Difference between revisions of "Part:BBa K1073024"
| (13 intermediate revisions by 2 users not shown) | |||
| Line 24: | Line 24: | ||
'''iGEM Team Braunschweig 2013:''' Supernatant after cell disruption containing chromoproteins of eforRed (<partinfo>BBa_K1073022</partinfo>) and amilGFP. Both chromoproteins fluoresce when excited by UV-light. | '''iGEM Team Braunschweig 2013:''' Supernatant after cell disruption containing chromoproteins of eforRed (<partinfo>BBa_K1073022</partinfo>) and amilGFP. Both chromoproteins fluoresce when excited by UV-light. | ||
| + | |||
| + | <h1> Translation Enhancing 5'-UTR (Improvement by iGEM TU_Darmstadt 2019) </h1> | ||
| + | <html> | ||
| + | <script id="MathJax-script" async | ||
| + | src="https://2019.igem.org/wiki/index.php?title=Template:TU_Darmstadt/MathjaxJS&action=raw&ctype=text/javascript"></script> | ||
| + | |||
| + | <div class="container"> | ||
| + | <div class="row"> | ||
| + | <div class="col mx-2"> | ||
| + | <h3>Usage and Biology</h3> | ||
| + | <hr class="head"> | ||
| + | <p> | ||
| + | Improved version: <a | ||
| + | href="https://parts.igem.org/Part:BBa_K3187015" | ||
| + | target="_blank"> BBa_K3187015</a> | ||
| + | <br></br> | ||
| + | The part was improved in terms of its expression level, based on the insertion of a 5’-untranslated region (5’-UTR) upstream of the coding sequence. This 5'-UTR was adapted from iGEM Bielefeld 2015 ( <a | ||
| + | href="https://parts.igem.org/Part:BBa_K1758100" | ||
| + | target="_blank"> BBa_K1758100</a>) and is based on the research of Olins <i>et</i> al | ||
| + | <sup id="cite_ref-1" class="reference"> | ||
| + | <a href="#cite_note-1">[1] </a> | ||
| + | </sup> | ||
| + | and Takahashi <i>et</i> al. | ||
| + | <sup id="cite_ref-2" class="reference"> | ||
| + | <a href="#cite_note-2">[2] </a> | ||
| + | </sup>. | ||
| + | It contains the strong ribosomal binding site (RBS) g10-L from the T7 bacteriophage and a sequence that plays a role in the regulation of mRNA binding to and release from the 30S ribosomal subunit. The 5'-UTR therefore enhances the translation efficiency of the following coding sequence (CDS) (Fig. 1). | ||
| + | |||
| + | </p> | ||
| + | |||
| + | <img | ||
| + | class="img-fluid center" | ||
| + | src="https://2019.igem.org/wiki/images/6/62/T--TU_Darmstadt--RibosomLiberation.png" | ||
| + | style="max-width:80%" | ||
| + | /> | ||
| + | <div class="caption"> | ||
| + | <p> | ||
| + | <b> | ||
| + | <center> Figure 1: | ||
| + | </b> Schematic depiction of the composition and interaction of the enhancer sequence with the 30S ribosomal subunit described by Takahashi <i>et.</i> al. <sup id="cite_ref-2" class="reference"> | ||
| + | <a href="#cite_note-2">[2] </a> | ||
