Part:BBa_K801070
xanthosine methyltransferase CaXMT1-strep
This part is based upon the mRNA sequences having been isolated out of coffea arabica by [http://www.ncbi.nlm.nih.gov/pubmed/12746542/ Uefuji et al.], 2003, and registered at [http://www.ncbi.nlm.nih.gov/nuccore/AB048794.1/ NCBI]. The sequence was modified in several ways, to make it iGEM compatible and improve the usage in general, respectively.
This part is characterized in Saccharomyces cerevisiae by having cloned it into a yeast shuttle vector (pTUM104) and the expression was proved successfully by SDS-PAGE and western blot analysis.
Background and principles
Caffeine is a purine-alkaloid and its biosynthesis occurs in coffee plants and tea plants. Its chemical structure is similar to that of the ribonucleoside adenosine. Hence it can block specific receptors in the hypothalamus. Adenosine binding leads to decreased neurotransmitter-release and therefore decreased neuron activity. This induces sleep and thus avoids overexertion of the brain. Since caffeine antagonizes adenosine and increases neuronal activity, it is used as a means to stay awake. On average, one cup (150 ml) of coffee contains about 50 - 130 mg caffeine and one cup of tea 25 - 90 mg. At higher doses (1g), however, caffeine leads to higher pulse rates and hyperactivity. Moreover, caffeine was shown to decrease the growth of E. Coli and yeast reversibly as of a concentration of 0.1 % by acting as a mutagen (Putrament et al., On the Specificity of Caffeine Effects, MGG, 1972), but previous caffeine synthesis experiments (see below) have only led to a concentration of about 5 µg/g (per g fresh weight of tobacco leaves), so it is not expected to reach critical concentrations and the amounts of caffeine in coffee or tea (leading to physiological effects) is usually a little bit lower.
Usage and Biology
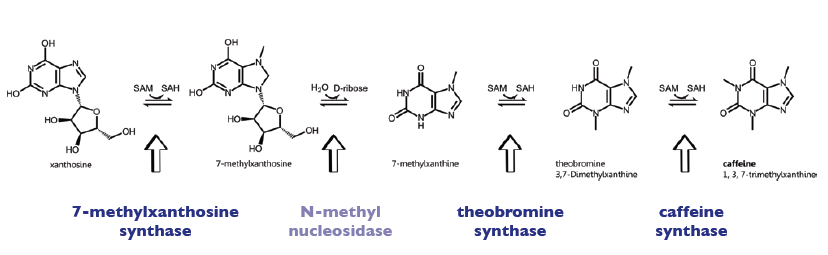
The enzyme CaXMT1 (xanthosine N-methyltransferase 1 of coffea arabica) catalyses the first reaction step of the caffeine biosynthesis pathway and convertes the sustrate Xanthosine into 7-Methylxanthosine. It uses SAM als methyl-donor and is located in the cytoplasm of the plants. Furthermore it exists as homodimer, being also able to form heterodimers with the other enzymes of the caffeine pathway (see [http://www.brenda-enzymes.info Brenda]). Besides, the enzyme CaXMT1 is highly imaginable to work in a bifunctional manner by catalyzing both, the methylation of the initial substrate xanthosine and the removal of the ribose moiety, thereby creating the intermediate 7-methylxanthine (which is the substrate for the second methyltransferase). A potential mechanism has been demonstrated by http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1914188/ McCarthy and McCarthy, 2007. As a matter of fact, a single N-methyl nucleosidase, as it is shown on the depicted pathway below, has indeed been partially purified out of tea leaves http://ci.nii.ac.jp/naid/110006323439/ Negishi et al. (1988), but neither the native enzyme nor its DNA have ever been isolated http://www.ncbi.nlm.nih.gov/pubmed/18068204 Ashihara et al., 2008.
Modifications
- the 5' UTR and 3' UTR of the original sequences were removed
- the yeast consensus sequence for improved ribosome binding (TACACA) was added 5' of the start codon ATG
- according to N- end rule and the yeast consensus sequence for improved ribosome binding, the first triplet after ATG (GAG) was exchanged with TCT (serine), to optimize both, protein stability and mRNA translation. This decision was made after proofing the 3D- structure of the enzyme CaDXMT1. Due to the fact, that the first two residues of the amino acid sequence are not shown in the crystalized structure (probably because of high flexibility), we chose to exchange this amino acid, because it is probably not necessary for the uptake of the ligands ([http://www.uniprot.org/uniprot/Q9AVK0 uniprot] entry further shows, that it is not immediately involved in ligand binding). Because of the high similarity of the enzyme sequences, we also exchanged this amino acid.
