Part:BBa_K3781009
GST + HRV 3C Motif, MocloMania B3
This basic part codes for a GST-tag that is fused to a downstream HRV 3C protease recognition motif.
The GST-tag has been used for purification purposes eversince the late 1980s and, unlike most other purification tags, doesn’t only consist of a small peptide, but rather an entire functional protein.[1] GST is short for glutathione S-transferase, a highly abundant cytosolic enzyme that is usually involved in protecting prokaryotic as well as eukaryotic cells from extracellular toxins.[2] It does this with the help of the redox peptide glutathione to whose reduced form it binds with very high affinity.[3] By fusing a gene of interest with a GST-tag, the resulting fusion protein can be purified from cell preparations with the help of affinity chromatography, employing reduced glutathione (GSH) as the respective column material. Since the affinity binding relies on the structural integrity of the GST protein, GST-tag purification can only be performed under native, non-denaturing conditions.[4]
The GST-tag encoded in this part is fused to a recognition site for the human rhinovirus 3C protease, a cysteine protease that usually mediates the disruption of host cell transcription during viral cell infection.[5] It binds and cleaves its recognition site with high specifity and, when its motif is fused with a tag, can be used for recovery of tag-free target protein after purification.
size 26.3 kDa
function affinity purification tag
cloning position B3
plasmid backbone pICH41258
Data
We were able to successfully clone this basic part into its respective L0 plasmid backbone and to confirm the integrity of the L0 construct via restriction digest and gel electrophoresis, see Figure 1. Furthermore, we were able to include it into a L1 construct, proving its correct adaptation towards MoClo assembly, see Figure 2. This part has been transfected into Leishmania, but it has not yet been tested for expression and purification functionality.
-
 Figure 1 | Test digest of L0 constructs using PstI and BstEII
Figure 1 | Test digest of L0 constructs using PstI and BstEII
1 | L0-GST-B3 | 2251 + 680 bp
2 | pICH41258 | 1850 + 995 bp
L | Thermofischer GeneRuler Plus Ladder [bp] -
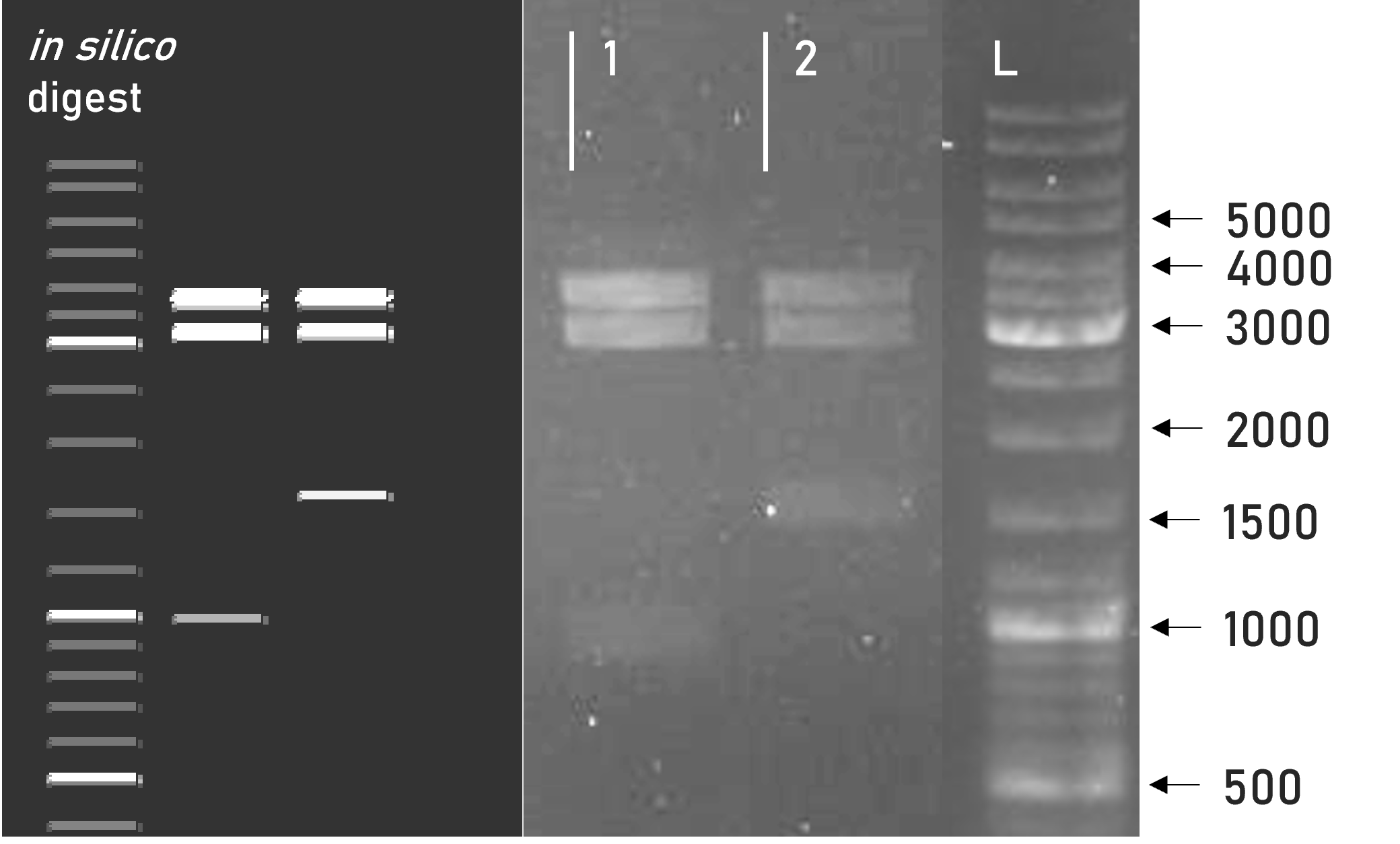 Figure 2 | Test digest of L1 constructs using HindIII
Figure 2 | Test digest of L1 constructs using HindIII
1 | L1_sAP_Strep_RBD_Strep8His | 3792 + 3198 + 1005 bp
2 | L1_sAP_GST_RBD_Strep8His | 3792 + 3198 + 1623 bp
L | Thermofischer GeneRuler Plus Ladder [bp]
The MocloMania collection
This basic part is part of the MocloMania collection, the very first collection of genetic parts specifically designed and optimized for Modular Cloning assembly and recombinant protein expression in the protozoan parasite Leishmania tarentolae.