| + | </sup>. | ||
| + | </center> | ||
| + | </p> | ||
| + | </div> | ||
| + | |||
| + | <p> | ||
| + | The sequence of the translation enhancing 5’-UTR can be divided into the four main features listed below: | ||
| + | </p> | ||
| + | |||
| + | |||
| + | |||
| + | <img | ||
| + | class="img-fluid center" | ||
| + | src="https://2019.igem.org/wiki/images/f/f3/T--TU_Darmstadt--EnhancerInColor.png" | ||
| + | style="text= align; max-width:85%" | ||
| + | /> | ||
| + | |||
| + | |||
| + | <br></br> | ||
| + | <div class="container-noborders"> | ||
| + | <div class="table-responsive-sm"> | ||
| + | <table class="table table-light"> | ||
| + | <tr> | ||
| + | <th scope="col">Sequence</th> | ||
| + | <th scope="col">Function</th> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="color:#d59b9b; word-wrap:break-word;"> | ||
| + | AATAATTTTGTT<br />TTAACTTTAA | ||
| + | </td> | ||
| + | <td> | ||
| + | The T7 g10 leader sequence (first described by Olins <i>et</i> al<sup id="cite_ref-1" class="reference"> | ||
| + | <a href="#cite_note-1">[1] </a> | ||
| + | </sup>)increases the efficiency of translation initiation. This sequence contains the epsilon motif TTAACTTTA which enhances the binding of the mRNA to the 16S rRNA. | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="color:#a5c8c8;">poly-A</td> | ||
| + | <td> | ||
| + | Referring to Takahashi et al.<sup id="cite_ref-2" class="reference"> | ||
| + | <a href="#cite_note-2">[2] </a> | ||
| + | </sup> a spacer between the epsilon motive and the RBS improves the translation rate. | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="color:#698888;">GAAGGAG</td> | ||
| + | <td> | ||
| + | According to Karig <i>et</i> al.<sup id="cite_ref-3" class="reference"> | ||
| + | <a href="#cite_note-3">[3] </a> | ||
| + | </sup> and Lentini et. al<sup id="cite_ref-4" class="reference"> | ||
| + | <a href="#cite_note-4">[4] </a> </sup> a distance of 4-9 bases between RBS and start codon increases the translation efficiency. | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td style="color:#cc9966;">AATAATCT</td> | ||
| + | <td> | ||
| + | According to Lentini et. al<sup id="cite_ref-4" class="reference"> | ||
| + | <a href="#cite_note-4">[4] </a> </sup> an AT-rich composition between the RBS and the start codon results in the best expression results. | ||
| + | </td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | </div> | ||
| + | </div> | ||
| + | |||
| + | <h3>Results</h3> | ||
| + | <hr class="head"> | ||
| + | <p> | ||
| + | <b>Fig. 2</b> shows the original version of the part (left) and the improved version (right) after overnight cultivation on an agar plate and after cultivation for 24 h in M9 minimal medium for better visualization of the emitted light at 512 nm<sup id="cite_ref-5" class="reference"> | ||
