Part:BBa_K3904426
nupG-specific sgRNA
Introduction
Vilnius-Lithuania iGEM 2021 project AmeByelooks at amebiasis holistically and comprehensively, therefore target E. histolytica from several angles: prevention and diagnostics. Our team's preventive solution consists of probiotics engineered to produce naringenin - an antiprotozoal compound. Two strains of genetically modified microorganisms were chosen as the main chassis - world-renowned Lactobacillus casei BL23 (Lactobacillus paracasei) and Escherichia coli Nissle 1917. Furthermore, the team made specific gene deletions to enhance naringenin production and adapted a novel toxin-antitoxin system to prevent GMO spreads into the environment. The diagnostic part includes a rapid, point of care, user-friendly diagnostic test identifying extraintestinal amebiasis. The main components of this test are aptamers specific to the E. histolytica secreted proteins. These single-stranded DNA sequences fold into tertiary structures for particular fit with target proteins.
Contents
Usage and Biology
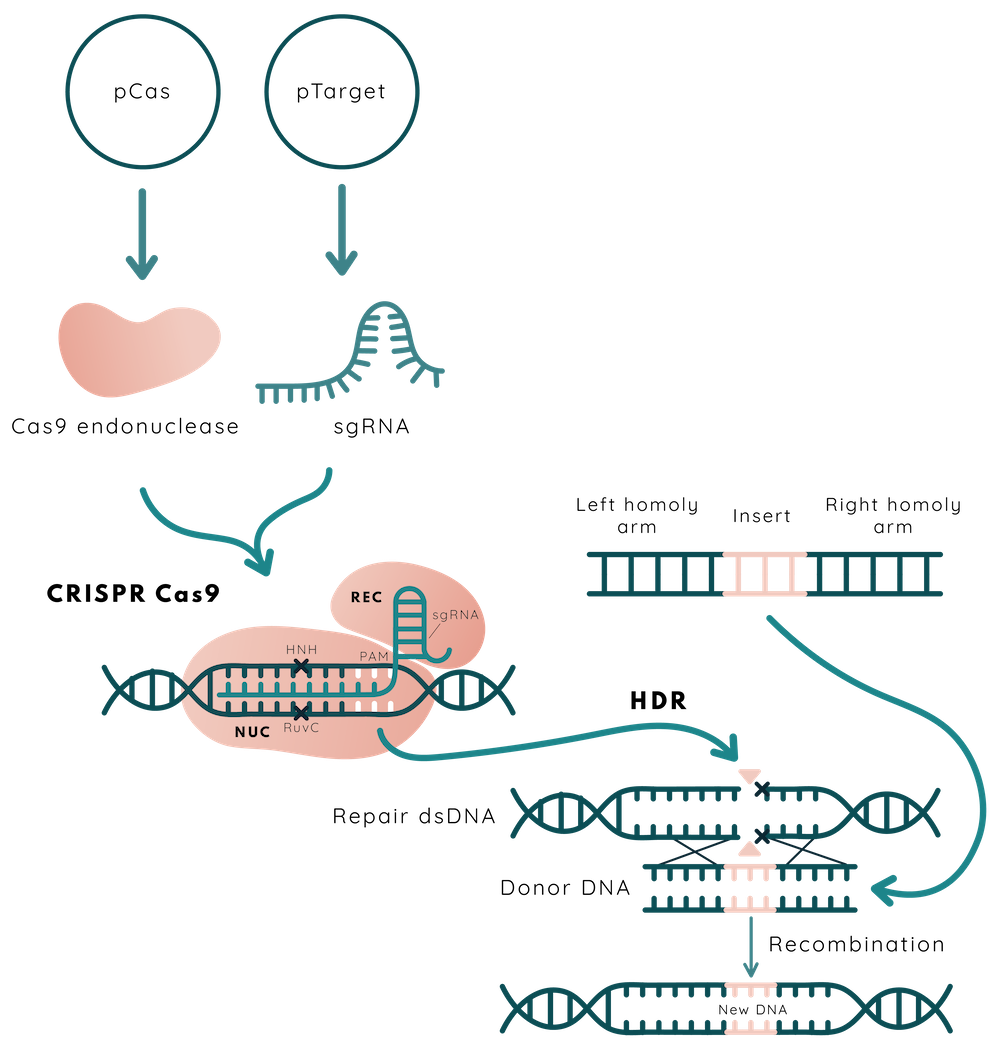
CRISPR-Cas9 is a versatile genome-editing technique. In our approach to editing E. coli Nissle 1917 genome, we have used two plasmid based system enabling to combine of Lambda Red recombination and CRISPR-Cas9 as counterselection tools encoded in pCas and pTarget.
