Part:BBa_K731500
[LacI + LacIq promoter] reverse + [tac promoter + lac operator] forward
This composite part consist of BBa_K731300 part (lacI-lacIq) and a tac promoter followed by a lac operator in the forward direction. It is a ready to use composite part and it allows the expression of a gene of interest with a strong E.coli promoter and under the control of IPTG. The lac operator decreases basal levels of expression.
This part is an improvement of BBa_K314111.
This part is in pSB1C3, but it is also available in pSB4K5 upon request. Contact us at igemtrento@gmail.com
Usage and Biology
LacI gene product is a trascriptional inhibitor inactivated by allolactose, a lactose metabolite. In its active conformation (no allolactose bound) it recognizes and binds lac operator sterically preventing the RNA polymerase binding to the tac promoter and owering constitutive gene expression. In its active conformation (allolactose bound) it releases lac operon, allowing polymerase recognition of Ptac. Protein expression can be induced with isopropyl β-D-1-thiogalactopyranoside (IPTG, an allolactose analogous).
This part allows IPTG-induced gene expression. lac operon and Ptac activity has been measured by placing a GFPmut3b under control of this regolative part and assaying protein level by fluorescence. See BBa_K731520 for sequence details.
This part has been characterized in both pSB4K5 and pSB1C3.
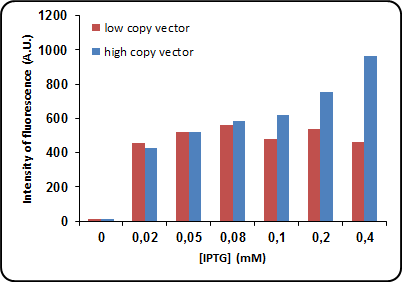
FIGURE 1 Dependence on protein expression of IPTG concentration. NEB10b cells transformed with BBa_K731520 both in low copy (pSB4K5) and high copy (pSB1C3) vectors. Cells were grown in LB at 37°C until OD of 0.6 and induced with different concentrations of IPTG. After 4 hours a 1.5 mL aliquot was taken from each sample. Cells were spun down and resuspended in 1.5 mL of PBS. Fluorescence measurements were taken with a Varian Cary Eclipse Spectrophotometer using an excitation wavelenght of 464 nm.
While in low copy vectors maximum protein expression is reached at low IPTG concentration such 0.05mM, high copy vectors show protein expression linearly proportional to strength of induction. This may be due the fact that lower IPTG concentrations are sufficient to trascriptionally activate all plasmid copies when plasmid concentration is small (i.e. when a low-copy vector such as pSB4K5 is used).
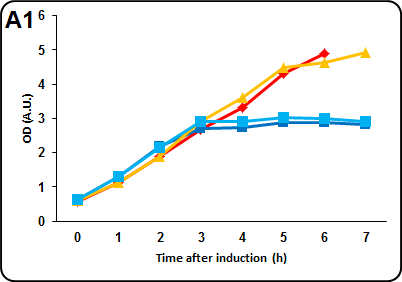
FIGURE 2 Effects of carbon source and induction on protein expression levels. Cells were grown at 37°C in LB until it was reached an OD of 0.4. The cells were at this point spun down and resuspended in an equal volume of MOPS medium and allowed to grow to an OD of 0.6. Prior induction the cells were splitted into two samples of equal volume and one of the two sample was induced with 0.1 mM IPTG. Every hour a 1.5 mL aliquot was taken to measure optical density, pelleted and stored overnight at 4°C. The following day samples were resuspended in 1.5 mL of PBS to measure fluorescence. In case of instrument saturation (A.U. > 1000) the volume of PBS was doubled. The assay was performed in two different MOPS media. MOPS A: 60 mM glycerol ("hot" colours). MOPS B: 30 mM glucose ("cold" colors). The experiment has been performed in triplicate. Panel A1& A2: Optical density at 600nm and intensity of fluorescence in pSB4K5. Please note that last MOPS A peak in this panel is not significative, since the instrument was saturated even at 1:2 diluition. Panel B1 & B2: Optical density at 600nm and intensity of fluorescence in pSB1C3. Fluorescence measurements were taken with a Varian Cary Eclipse Spectrophotometer using an excitation wavelenght of 464 nm.
Contribution: BNDS_China 2019
Group: BNDS_China iGEM 2019
Author: Wanji Li
Introduction & Design:
We observed better cell growth and protein expression when NEB10b cells were transformed with the low copy vector. We also noted a difference in cell growth and protein expression depending on carbon source, with glycerol-containing media allowing better growth. No fitness decrease has been observed when cells were induced with IPTG 0.1 mM. Difference in protein expression may be due inductor exclusion, which occurs when glucose may be used as a carbon source instead of lactose. This is more evident in low copy-transformed NEB10b.
In our study, we aim to find an equation describing the relationship between the expression level of proteins and IPTG concentration and between the cell’s growth and the IPTG concentration. Besides, we also want to show the data in the extreme conditions. Therefore, we decided to do the experiment when the concentration of IPTG is extremely high.
We firstly set the IPTG concentration: 0.0mM, 0.1mM, 0.2mM, 0.3mM, 0.4mM, 0.5mM, 0.7mM, 0.9mM, 1.1mM, 1.3mM, 1.5mM, 1.7mM, 1.9mM, 2.1mM, 2.3mM, 2.5mM, 2.7mM. Then, with an initial OD value of 0.6, we add different amount of IPTG corresponding to the concentration mentioned previously to the liquid bacteria culture and reconcile the volume of bacteria culture to 100ul in 96-well plate. After that, through the measurement of plate reader, we got the data showing the relationship among time, cell’s growth, and IPTG concentration, shown in the three dimension pictures, FIGURE 3:

FIGURE 3data showing the relationship among time, cell’s growth, and IPTG concentration
By analyzing the trend of the mechanism, we create a differential equation that explains the relationship among time, cell’s growth, and IPTG concentration. Furthermore, by solving the equation and fitting the data we got from the experiment, the relationship among time, cell’s growth, and IPTG concentration are quantitatively shown below, by

FIGURE 4Equations that quantitatively show the relationship among time, cell’s growth, and IPTG concentration
Therefore, we get the conclusion that The result of programming instructs us that when the concentration of IPTG is 0.764906mM, E.coli could produce the largest amount of todD. The graphics of objective function shows that the producing rate of todD would slowing down significantly when time value approaches 50, which is 12.5 hours in the real world. To increase the efficiency of the lab and to save time, 12.5 hours of IPTG induction is enough for the production of todD. The fitting three dimension graphs are shown below, by FIGURE 5&6

FIGURE 5the fitting three dimension garphs

FIGURE 6the fitting three dimension graphs
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
| None |