| + | <a href="#cite_note-5">[5] </a> </sup>. Both the agar plate and the liquid culture were supplemented with chloramphenicol according to the cmp resistance on the pSB1C3 backbone. Both pictures were taken using a dark reader (Dark Reader Clare Chemical Research) for better visualization of the yellow color. | ||
| + | </p> | ||
| + | <br></br> | ||
| + | |||
| + | <img | ||
| + | class="img-fluid center" | ||
| + | src="https://2019.igem.org/wiki/images/0/0a/T--TU_Darmstadt--amilGFPComparison.png" | ||
| + | style="max-width:90%" | ||
| + | /> | ||
| + | <div class="caption"> | ||
| + | <p> | ||
| + | <b> | ||
| + | <center> Figure 2: | ||
| + | </b><i>E. coli</i> BL21 DE3 transformed with BBa_K1073024 after cultivation on LB-agar (left) and in a liquid culture (M9 minimal medium) supplemented with cmp. The improved version of the part shows increased amilGFP expression </center> | ||
| + | </p> | ||
| + | </div> | ||
| + | <br></br> | ||
| + | <p> | ||
| + | Both the agar plate and the liquid culture show an enhanced expression of the amilGFP gene after the upstream insertion of the translation enhancing 5’-UTR. | ||
| + | </p> | ||
| + | |||
| + | <h2>References</h2> | ||
| + | <ol class="references"> | ||
| + | <li id="cite_note-1"> | ||
| + | <span class="mw-cite-backlink"> | ||
| + | <a href="#cite_ref-1">↑</a> | ||
| + | </span> | ||
| + | <span class="reference-text"> | ||
| + | Peter O. Olins and Shaukat H. Rangwala, A Novel Sequence Element Derived from Bacteriophage T7 mRNA Acts as an Enhancer of Translation of the lacZ Gene in Escherichia coli, The Journal of Biological Chemistry, Vol. 264, No. 29, Issue of Ovtobet 15, pp. 16973-16976, 1989 | ||
| + | <a rel="nofollow" class="external autonumber" href=" https://www.ncbi.nlm.nih.gov/pubmed/2676996" | ||
| + | target="_blank">[1] </a> | ||
| + | </span> | ||
| + | </li> | ||
| + | |||
| + | <li id="cite_note-2"> | ||
| + | <span class="mw-cite-backlink"> | ||
| + | <a href="#cite_ref-2">↑</a> | ||
| + | </span> | ||
| + | <span class="reference-text"> | ||
| + | Shuntaor Takahashi, Hiroyuki Furusawa, Takuya Ueda and Yoshio Okahata, Translation Enhancer Improves the Ribosome Liberation from Translation Initiation, J. Am. Chem. Soc. 2013, 135 35, 13096-13106 | ||
| + | <a rel="nofollow" class="external autonumber" | ||
| + | href=" https://www.ncbi.nlm.nih.gov/pubmed/23927491 | ||
| + | " target="_blank">[2] | ||
| + | </a> | ||
| + | </span> | ||
| + | </li> | ||
| + | |||
| + | <li id="cite_note-3"> | ||
| + | <span class="mw-cite-backlink"> | ||
| + | <a href="#cite_ref-3">↑</a> | ||
| + | </span> | ||
| + | <span class="reference-text"> | ||