Are you trying to express complexly glycosylated proteins? Large antibody side chains? Human proteins that require accurate post-translational modification? Then Leishmania might be just the right organism for you! Leishmania tarentolae’s glycosylation patterns resemble those of human cells more closely than any other microbial expression host, while still delivering all the benefits of microbial production systems like easy transfection and cultivation.[6] So instead of relying on mammalian cell lines, try considering Leishmania as your new expression host of choice!
Our MocloMania collection will allow you to easily modify your protein of choice and make it suitable for downstream detection and purification procedures - all thanks to the help of Modular Cloning. This cloning system was first established by Weber et al. in 2011 and relies on the ability of type IIS restriction enzymes to cut DNA outside of their recognition sequence, hereby generating four nucleotide overhangs.[7] Every basic part in our collection is equipped with a specified set of overhangs that assign it to its designated position within the reading frame. These so-called cloning positions are labelled B2-B5 from upstream to downstream. By filling all positions with the basic parts of your choice, you can easily generate variable genetic constructs that code for the fusion protein of your desire.
We furthermore provide a specifically domesticated Leishmania expression vector, named weird_plex, which will package your fusion construct into a functional transcriptional unit that is optimized for high expression in Leishmania.
The best part? Because of the type IIS restriction properties and the specifity of the generated overhangs, restriction and ligation of your construct can all happen simultaneously in a simple one-step, one-pot reaction. This will safe you a lot of time and frustration in your cloning endeavours!
Do we have your attention? In the table below you can find some basic information on how our cloning system, along with most other MoClo systems, is set up. Please feel free to check out our wiki to find more information on Leishmania and Modular Cloning as well as to understand how this basic part integrates into our part collection. See you there!
| Level | What does this level contain? | antibiotic resistance | Enzyme used for ligation |
| L0 | The foundation to every MoClo construct which are basic genetic units, such as coding sequences, promoters, terminators | spectinomycin | BbsI |
| L1 | Several L0 parts assembled into a functional transcriptional unit, e.g. consisting of promoter, coding region and terminator | ampicillin | BsaI |
| L2 | Multiple transcriptional units added into one multi-gene construct, e.g. a protein of interest fused to a selection marker | kanamycin | BbsI |
Sequence and Features
IMPORTANT
This basic part has been altered in its sequence in order to make it compatible with the iGEM RCF 1000 cloning standard.
