Part:BBa_K4342011
pbpG Upstream
Introduction
The 2022 UT Austin iGEM Team’s Part Collection provides a number of DNA sequences and procedures for genetically engineering Acinetobacter baylyi ADP1. We were able to effectively engineer ADP1's genome using a two-step genetic engineering protocol. See the Engineering Page for more details on how we modified ADP1's genome. On this page, we explain how our part collection can be used alongside this two-step protocol to delete ADP1 genes, insert DNA sequences into any chromosomal location, and engineer an ADP1-based biosensor to detect any DNA sequence of interest.
We hope this part collection guides future iGEM teams in engineering ADP1 and utilizing ADP1’s flexibility to tackle any challenge in synthetic biology.
Categorization
For our parts collection, we categorize our parts into the following categories:
Upstream
An Upstream basic part is a DNA sequence directly upstream of a target gene. These basic parts are homology flanks that are used for ADP1 Genetic Engineering. Examples include the ACIAD2049 Upstream for P. destructans detector (BBa_4342003) and pbpG Upstream (BBa_4342011).
Downstream
A Downstream basic part is a DNA sequence directly downstream of a target gene. These basic parts are homology flanks that are used for ADP1 Genetic Engineering. Examples include ACIAD2049 Downstream for P. destructans detector (BBa_4342004) and pbpG Downstream (BBa_4342012).
Integration Cassettes
An "Integration" cassette is a composite part consisting of an "Upstream" basic part, the tdk/kan basic part (BBa_4342000), and a "Downstream" basic part. These parts are designed to use in the first transformation step in ADP1 Genetic Engineering. Examples include the ACIAD2049 Integration cassette (BBa_4342019) and the acrB Integration cassette (BBa_4342023).
Rescue Cassettes
"Rescue" cassette is a composite part consisting of an "Upstream" basic part, an optional genetic device, and a "Downstream" basic part. These parts are designed to use in the second transformation step in ADP1 Genetic Engineering. Examples include the ACIAD2049 Rescue cassette (BBa_4342020, Upstream + Downstream), the YFP Rescue cassette (BBa_4342030, Upstream + Genetic Device + Downstream), and the nptII Detector Rescue cassette (BBa_4342031, Upstream + Composite Part + Downstream).
Genetic Device
"Genetic Device" is a basic part that can be any DNA sequence to be integrated into ADP1. Examples include the CymR YFP (BBa_4342008) and the nptII Broken Gene (BBa_4342015).
We further categorize each part with a standardized Golden Gate Assembly (GGA) Type 1-8 Overhang [2]. Each type is ligated to a complementary type (ex. Type 2 can be ligated to Type 1 and Type 3). Moreover, some parts contain consecutive GGA Type numbers, such as Type 234. These DNA sequences start with a Type 2 Overhang and end with a Type 4 Overhang (ex. tdk/kan cassette (BBa_4342000).
Usage and Biology
pbpG is a nonessential gene in Acinetobacter baylyi ADP1 that codes for a protein involved in peptidoglycan synthesis, contributing to β-lactam antibiotic resistance [1]. Knocking out this gene allows for the integration of other DNA sequences in its chromosomal location. Using this part, we demonstrate that pbpG can be replaced with any DNA construct.
Design
The pbpG Upstream part comprises the 1035 bp homology directly upstream of the pbpG gene in ADP1. We designed optimized primers, which include GC contents of over 40% and melting temperatures of under 70 °C. BsaI and BsmBI restriction sites are attached to the 3’ end, which are designed to ligate to the 5' end of the tdk/kan cassette (BBa_4342000) and the pbpG Downstream part (BBa_4342012) respectively. See Figure 4 on the Engineering Page for more details on how to design primers containing the correct GGA Type Overhang and restriction sites.
This part contains a BsaI restriction site with a standard 4 bp GGA Type 2 Prefix [2] and a BsmBI restriction site with a 4 bp “rescue” complementary scar. See the Contribution page on our wiki for more details on GGA Type Overhangs. This design allows for easy ligation with any part that contains a complementary 4 bp GGA Type 2 Prefix (BsaI) or the same 4 bp “rescue” complementary scar.