| + | David K. Karig, Sukanya Iyer, Michael L. Simpson and Mitchel J. Doktycz, Expression optimization and synthetic gene networks in cell-free systems, Nucleic Acids Res. 2012 Apr; 40(8): 3763.3774 | ||
| + | <a rel="nofollow" class="external autonumber" | ||
| + | href="https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3333853/" target="_blank">[3] | ||
| + | </a> | ||
| + | </span> | ||
| + | </li> | ||
| + | |||
| + | <li id="cite_note-4"> | ||
| + | <span class="mw-cite-backlink"> | ||
| + | <a href="#cite_ref-4">↑</a> | ||
| + | </span> | ||
| + | <span class="reference-text"> | ||
| + | Roberta Lentini, Silvia Perez Santero, Fabio Chizzolini, Dario Cecchi, Jason Fontana, Marta Marchioretto, Christina Del Bianco, Jessica L. Terrell, Amy C. Spencer, Laura Martini, Michele Forlin, Michael Assfalg, Mauro Dalla Serra, William E. Bentley and Sheref S. Mansy, Integrating artificial with natural cells to translate chemical messages that direct E. coli behavior, Nature Communications 5, Article number: 4012 (2014) | ||
| + | <a rel="nofollow" class="external autonumber" | ||
| + | href=" https://www.nature.com/articles/ncomms5012 " | ||
| + | target="_blank">[4] </a> | ||
| + | </span> | ||
| + | </li> | ||
| + | |||
| + | <li id="cite_note-5"> | ||
| + | <span class="mw-cite-backlink"> | ||
| + | <a href="#cite_ref-5">↑</a> | ||
| + | </span> | ||
| + | <span class="reference-text"> | ||
| + | Joesfine Liljeruhm, Saskia K. Funk, Sandra Tietscher, Anders D. Edlund, Sabri Jamal, Pikkei Wistrand-Yuen, Karl Dyrhage, Arvid Gynna, Katarina Ivermark, Jessica Lövgren, Viktor Törnblom, Anders Virtanen, Erik R. Lundin, Erik Wistrand-Yuen and Anthony C. Forster, Enginerring a palette of eukaryotic chromoproteins for bacterial synthetic biology, Journal of Biological Engineering 12, Article number: 8 (2018) | ||
| + | |||
| + | <a rel="nofollow" class="external autonumber" | ||
| + | href=" https://jbioleng.biomedcentral.com/articles/10.1186/s13036-018-0100-0 " | ||
| + | target="_blank">[5] </a> | ||
| + | </span> | ||
| + | </li> | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | </ol> | ||
| + | |||
| + | </div> | ||
| + | </div> | ||
| + | </div> | ||
| + | </div> | ||
| + | <br></br> | ||
| + | </html> | ||
| + | |||
| + | |||
| + | ==Nanjing_High_School 2019's characterization== | ||
| + | ==BBa_K1073024 Constitutively expressed chromoprotein amilGFP== | ||
| + | <p>The level of fluorescent protein expression in a unit of bacterial cells changes with cell growth. We investigated this phenomenon by measuring the growth of cells and the fluorescence value of cell unit OD600 over time.</p> | ||
| + | <p>1. We transformed the plasmid contanining BBa_K1073024 into bacterial competent cells, plated and cultured overnight at 37 °C.</p> | ||