- we added a c- terminal strep-tag for purification and detection
- the remaining coding sequence was extended with the standard RFC10 prefix and suffix, respectively
- at last we made an optimization of the coding sequences with respect to the codon usage and mRNA structures
- remove of all critical restriction sites (RFC10 and RFC25)
Note: Because of the yeast consensus sequence, this coding part does not start with ATG!
Biosynthesis Pathway
Characterization
Cloning into pSB1C3
In order to submit the enzyme xanthosine methyltransferase (CaXMT1) to the parts.igem, we cloned the generated sequence into pSB1C3, making the system RFC10 compatible. However, since the sequence does not contain any restriction sites of the RFC25 standard (NgoMIV and Age1), RFC25 compatibility can easily be reached without required quick changes by the use of PCR upon usage of appropriate primers.
To check the successful cloning, we performed an analytical digest with XbaI and PstI.
The expected lengths of the fragments were:
- Insert (CaXMT1): ca. 1158bp
- Backbone (pSB1C3): ca. 2050 bp
The picture on the left shows the analytical digest of BioBrick BBa_K801070 with Xba1 and Pst1, separated by gel-electrophoresis on 1% agarose gel upon usage of ethidium bromide.
Westernblot
Western blot of the crude extract after expression of Saccharomyces cerevisiae INVSc1 transformed with pTUM104_CaXMT1, pTUM104_CaMXMT1 and pYES2_eGFP cultivated in selective expression Sc minimal medium lacking uracil with 2% galactose as inducer at different times after start of expression.
| prestained protein ladder | eGFP (uninduced) | CaMXMT1 (uninduced) | CaXMT1 (uninduced) | eGFP (20 h) | CaMXMT1 (20 h) | CaXMT1 (20 h) | unstained protein ladder * |
* previously stained with ponceau's reagent
Note: Because of probable posttranslational modifications in yeast, the apparent weight of the protein differs from the theoretical weight.
[http://2d.bjmu.edu.cn/show2d/Proteomics%20tools.asp ExPASy Proteomics tools] predicts (amongst others) the following modifications:
- acetylation at serine (second amino acid)
- O-GlcNAc modifikation at several positions
- phosphorylation at several positions
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
References
- http://www.ncbi.nlm.nih.gov/pubmed/18068204 Ashihara et al., 2008 Ashihara, H., Sano, H., and Crozier, A. (2008). Caffeine and related purine alkaloids: biosynthesis, catabolism, function and genetic engineering. Phytochemistry, 69(4):841–56.
- http://www.ncbi.nlm.nih.gov/pubmed/22849837 Franco et al., 2012 Franco, L., Sánchez, C., Bravo, R., Rodriguez, A., Barriga, C., and Juánez, J. C. (2012). The sedative effects of hops (humulus lupulus), a component of beer, on the activity/rest rhythm. Acta Physiol Hung, 99(2):133–9.
- http://www.ncbi.nlm.nih.gov/pubmed/18036626 Kim and Sano, 2008 Kim, Y.-S. and Sano, H. (2008). Pathogen resistance of transgenic tobacco plants producing caffeine. Phytochemistry, 69(4):882–8.
- http://www.ncbi.nlm.nih.gov/pubmed/16925551 Kuranda et al., 2006 Kuranda, K., Leberre, V., Sokol, S., Palamarczyk, G., and François, J. (2006). Investigating the caffeine effects in the yeast Saccharomyces cerevisiae brings new insights into the connection between TOR, PKC and Ras/cAMP signalling pathways. Mol Microbiol, 61(5):1147–66.
- http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1914188/ McCarthy and McCarthy, 2007 McCarthy, A.A., McCarthy, J.G. (2007). The Structure of Two N-Methyltransferases from the Caffeine Biosynthetic Pathway. Plant Physiology, 144(2):879-889.
- http://ci.nii.ac.jp/naid/110006323439/ Negishi et al. (1988) Negishi O, Ozawa T and Imagawa H (1988). N-Methyl nucleosidase from tea leaves. Agric. Biol. Chem. 52: 169–175.
- http://www.ncbi.nlm.nih.gov/pubmed/12746542 Uefuji et al., 2003 Uefuji, H., Ogita, S., Yamaguchi, Y., Koizumi, N., and Sano, H. (2003). Molecular cloning and functional characterization of three distinct n-methyltransferases involved in the caffeine biosynthetic pathway in coffee plants. Plant Physiol, 132(1):372–80.
- http://www.ncbi.nlm.nih.gov/pubmed/16247553 Uefuji et al., 2005 Uefuji, H., Tatsumi, Y., Morimoto, M., Kaothien-Nakayama, P., Ogita, S., and Sano, H. (2005). Caffeine production in tobacco plants by simultaneous expression of three coffee n-methyltrasferases and its potential as a pest repellant. Plant Mol Biol, 59(2):221–7.
//cds/biosynthesis
//chassis/eukaryote/yeast
| None |