Composite Parts
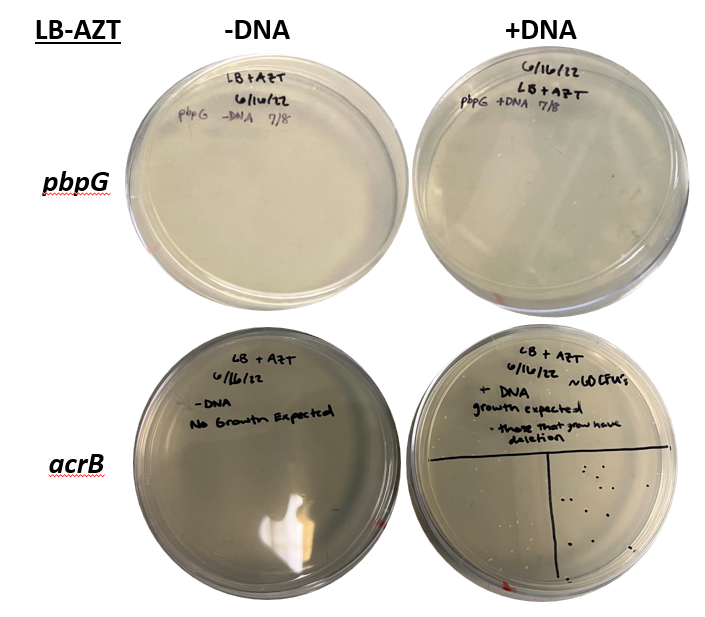
This basic part is used to assemble the pbpG integration cassette (BBa_4342021) and the pbpG rescue cassette (BBa_4342022)composite parts. Figures 1 and 2 show how these composite parts can be used in our two-step ADP1 Genetic Engineering protocol to create a minimal 4 bp scar in the deletion of the pbpG gene.
Step 1
This part is designed to ligate to the 5' end of the tdk/kan cassette, BBa_4342000, creating the pbpG- tdk/kan cassette composite part (BBa_4342021). This composite part allows for successful transformant selection on Kanamycin (Kan) via the kanR gene (Fig. 1).
Step 2
The tdk/kan cassette can subsequently be knocked out to create a scarless deletion of pbpG via BsmBI digestion, BBa_4342022. During this reaction, this part is ligated to the 5' end of the pbpG Downstream part BBa_4342012. This composite part serves as a “rescue” cassette to select for successful transformants on Azidothymidine (AZT) (Fig. 2).Characterization
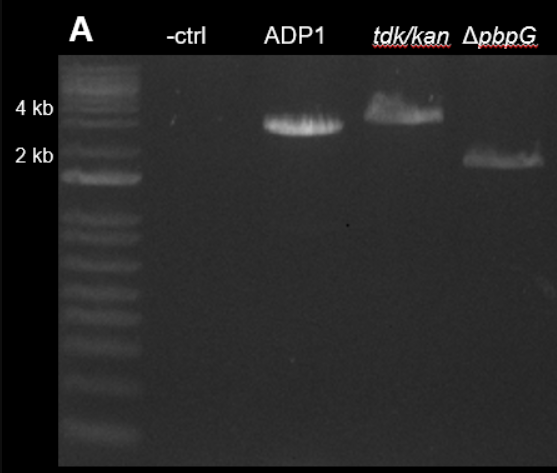
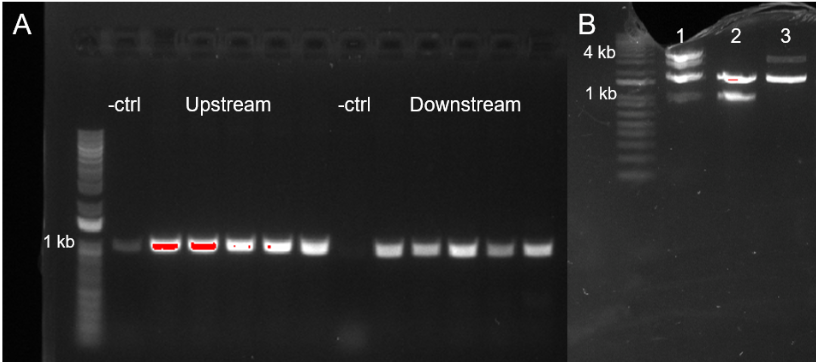
To confirm that we successfully created this part, we performed a PCR and gel electrophoresis using genomic DNA from the ADP1-ISx strain as a template. Bands were visible at ~1000 bp, confirming the amplification of the pbpG Upstream part. A PCR master mix with diH2O in place of template DNA was used as negative control.
References
[1] Gomez, M. J., & Neyfakh, A. A. (2006). Genes involved in intrinsic antibiotic resistance of Acinetobacter baylyi. Antimicrobial agents and chemotherapy, 50(11), 3562-3567. https://doi.org/10.1128/AAC.00579-06
[2] Lee, M.E., DeLoache, W.C., Cervantes, B., and Dueber, J.E. (2015). A highly characterized yeast toolkit for modular, multipart assembly. ACS synthetic biology 4, 975–986. 10.1021/sb500366v.
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
| None |