| + | <p>2. On the evening of the next day, we picked a single clone into a tube containing 5 ml LB which is called 0h. We culture bacterial cells at 37 ° C, 220 rpm. The sampling time points are 24h and 30h. </p> | ||
| + | <p>3. We took 100ul of the bacteria culture and measured the total fluorescence with a Thermo fluorescence microplate reader. We took 200ul of the ten-fold diluted bacterial solution and measured the OD600 with BioTek optical microplate reader. Total fluorescence is divided by OD600 to obtain fluorescence value per OD600.</p> | ||
| + | [[Image:K1073024-1.png|400px|Characterization of popular BioBrick RBSs]] | ||
| + | |||
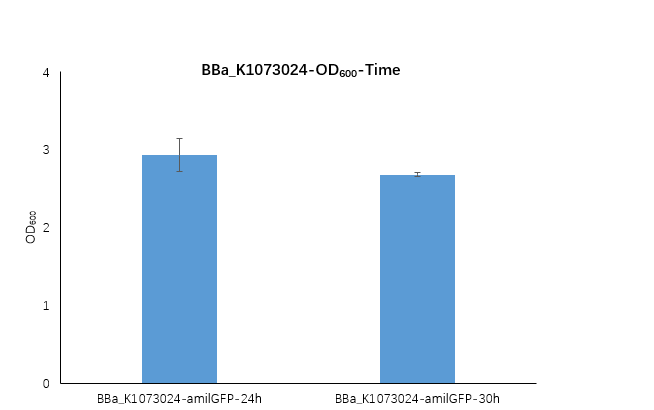
| + | <strong>Figure1. BBa_K1073024 containing strain OD600 at 24h and 30h.</strong> | ||
| + | <p>We measured the OD600 of the strain at 24h and 30h, respectively. The OD600 at 24h is 2.98 ,which is slightly higher than the OD600 2.68 at 30h.</p> | ||
| + | [[Image:K1073024-2.png|400px|Characterization of popular BioBrick RBSs]] | ||
| + | |||
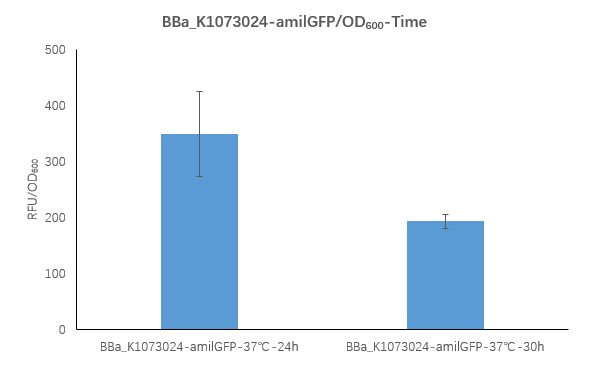
| + | <strong>Figure2. BBa_K1073024 containing strain Fluorescence per OD600 at 24h and 30h.</strong> | ||
| + | <p>We measured the RFU/OD600 of the strain at 24h and 30h, respectively. The RFU/OD600 at 24h is 348.4 ,which is higher than the RFU/OD600 192.9 at 30h.</p> | ||
| + | |||
| + | <p>These results indicate that the bacterial strain in the medium reached the stationary phase at 24h and 30h.</p> | ||
| + | <p>During the stationary phase, the strain dies, causing the number of cells to gradually decrease. The protein in the cell will only degrade, but will not be newly expressed, resulting in a decrease in protein levels.</p> | ||
| + | |||
| + | ==Shanghai_High_School 2019’s Characterisation== | ||
| + | ==BBa_K1073024 Constitutively expressed chromoprotein amilGFP== | ||
| + | [[Image:K1073024-S1.png|400px|Characterization of popular BioBrick RBSs]] | ||
| + | |||
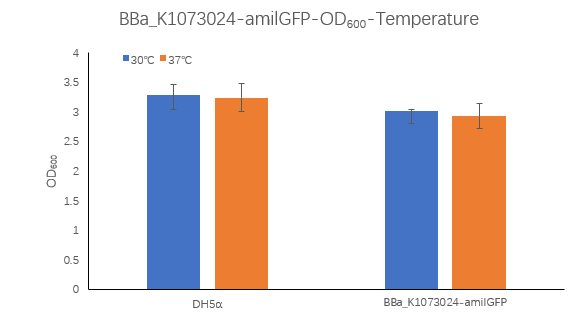
| + | <strong>Figure1. BBa_K1073024 containing strain OD600 at 30℃ and 37℃. </strong> | ||
| + | <p>The OD600 of blank control DH5α cells grown at 30 ° C and 37 ° C were 3.285 and 3.245, respectively, which is very close. The OD600 of the strain containing BBa_K1073024 grown at 30 ° C was 3.015, which was higher than the OD6002.93 at 37 ° C.</p> | ||
| + | [[Image:K1073024-S3.png|400px|Characterization of popular BioBrick RBSs]] | ||
| + | |||
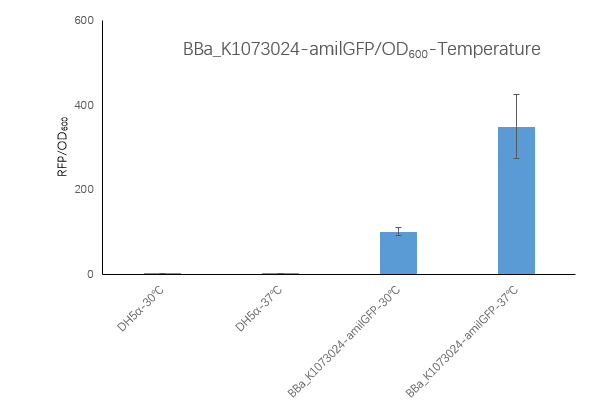
| + | <strong>Figure2. BBa_K1073024 containing strain Fluorescence per OD600 at 30℃ and 37℃. </strong> | ||
| + | <p>We measured the RFU/OD600 of the strain at 30℃and 37℃, respectively. The DH5α RFU/OD600 at 24h and 30h is close to zero. The Bba_K516132 containing strain</p> | ||
