of OMEGA project (Operable Magic to Efficiently Getting over AHPND). Based on the efforts of our previous projects in 2020 (AnTea-Glyphosate) and 2021 (SALVAGE), we further developed the surface display system on the OMVs released by the engineered bacteria. The usage of cargo proteins were no more limited to enzymes that are usually utilized to catalyze series bio-chemical reactions, since some receptors or ligands involved in complex protein-protein interaction (PPI) were selected as the cargo candidates. This year, we chose two classic anchor proteins, ClyA and INPNC, to construct the display cassette with various cargo proteins including rFET (receptor), rLvAPN1 (receptor), TTPA (ligand) and TTPB (ligand) (Fig. 1). On one hand, with the receptors displayed, OMVs will gain the function of neutralizing toxins secreted by V. parahaemolyticus. On the other hand, with the assistance of ligands displayed on the surface, OMVs will become a special vector to deliver antimicrobials for the specific pathogen. In summary, we have taken a step closer to the collections of extracellular functional elements (EFE), combining the OMVs, secretion systems and surface display systems which we have been dedicated to since 2020. Learn more information from our Design page.
Fig. 1 Graphic description of the expression gene circuits for display cassette designed in OMEGA project.
For this part (INPNC-rLvAPN1-his), a His-tag (6×His) was added to the C-terminal of INPNC-rLvAPN1 to verify whether the rLvAPN1 protein is displayed on the surface of engineered bacteria or not. Arabinose-inducible system was used in the expression circuit of this part in pSB1C3 then composite part BBa_K4195133 was obtained. We transformed the constructed plasmid into E. coli BL21(DE3) for further verification of its location on the surface of E. coli.
Characterization
1. Identification
When constructing this circuit, colony PCR and gene sequencing were used to verify that the transformatants were correct. Target bands (3900 bp) can be observed at the position between 3000 and 5000 bp (Fig. 2).
Fig. 2 DNA gel electrophoresis of the colony PCR products of BBa_K4195133_pSB1C3.
2. Characterization of displaying cargo proteins
We used BBa_I0500 promoter and RBS (BBa_B0034) to express INPNC-rLvAPN1-his protein in E. coli BL21(DE3). We used BBa_K4195134 which has no surface display system (like INPNC or ClyA) as negative control and BBa_K4195133 as positive control. The arabinose-induced overnight culture was then incubated with FITC-labeled anti-His-tag antibody to verify whether the display system is functional or not.
Fig. 3 The results of immunofluorescence to characterize the function of the display system (p = 0.0406).
The ratio of fluorescence intensity (λEx = 492 nm, λEm = 528 nm) to OD600 of positive control (culture was incubated with PirA-his or his-PirB) is higher than that of negative control (culture was incubated with 1×TBST) (Fig. 3), which indicates that our surface display system works well and the binding ability of rLvAPN1 to PirA-his and his-PirB is retained on the surface of bacteria.
Reference
1. E. van Bloois, R. T. Winter, H. Kolmar, M. W. Fraaije, Decorating microbes: surface display of proteins on Escherichia coli. Trends Biotechnol. 29, 79-86 (2011).
2. W. Luangtrakul et al., Cytotoxicity of Vibrio parahaemolyticus AHPND toxin on shrimp hemocytes, a newly identified target tissue, involves binding of toxin to aminopeptidase N1 receptor. PLoS Pathog. 17, e1009463 (2021).
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12INCOMPATIBLE WITH RFC[12]Illegal NheI site found at 469
Illegal NheI site found at 2152 - 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 72
Illegal NgoMIV site found at 682
Illegal NgoMIV site found at 1791
Illegal AgeI site found at 429
Illegal AgeI site found at 823 - 1000INCOMPATIBLE WITH RFC[1000]Illegal BsaI site found at 139
Illegal SapI site found at 2156
| None |