Part:BBa_K1343016
Pcat->RBS->TaZ->dTer
This part generates the TaZ receptor. The TaZ is under a Pcat constitutive promoter. TaZ is a fused Tar-EnvZ receptor meant for Aspartate dependent response, by activating the EnvZ/OmpR two component signalling system and thus activating the PompC promoter. We used this part for prove-of-concept for low concentration detection. By using this TaZ Generator we were able to test different concentrations of Aspartate and their effect on the PompC promoter present in another plasmid.
To test the activity of the Taz construct we created, varying concentrations of L-aspartic acid were added to a culture of E. coli expressing the Taz construct on plasmid pSB1AK3 and RFP under the promoter PompC (biobrick Bba_M30011) on plasmid pSB1C3.
Two isogenic strains of E. coli K-12, BW25113 (parent strain for the Keio collection) and JW3367-3 (with ΔEnvZ mutation) were transformed with pSB1AK3 carrying our Taz construct (BBa_K1343016) and pSB1C3 carrying the BBa_M30011 reporter. The bacteria were cultured in growth media containing varying concentrations of L-aspartic acid. After two hours of growth the relative RFP fluorescence of the cultures was determined (fluorescence/OD).
OD was measured at 600nm. Fluorescence excitation wavelength: 560nm Fluorescence emission wavelength: 612nm
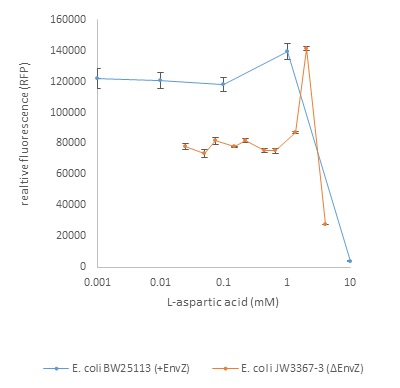
Figure 1: Relative fluorescence dependent on L-aspartic acid concentration (mM)
Figure 1 shows that at low concentrations of L-aspartatic acid (below 1mM), there is a steady, constant expression of the reporter in both the wild type and the mutant strain. The relative fluorescence observed by the mutant strain (ΔEnvZ) is lower than that of the parent strain. This is because the natural histidine kinase protein which detects a high level of osmolarity in the cells environment (EnvZ) is present in the wild type. This contributes to phosphorylation of the ompR. The low level of fluorescence observed in the mutant strain could be due to another mechanism (such as an acetyl phosphate dependent mechanism) which phosphorylates the ompR, leading to activation of the PompC promoter.
The mutant shows an increase in relative fluorescence is observed at 1.33mM L-aspartic acid, with a peak at 2mM followed by a drop in relative fluorescence at 4mM. The drop in relative fluorescence in both the mutant and wild type indicates a toxic concentration level.
In the mutant strain, the relative fluorescence increases 202% in comparison to the basal level. This occurs over a narrow concentration range, which reflects the sensitivity of the two-component signaling system.
References
Harrison, K. (2013). Reporter ompC-GFP. Retrieved from Toximop: http://2013.igem.org/Team:Dundee/Project/ReporterOmpC Heyde, M., Laloi, P., & Portalier, R. (2000). Involvement of Carbon Source and Acetyl Phosphate in the External-pH-Dependent Expression of Porin Genes in Escherichia coli. Journal of Bacteriology, 182(1), 198-202.
Levskaya, A., Chevalier, A. A., & Tabor, J. J. (2005). Engineering Escherichia Coli to see light. Nature, 438(24), 441-442. Michalodimitrakis, K. M., Sourjik, V., & Serrano, L. (2005). Plasticity in amino acid sensing of the chimeric receptor Taz. Molecular Microbiology, 58(1), 257–266.
Tabor, J. J., Groban, E. S., & Voigt, C. A. (2009). Performance Characteristics for Sensors and Circuits Used to Program E. coli. In S. Y. Lee (Ed.), Systems Biology and Biotechnology of Escherichia coli (pp. 401-439). Springer Science+Business Media B.V. Yoshida, T., Phadtare, S., & Inouye, M. (2007). The Design and Development of Tar-EnvZ Chimeric Receptors. Methods in Enzymology, 423, 166-183.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000INCOMPATIBLE WITH RFC[1000]Illegal SapI.rc site found at 167
| None |