| + | |||
| + | <p>RFU/OD600 is 348.42 at 37℃,which is higher than 100.71 at 30℃.</p> | ||
| + | |||
| + | <p>To sum up: the growth OD600 of the BBa_K1073024-amilGFP strain is similar to the control DH5α, and this strain should have reached a stable phase at 24 h. The B BBa_K1073024-amilGFP strain has a much higher fluorescence at 37 ° C than 30 ° C. | ||
| + | These results indicate that the BBa_K1073024-amilGFP protein may be expressed more or more stably at 37 °C, which also means that it may be necessary to find a suitable culture temperature when expressing an unknown protein.</p> | ||
Latest revision as of 10:42, 20 October 2019
Constitutively expressed chromoprotein amilGFP
This device is an enhanced construct of 2012 Uppsala's BBa_K592010. It consists of a strong promoter (BBa_J23100) combined with a RBS (BBa_B0032) and the encoding region of the chromoprotein amilGFP (BBa_K592010). This results in a constitutively high expression of amilGFP.
Usage and Biology
During cultivation, the chromoprotein amilGFP is expressed and stored intracellularly. The yellow color of the protein can be seen by the naked eye, thus the chromoprotein can be used as a reporter or selection marker to distinguish cells easily.
iGEM Team Braunschweig 2013:
Left: Liquid cultures of E. coli XL1 expressing chromoproteins amilGFP , eforRed (BBa_K1073022) and aeBlue (BBa_K1073020).
Right: Cell pellet of E. coli XL1 expressing amilGFP.
The yellow color of amilGFP can be seen by the naked eye. The chromoprotein also fluoresce when excited. Thus it can be used to identify cells with fluorescence microscopy or flow cytometry. For further information as well as absorption and emission spectra see experience page.
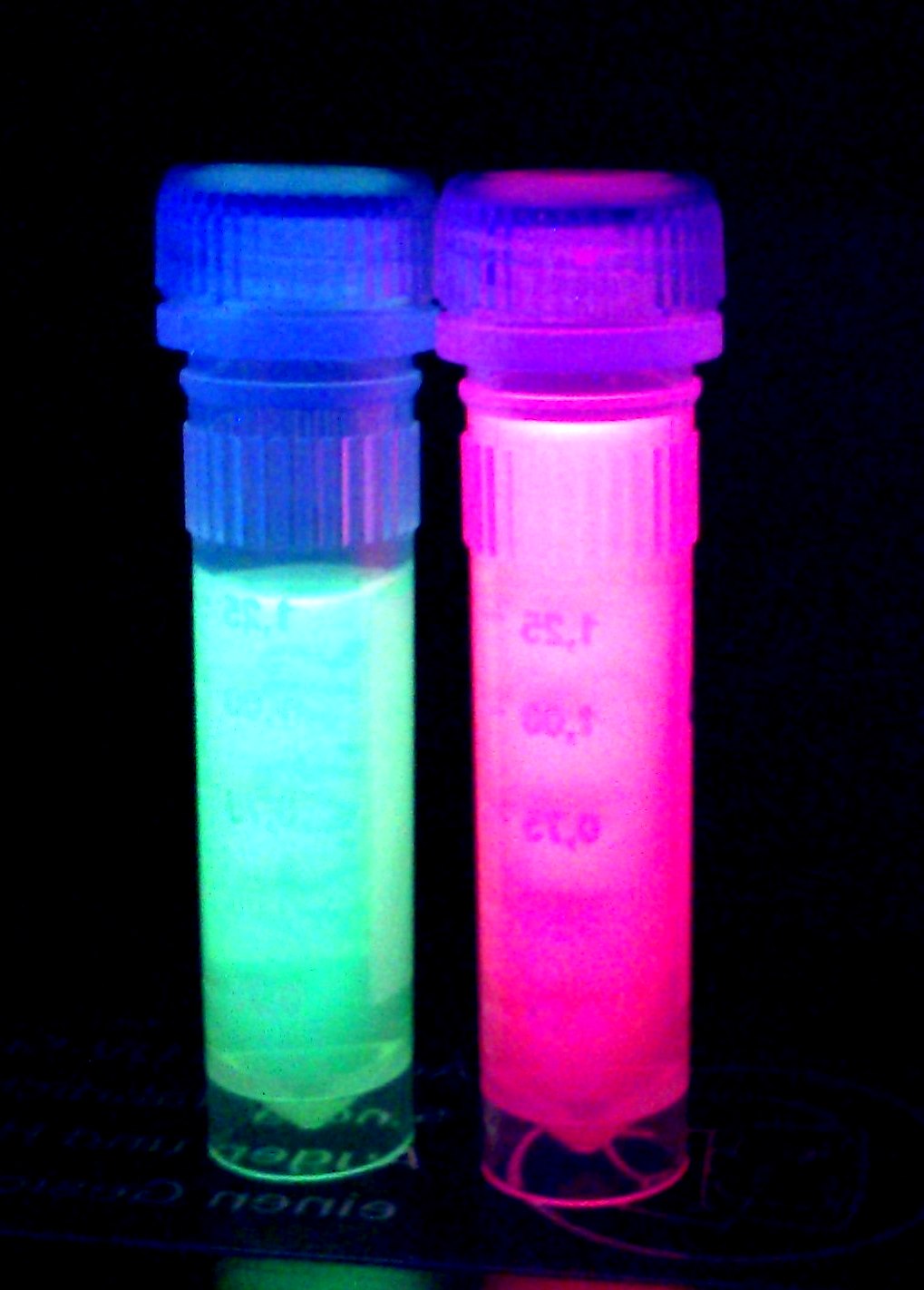
iGEM Team Braunschweig 2013: Supernatant after cell disruption containing chromoproteins of eforRed (BBa_K1073022) and amilGFP. Both chromoproteins fluoresce when excited by UV-light.
Translation Enhancing 5'-UTR (Improvement by iGEM TU_Darmstadt 2019)
Usage and Biology
Improved version: BBa_K3187015
The part was improved in terms of its expression level, based on the insertion of a 5’-untranslated region (5’-UTR) upstream of the coding sequence. This 5'-UTR was adapted from iGEM Bielefeld 2015 ( BBa_K1758100) and is based on the research of Olins et al
[1]
and Takahashi et al.
[2]
.
It contains the strong ribosomal binding site (RBS) g10-L from the T7 bacteriophage and a sequence that plays a role in the regulation of mRNA binding to and release from the 30S ribosomal subunit. The 5'-UTR therefore enhances the translation efficiency of the following coding sequence (CDS) (Fig. 1).
