Part:BBa_K2715001
Constitutive clostridial promoter Pcac_thl (BBa_K2715010), strong RBS and GFP reporter
Usage and Biology
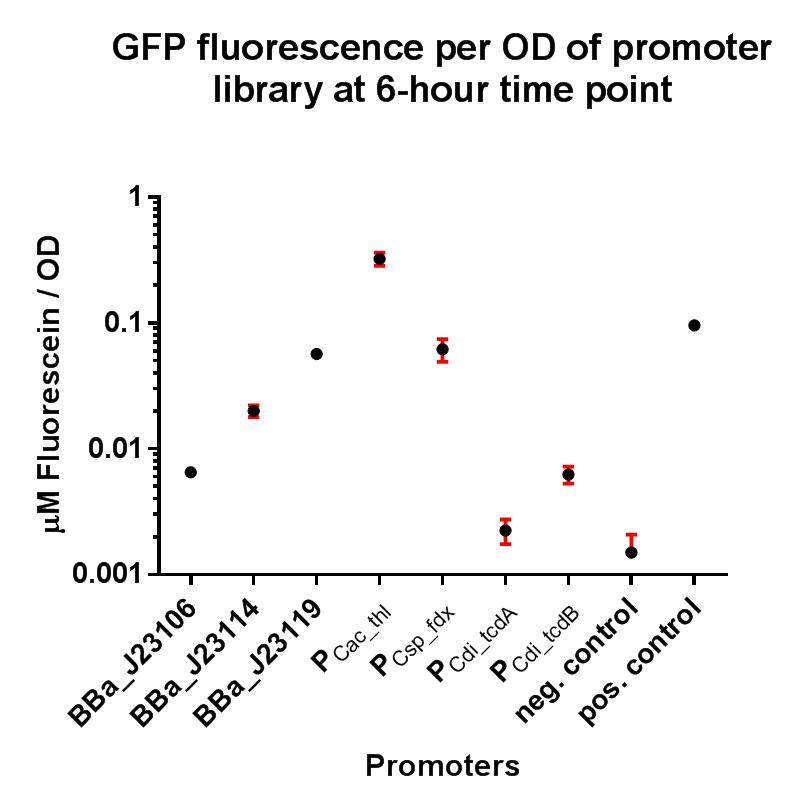
Our project required the use of strong constitutive promoters which would function well in the non-model Gram-positive organism Clostridium difficile. We also wanted to establish whether these promoters functioned in E. coli, as this could have implications for cloning stages and vector assembly when trying to build constructs containing potentially toxic genes. In order to put the strength of these promoters into context, we decided upon a GFP fluorescence assay using the iGEM Interlab calibration curves, and also to compare their strengths to the Interlab positive and negative controls. This promoter was taken from the related species Clostridium acetobutylicum, driving expression of the thiolase gene, which has been shown in previous research to generally have strong expression in clostridial species.
Characterisation
In this composite part we've added a strong RBS BBa_K2715009, shown to function in Gram-positive and Gram-negative organisms, downstream of the BBa_J23010 clostridial promoter, driving expression of GFP taken from BBa_E0040. This composite part also includes the 5’ UTR region of the thiolase promoter BBa_K2715019. The construct is part of a family of composite parts which all share the same strong RBS and GFP gene and were all characterised in the same plasmid backbone and in parallel in a fluorescence assay, the results of which can be seen below. The positive and negative controls are parts BBa_I20270 and BBa_R0040 respectively, used in the Interlab 2018 study.
The composite parts tested in this assay under the same conditions using a range of alternative promoters are as follows:
BBa_K2715106
BBa_K2715114
BBa_K2715119
BBa_K2715001
BBa_K2715002
BBa_K2715003
BBa_K2715004
These composite parts were assembled in the shuttle cloning vector pMTL84151, and characterised in E. coli within this plasmid. Additionally these promoters were characterised in C. difficile using the gusA biobrick BBa_K330002 as a reporter gene in place of GFP, as GFP requires oxygen in order to function, and C. difficile is an anaerobic organism. Unfortunately despite our efforts we were unable to clone the gusA composite variant of the thiolase promoter, BBa_K2715025, despite successfully cloning all other members of this promoter library upstream of gusA. We hypothesised this was likely due to the very high strength of the promoter in E. coli, and the fact that over expression of gusA is toxic to the cells. A number of methods were attempted in order to achieve this cloning step, including changing the host strain for HB101, using a lower copy Gram-positive replicon in the plasmid backbone, and incubating the cultures at a reduced temperature. Unfortunately none of these methods enabled us to overcome the toxicity issues. The attempted composite part driving gusA is also listed in the group below:
The gusA containing composites used to assay the promoter activities in C. difficile are listed below.
BBa_K2715025
BBa_K2715026
BBa_K2715027
BBa_K2715028
BBa_K2715029
BBa_K2715030
BBa_K2715031
The plasmid used for this characterisation in E. coli is displayed below.
Conclusions
This composite part has enabled a standardised characterisation of K2715010 when used in conjugation with a strong RBS shown to function in both Gram-positive and Gram-negative organisms, and its strength can be quantified in E. coli using the iGEM 2018 interlab units of fluorescence. It has been demonstrated in this assay to be an exceptionally strong promoter in E. coli when in conjugation with this RBS, resulting in greater GFP production then any other promoter tested, including the Interlab positive control vector. We foresee this promoter being of use to future iGEM teams who which to use a promoter that will result in very high expression in both Gram-positive and Gram-negative organisms, including non-model organisms such as clostridial species.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000INCOMPATIBLE WITH RFC[1000]Illegal BsaI.rc site found at 797
References
Heap, J.T., Pennington, O.J., Cartman, S.T. and Minton, N.P., 2009. A modular system for Clostridium shuttle plasmids. Journal of microbiological methods, 78(1), pp.79-85.
Davis, D.F., Ward, W.W. and Cutler, M.W., 1994. Posttranslational chromophore formation in recombinant GFP from E. coli requires oxygen. In Bioluminescence and Chemiluminescence: Fundamentals and Applied Aspects. Proceedings of the 8th International Symposium on Bioluminescence and Chemiluminescence, Cambridge. Wiley, New York, NY (pp. 569-599).
Chiu, N.H. and Watson, A.L., 2017. Measuring β‐Galactosidase Activity in Gram‐Positive Bacteria Using a Whole‐Cell Assay with MUG as a Fluorescent Reporter. Current protocols in toxicology, 74(1), pp.4-44.
| None |