Part:BBa_K192000
Contents
Encapsulin
Toronto 2009
BBa_K192000 is a shell-forming protein known as "encapsulin", which we derived from the TM0785 plasmid in T. maritima. The protein is a monomer which assembles into a thin icosahedral shell with a diameter of 240 angstroms resulting in a nanocompartment lined with conserved binding sites for short polypeptide tags present as C-terminal extensions of enzymes. In T. maritima, these enzymes are involved in oxidative-stress response. We have isolated the encapsulin gene in E. coli, in which we plan to characterize as an expression system for the application of channeling candidate enzyme pairs identified in silico.
Improvement from iGEM EPFL 2018
Overview
The 2018 EPFL iGEM team has improved upon this part by using a different sequence derived from a plasmid used in David Savage's lab (Cassidy-Armstutz et al., 2016). Additionally the team introduced a HexaHistidine loop (GGGGGGHHHHHHGGGGG) into the protein's sequence to improve the heat resistance and to provide better hydrodynamic properties (Moon et al., 2014). The composite part BBa_K2686005 encodes this modified Encapsulin sequence and presents an upgrade due to BsaI cut sites and sfGFP under a native promoter which allow for the rapid introduction of antigen encoding sequences to the Encapsulin monomer's C terminus.
Purification
After having tested a variety of purification procedures, heat purification at 70C for 20 minutes followed by cooling on ice for 15 minutes and a subsequent centrifugation at 12000g for 10 minutes was found to be the most efficient way of isolating the encapsulin (encapsulin ends up in supernatant).
Expression of BBa_K2686005 precursors compared to BBa_K192000
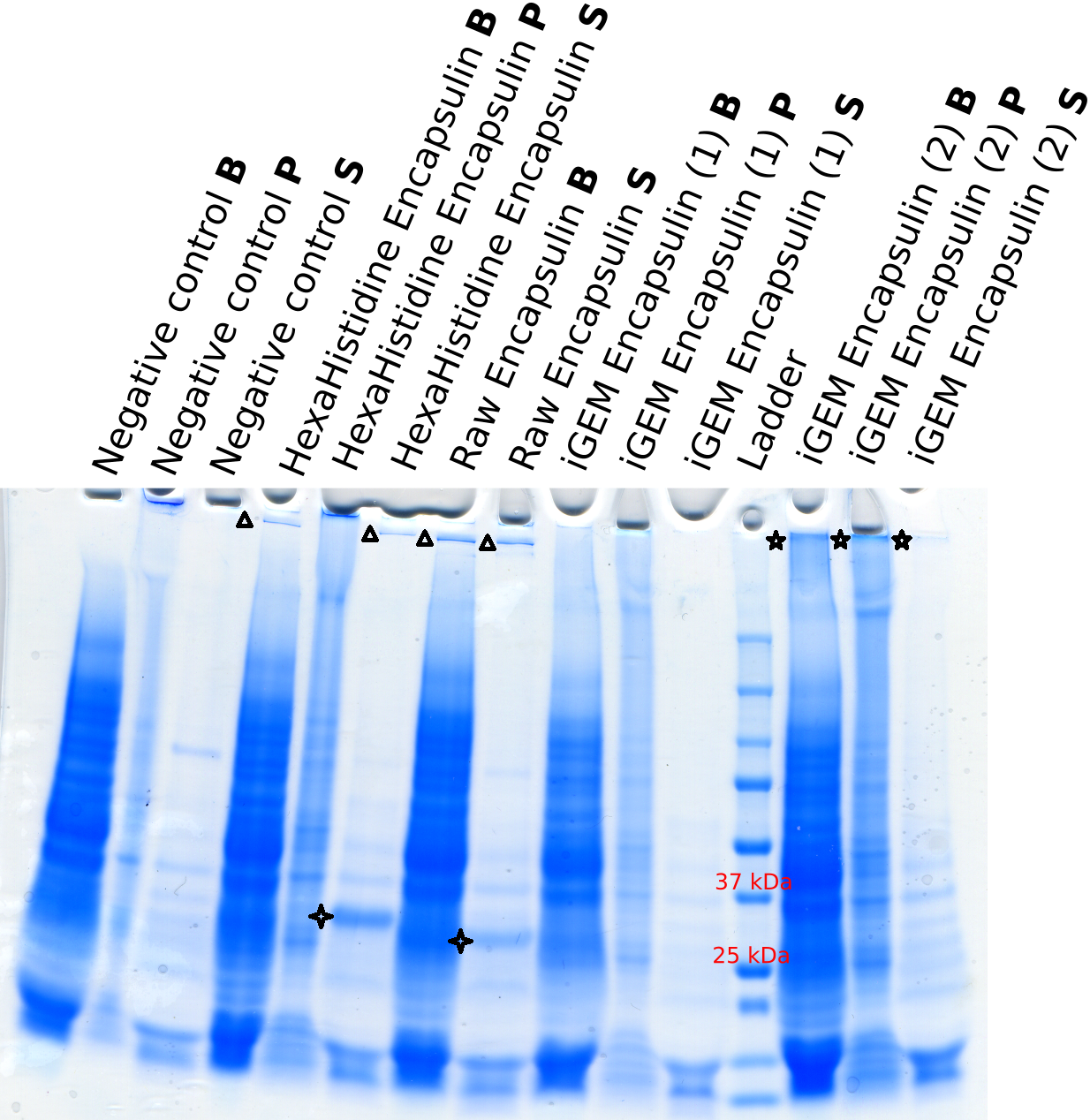
Conclusion
We were unable to prove for certain that BBa_K192000 was successfully expressed in cell free and that the part assembles to a 60-mer.
| None |