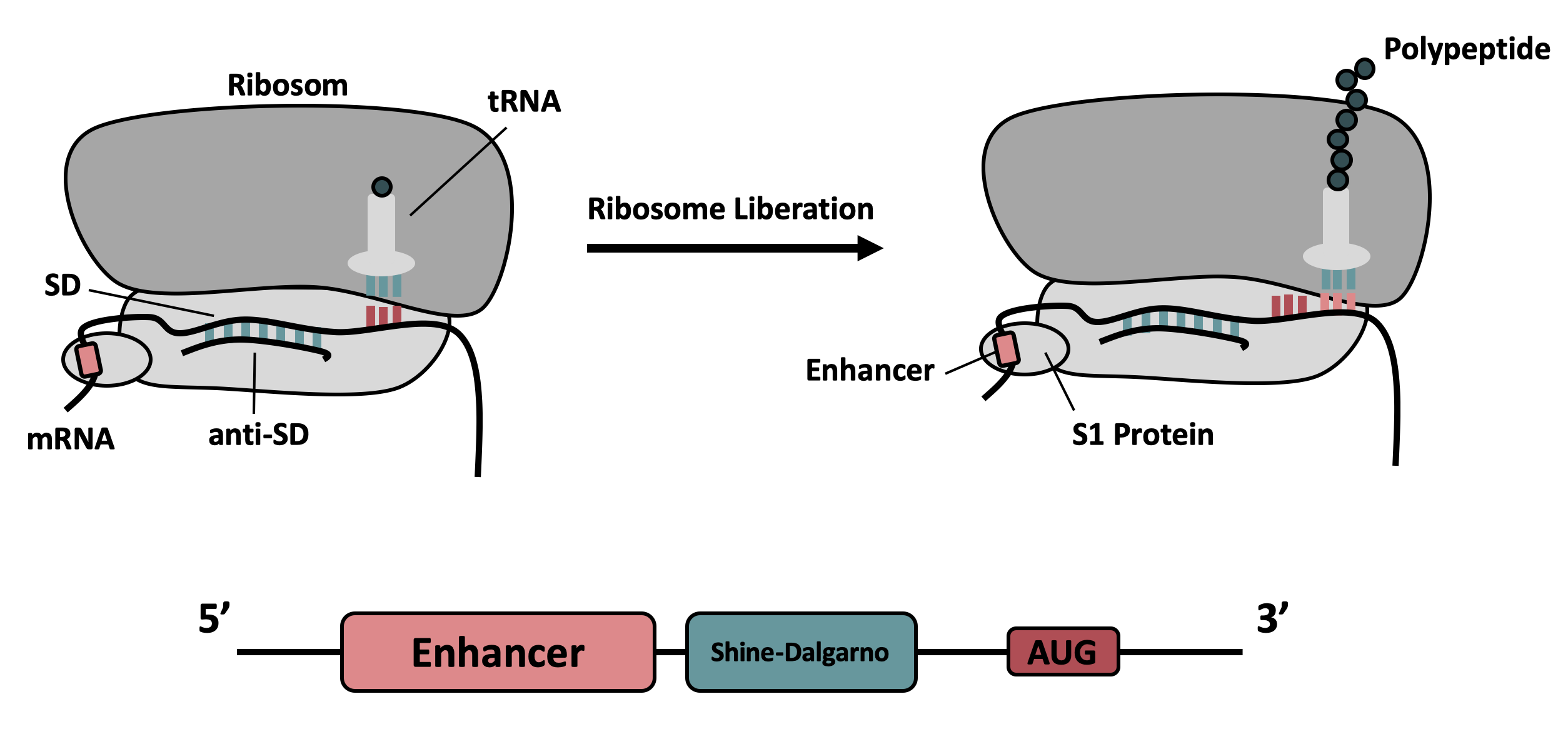
The sequence of the translation enhancing 5’-UTR can be divided into the four main features listed below:

| Sequence | Function |
|---|---|
|
AATAATTTTGTT TTAACTTTAA |
The T7 g10 leader sequence (first described by Olins et al [1] )increases the efficiency of translation initiation. This sequence contains the epsilon motif TTAACTTTA which enhances the binding of the mRNA to the 16S rRNA. |
| poly-A | Referring to Takahashi et al. [2] a spacer between the epsilon motive and the RBS improves the translation rate. |
| GAAGGAG | According to Karig et al. [3] and Lentini et. al [4] a distance of 4-9 bases between RBS and start codon increases the translation efficiency. |
| AATAATCT | According to Lentini et. al [4] an AT-rich composition between the RBS and the start codon results in the best expression results. |
Results
Fig. 2 shows the original version of the part (left) and the improved version (right) after overnight cultivation on an agar plate and after cultivation for 24 h in M9 minimal medium for better visualization of the emitted light at 512 nm [5] . Both the agar plate and the liquid culture were supplemented with chloramphenicol according to the cmp resistance on the pSB1C3 backbone. Both pictures were taken using a dark reader (Dark Reader Clare Chemical Research) for better visualization of the yellow color.

Both the agar plate and the liquid culture show an enhanced expression of the amilGFP gene after the upstream insertion of the translation enhancing 5’-UTR.