Mechanism of genome editing
pCas plasmid is used for Cas9, Lambda Red system expression, and plasmid curing of pTarget. Cas9 - the RNA-guided endonuclease - is expressed constitutively, while the expression of Lambda Red genes (Gam, Exo, Beta) is under the control of arabinose inducible promoter araBp. pTarget plasmid caries constitutively expressed single-guide RNA (sgRNA). This RNA molecule, as and in nature, is composed of 17-20 nt length guide RNA (gRNA) sequence complementary to the targeted DNA adjacent to the protospacer adjacent motif (PAM) present at the 3' end, and the scaffold for the Cas9 nuclease binding to sgRNA and forming the ribonucleoprotein complex (1). Although in nature sgRNA exists as two separate RNA molecules, in laboratory experiments they are usually combined into one single-guide RNA (sgRNA) obviating additional maturation steps. As both pCas and pTarget plasmids are in a cell, Cas9 nuclease and sgRNA are able to form ribonucleoprotein complex, scan DNA for PAM sequences and perform a double-strand break in a part of the DNA which is complementary to the gRNA and adjacent to the PAM sequence - NGG. However, if arabinose has been added to the cell culture and a double-stranded DNA repair template is present in the cell, the Lambda Red system performs homology directed repair (HDR). If this process is unsuccessful, the Cas9-sgRNA complex will cause a double-strand break and will cause cell death (2). This is employed as a counter selection in order to avoid the additional antibiotic as selection marker usage.
Table 1. sgRNA collection for E. coli Nissle 1917 genome editing.
| ackA-specific sgRNA | BBa_K3904401 |
| pta-specific sgRNA | BBa_K3904402 |
| colicin-specific sgRNA | BBa_K3904405 |
| nupG-specific sgRNA | BBa_K3904426 |
Table 2. Parts collection for genomic insertion into E. Coli Nissle 1917 nupG gene.
| Short description | Part number |
| nupG-specific sgRNA | BBa_K3904426 |
| Forward primer for part of nupG gene amplification | BBa_K3904429 |
| Reverse primer for part of nupG gene amplification | BBa_K3904430 |
| First forward primer for generating a left homology arm of nupG gene | BBa_K3904428 |
| First reverse primer for generating a right homology arm of nupG gene | BBa_K3904427 |
| Second forward primer for generating a left homology arm of nupG gene | BBa_K3904431 |
| Second reverse primer for generating a right homology arm of nupG gene | BBa_K3904432 |
nupG-specific sgRNA importance to our project
Particularly in our project, we needed to insert the metabolic pathway for naringenin production into the genomes of our target probiotic strains. We have chosen two genomic sites for genomic insertions, which would do not harm or negatively affect our probiotic bacteria growth or overall performance - a putative colicin-encoding and nupG genes. We have inserted sfGFP encoding gene under the control of constitutive slpA promoter into these sequences. SfGFP fluorescence was significantly higher in samples with insertion in nupG, however, this case also demonstrated higher fluorescence fluctuations in comparison with putative colicin samples.
Fig. 1. Transcriptional activity comparison of GFP transcription from its constructs insertion in nupG or colicin genes.
Genome editing efficiency
nupG genomic VapXD system with constitutive slpA and bile-inducible promoter insertion with this sgRNA have been achieved by 100 % efficiency (fig. 1).
Fig. 2. cPCR of E. coli Nissle 1917 transformants. 1.3 kb PCR product formation from genomic DNA proves VapXD construct occurrence in the colicin genomic locus. All colonies are proved to be mutants harboring the VapXD system, where toxin encoding gene translation is induced by constitutive PslpA promoter, while antitoxin - by bile acids inducible promoter. L - GeneRuler 1 kb Ladder, 1 - negative control (wild type E. coli Nissle 1917), 2 - VapXD-cold&bile mutant (1), 3 - VapXD-cold&bile mutant (2), 4 - VapXD-cold&bile mutant (3), 5 - VapXD-cold&bile mutant (4), 6 - VapXD-cold&bile mutant (5), 7 - VapXD-cold&bile mutant (6), 8 - VapXD-cold&bile mutant (7), 9 - VapXD-cold&bile mutant (8), 10 - VapXD-cold&bile mutant (9), 11 - VapXD-cold&bile mutant (10), 12 - VapXD-cold&bile mutant (11), 13 - VapXD-cold&bile mutant (12).
nupG genomic VapXD system with cold and bile-inducible promoters insertion with this sgRNA have been achieved by 100 % efficiency (fig. 3).
Fig. 3. cPCR of E. coli Nissle 1917 transformants. 1.3 kb PCR product formation from genomic DNA proves VapXD construct occurrence in the colicin genomic locus. All except 6th colony are proved to be mutants harboring the VapXD system, where toxin encoding gene translation is induced by cold and antitoxin - by bile acids. L - GeneRuler 1 kb Ladder, 1 - VapXD-cold&bile mutant, 2 - VapXD-cold&bile mutant, 3 - VapXD-cold&bile mutant, 4 - VapXD-cold&bile mutant, 5 - VapXD-cold&bile mutant, 6 - negative control (wild type E. coli Nissle 1917).
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
References
- Doudna, J. A., & Charpentier, E. (2014). The new frontier of genome engineering with CRISPR-Cas9. Science, 346(6213).
- Jiang, Y., Chen, B., Duan, C., Sun, B., Yang, J., & Yang, S. (2015). Multigene editing in the Escherichia coli genome via the CRISPR-Cas9 system. Applied and environmental microbiology, 81(7), 2506-2514.
- Ku, J. T., Chen, A. Y., & Lan, E. I. (2020). Metabolic engineering design strategies for increasing acetyl-CoA flux. Metabolites, 10(4), 166.
- Wu, J., Du, G., Chen, J., & Zhou, J. (2015). Enhancing flavonoid production by systematically tuning the central metabolic pathways based on a CRISPR interference system in Escherichia coli. Scientific reports, 5(1), 1-14.
| None |