In its original form, this part contains two endogenous SapI restriction sites. This is because it was specifically designed to be utilized within the Modular Cloning system as established by Weber et al. in 2011, relying on BsaI and BbsI as the executive type IIS restriction enzymes.[8] This MoClo standard is a very frequently used type IIS standard, especially when it comes to plant-based expression systems.[9][10]
Because of its intended use within the MoClo system, this basic part is domesticated against BsaI and BbsI, but not against SapI. Upon registration of this part in the iGEM Registry, the endogenous SapI restriction sites were disrupted via in silico introduction of a silent mutation. We decided to do this in order to increase the chance of the judges actually taking a look at this part page and not having the part overlooked due to incompatibility.
Since the part’s amino acid sequence remains unchanged, users interested in utilizing this part for type IIS loop assembly can rely on the sequence as listed on this page. For users interested in applying this basic part to their MoClo based cloning approaches, in silico reversion of the mutations might be beneficial, since SapI proved to be a valuable enzyme for confirming part insertion via restriction digest.
We highly recommend that researchers and iGEM teams utilize this basic part within the MocloMania cloning system, since it is codon optimized towards Leishmania tarentolae and may otherwise negatively influence expression yield in other organisms.
MUTATION 1 | bp 583 - 585 | AGC → TCG
MUTATION 2 | bp 655 - 657 | AGC → TCG
- 10INCOMPATIBLE WITH RFC[10]Illegal PstI site found at 607
- 12INCOMPATIBLE WITH RFC[12]Illegal PstI site found at 607
- 21COMPATIBLE WITH RFC[21]
- 23INCOMPATIBLE WITH RFC[23]Illegal PstI site found at 607
- 25INCOMPATIBLE WITH RFC[25]Illegal PstI site found at 607
- 1000COMPATIBLE WITH RFC[1000]
Reference Literature
- ↑ Smith DB, Johnson KS. Single-step purification of polypeptides expressed in Escherichia coli as fusions with glutathione S-transferase. Gene. 1988 Jul 15;67(1):31-40. doi: 10.1016/0378-1119(88)90005-4. PMID: 3047011.
- ↑ Hayes JD, Flanagan JU, Jowsey IR. Glutathione transferases. Annu Rev Pharmacol Toxicol. 2005;45:51-88. doi: 10.1146/annurev.pharmtox.45.120403.095857. PMID: 15822171.
- ↑ Lohning, Anna, Salinas, Anna, Wong, M.G., 1999/04/01, Glutathione S-transferases--a review, Current Medicinal Chemistry
- ↑ Harper S, Speicher DW. Purification of proteins fused to glutathione S-transferase. Methods Mol Biol. 2011;681:259-280. doi:10.1007/978-1-60761-913-0_14
- ↑ Amineva SP, Aminev AG, Palmenberg AC, Gern JE (October 2004). "Rhinovirus 3C protease precursors 3CD and 3CD' localize to the nuclei of infected cells". The Journal of General Virology. 85 (Pt 10): 2969–79. doi:10.1099/vir.0.80164-0. PMID 15448360
- ↑ Langer T, Corvey C, Kroll K, Boscheinen O, Wendrich T, Dittrich W. Expression and purification of the extracellular domains of human glycoprotein VI (GPVI) and the receptor for advanced glycation end products (RAGE) from Rattus norvegicus in Leishmania tarentolae. Prep Biochem Biotechnol. 2017 Nov 26;47(10):1008-1015. doi: 10.1080/10826068.2017.1365252. Epub 2017 Aug 31. PMID: 28857681.
- ↑ Weber E, Engler C, Gruetzner R, Werner S, Marillonnet S (2011) A Modular Cloning System for Standardized Assembly of Multigene Constructs. PLoS ONE 6(2): e16765. https://doi.org/10.1371/journal.pone.0016765
- ↑ Weber E, Engler C, Gruetzner R, Werner S, Marillonnet S (2011) A Modular Cloning System for Standardized Assembly of Multigene Constructs. PLoS ONE 6(2): e16765. https://doi.org/10.1371/journal.pone.0016765
- ↑ Patron, N.J., Orzaez, D., Marillonnet, S. et al., (2015), Standards for plant synthetic biology: a common syntax for exchange of DNA parts. New Phytol, 208: 13-19. https://doi.org/10.1111/nph.13532
- ↑ https://www.addgene.org/cloning/moclo/, last visited 10/16/21, 19:00 ECT
| None |