Part:BBa_K1732004
pSB1C3-J23100-B0034-CDcel-Gaussia-His-B0015
J23100 constitutive promoter (BBa_J23100), a RBS (BBa_B0034), CDcel domain (BBa_K1732002), Gaussia luciferase (BBa_K1732003), and B0015 terminator (BBa_B0015). ______________________________________________________________________________________________________________________________
This DNA sequence, expressed from a constitutive, strong promoter allows for the Gaussia luciferase protein to be synthesized and was targeted extrecellularly into the media. The Gaussia luciferase proteins were reacted with the optimal concentration of coelenterazine to express luminescence, which was used to test the Carnegie Mellon DIY luminometer. The sequence was codon optimized for E.coli and contains a 6X His-tag for easier purification of the protein.
Gaussia Luciferase (GLuc) is a naturally secreted protein from Gaussia princeps. It is considered the brightest luciferase and its size is around 19kDa. This luciferase is stable at high temperatures due to the presence of disulfide bonds in its structure. This stability makes it an ideal reporter gene, specifically for coelenterazine [1].
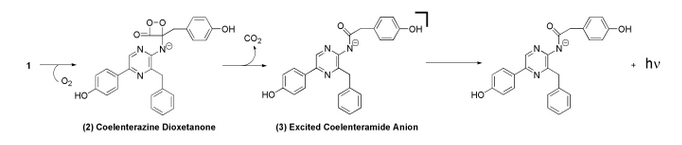
Bioluminescent Mechanism of Coelenterazine
Coelenterazine is found in several aquatic organisms and is a luminescent substrate for many luciferase enzymes. For this study, PelB Gaussia Luciferase interacted with coelenterazine to determine relative light output and kinetics [3].
In the mechanism, coelenterazine reacts with oxygen to yield 1,2-dioxetane (compound 2). Subsequent loss of carbon dioxide leads to the intermediate, followed by emission of a photon [2].
The image above is the chemiluminescent mechanism of coelenterazine. The bioluminescent mechanism is similar, except that the excited state molecule in bioluminescent is phenolate anion instead of the amide anion [2].
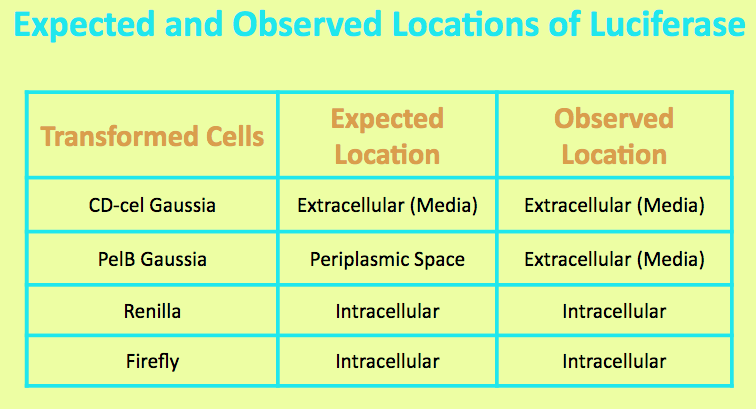
Before running experiments with CD-cel Gaussia Luciferase, the location of the luciferase in the media was determined by comparing the light output of overnight grown culture of CD-cel cells with those of the pellet and the supernatant. The CD-cel Gaussia culture was spun down, and the pellet represented intracellular localization while the supernatant represented extracellular localization.
It was expected that the Gaussia Luciferase was localized in the cell, but the level of light output from the media matched that of the overnight culture and was significantly higher than that of the pellet. This suggested that the luciferase is located in the media and secreted from the cells.
The graph shows the amount of light output produced from varying concentrations of coelenterazine when added to 100uL of CD-cel Gaussia cells grown overnight (5mL LB broth, 5uL Chloramphenicol, single cell colony). 10uL of coelenterazine was added and this volume stayed consistent. The overnight culture was diluted 1/10 with LB broth in order to measure the Klett OD and stay within accurate measurement range. The purpose of this is to see the effect of light output with increasing concentrations of coelenterazine. The goal was to find a concentration that plateau without maxing out the luminometer (TECAN). It was discovered that at a certain concentration, the light output stop increasing because the acidic methanol used to make the stock solution of coelenterazine began to interfere with the enzyme activity.
The competent cells used were Mach cells. Mach cells are one of the fastest growing competent strain and they are able to replicate high number of plasmids at stable levels. Top10 cells were not tested because transformation was unsuccessful.
From the graphs, it can be determined that higher concentrations of coelenterazine allows for a higher light output. However, it is important to note that CD-cel luciferase is not as sensitive to coelenterazine when compared to PelB Gaussia. This is evident by the high concentrations of coelenterazine tested. As the concentration of coelenterazine increases, the increase in light output gets smaller. The culture may have become saturated with coelenterazine or the acidic methanol was interfering with the reaction. Also, the plateaus are not proportional to the concentrations of coelenterazine, and therefore, it may be detrimental to have too much coelenterazine.
References
[1]Gaussia Luciferase [Fact sheet]. (n.d.). Retrieved September 14, 2015, from New England BioLabs Inc.
website: https://www.neb.com/applications/cellular-analysis/reporter-systems/gaussia-luciferase
[2]Gonzalez, V. M., Jr. (2007). Synthesis, Luminescence, and Applications of Coelenterazine and its
Analogs. University of Illinois Urbana-Champaign.
[3]Coelenterazine [Fact sheet]. (n.d.). Retrieved September 14, 2015, from Gold Biotechnology website:
https://www.goldbio.com/product/1012/coelenterazine
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12INCOMPATIBLE WITH RFC[12]Illegal NheI site found at 7
Illegal NheI site found at 30 - 21INCOMPATIBLE WITH RFC[21]Illegal BglII site found at 1517
Illegal BamHI site found at 975 - 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000INCOMPATIBLE WITH RFC[1000]Illegal BsaI.rc site found at 715
| None |