References
- ↑ Peter O. Olins and Shaukat H. Rangwala, A Novel Sequence Element Derived from Bacteriophage T7 mRNA Acts as an Enhancer of Translation of the lacZ Gene in Escherichia coli, The Journal of Biological Chemistry, Vol. 264, No. 29, Issue of Ovtobet 15, pp. 16973-16976, 1989 [1]
- ↑ Shuntaor Takahashi, Hiroyuki Furusawa, Takuya Ueda and Yoshio Okahata, Translation Enhancer Improves the Ribosome Liberation from Translation Initiation, J. Am. Chem. Soc. 2013, 135 35, 13096-13106 [2]
- ↑ David K. Karig, Sukanya Iyer, Michael L. Simpson and Mitchel J. Doktycz, Expression optimization and synthetic gene networks in cell-free systems, Nucleic Acids Res. 2012 Apr; 40(8): 3763.3774 [3]
- ↑ Roberta Lentini, Silvia Perez Santero, Fabio Chizzolini, Dario Cecchi, Jason Fontana, Marta Marchioretto, Christina Del Bianco, Jessica L. Terrell, Amy C. Spencer, Laura Martini, Michele Forlin, Michael Assfalg, Mauro Dalla Serra, William E. Bentley and Sheref S. Mansy, Integrating artificial with natural cells to translate chemical messages that direct E. coli behavior, Nature Communications 5, Article number: 4012 (2014) [4]
- ↑ Joesfine Liljeruhm, Saskia K. Funk, Sandra Tietscher, Anders D. Edlund, Sabri Jamal, Pikkei Wistrand-Yuen, Karl Dyrhage, Arvid Gynna, Katarina Ivermark, Jessica Lövgren, Viktor Törnblom, Anders Virtanen, Erik R. Lundin, Erik Wistrand-Yuen and Anthony C. Forster, Enginerring a palette of eukaryotic chromoproteins for bacterial synthetic biology, Journal of Biological Engineering 12, Article number: 8 (2018) [5]
Nanjing_High_School 2019's characterization
BBa_K1073024 Constitutively expressed chromoprotein amilGFP
The level of fluorescent protein expression in a unit of bacterial cells changes with cell growth. We investigated this phenomenon by measuring the growth of cells and the fluorescence value of cell unit OD600 over time.
1. We transformed the plasmid contanining BBa_K1073024 into bacterial competent cells, plated and cultured overnight at 37 °C.
2. On the evening of the next day, we picked a single clone into a tube containing 5 ml LB which is called 0h. We culture bacterial cells at 37 ° C, 220 rpm. The sampling time points are 24h and 30h.
3. We took 100ul of the bacteria culture and measured the total fluorescence with a Thermo fluorescence microplate reader. We took 200ul of the ten-fold diluted bacterial solution and measured the OD600 with BioTek optical microplate reader. Total fluorescence is divided by OD600 to obtain fluorescence value per OD600.
Figure1. BBa_K1073024 containing strain OD600 at 24h and 30h.
We measured the OD600 of the strain at 24h and 30h, respectively. The OD600 at 24h is 2.98 ,which is slightly higher than the OD600 2.68 at 30h.
Figure2. BBa_K1073024 containing strain Fluorescence per OD600 at 24h and 30h.
We measured the RFU/OD600 of the strain at 24h and 30h, respectively. The RFU/OD600 at 24h is 348.4 ,which is higher than the RFU/OD600 192.9 at 30h.
These results indicate that the bacterial strain in the medium reached the stationary phase at 24h and 30h.
During the stationary phase, the strain dies, causing the number of cells to gradually decrease. The protein in the cell will only degrade, but will not be newly expressed, resulting in a decrease in protein levels.
Shanghai_High_School 2019’s Characterisation
BBa_K1073024 Constitutively expressed chromoprotein amilGFP
Figure1. BBa_K1073024 containing strain OD600 at 30℃ and 37℃.
The OD600 of blank control DH5α cells grown at 30 ° C and 37 ° C were 3.285 and 3.245, respectively, which is very close. The OD600 of the strain containing BBa_K1073024 grown at 30 ° C was 3.015, which was higher than the OD6002.93 at 37 ° C.
Figure2. BBa_K1073024 containing strain Fluorescence per OD600 at 30℃ and 37℃.
We measured the RFU/OD600 of the strain at 30℃and 37℃, respectively. The DH5α RFU/OD600 at 24h and 30h is close to zero. The Bba_K516132 containing strain
RFU/OD600 is 348.42 at 37℃,which is higher than 100.71 at 30℃.
To sum up: the growth OD600 of the BBa_K1073024-amilGFP strain is similar to the control DH5α, and this strain should have reached a stable phase at 24 h. The B BBa_K1073024-amilGFP strain has a much higher fluorescence at 37 ° C than 30 ° C. These results indicate that the BBa_K1073024-amilGFP protein may be expressed more or more stably at 37 °C, which also means that it may be necessary to find a suitable culture temperature when expressing an unknown protein.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12INCOMPATIBLE WITH RFC[12]Illegal NheI site found at 7
Illegal NheI site found at 30 - 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]