Part:BBa_K1321350
CBDcenA with linker fused to sfGFP in Freiburg format (RFC 25)
Super-folder GFP fused N-terminally to CBDcenA (a cellulose-binding domain), which contains an endogenous C-terminal linker sequence.
This construct is part of a library of Super-folder GFP fusions with cellulose binding domains, which we used to assay the CBD binding affinity. Please see our [http://2014.igem.org/Team:Imperial/Functionalisation project page] for more information. The collection of sfGFP-CBD fusion parts can be seen in the table below: 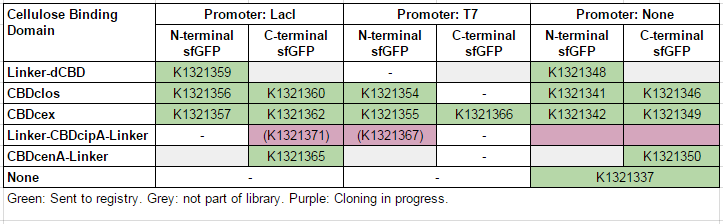
Note that the start and stop codon, plus 6 bp either side of the sequence, are included the RFC25 prefix and suffix which is not shown.
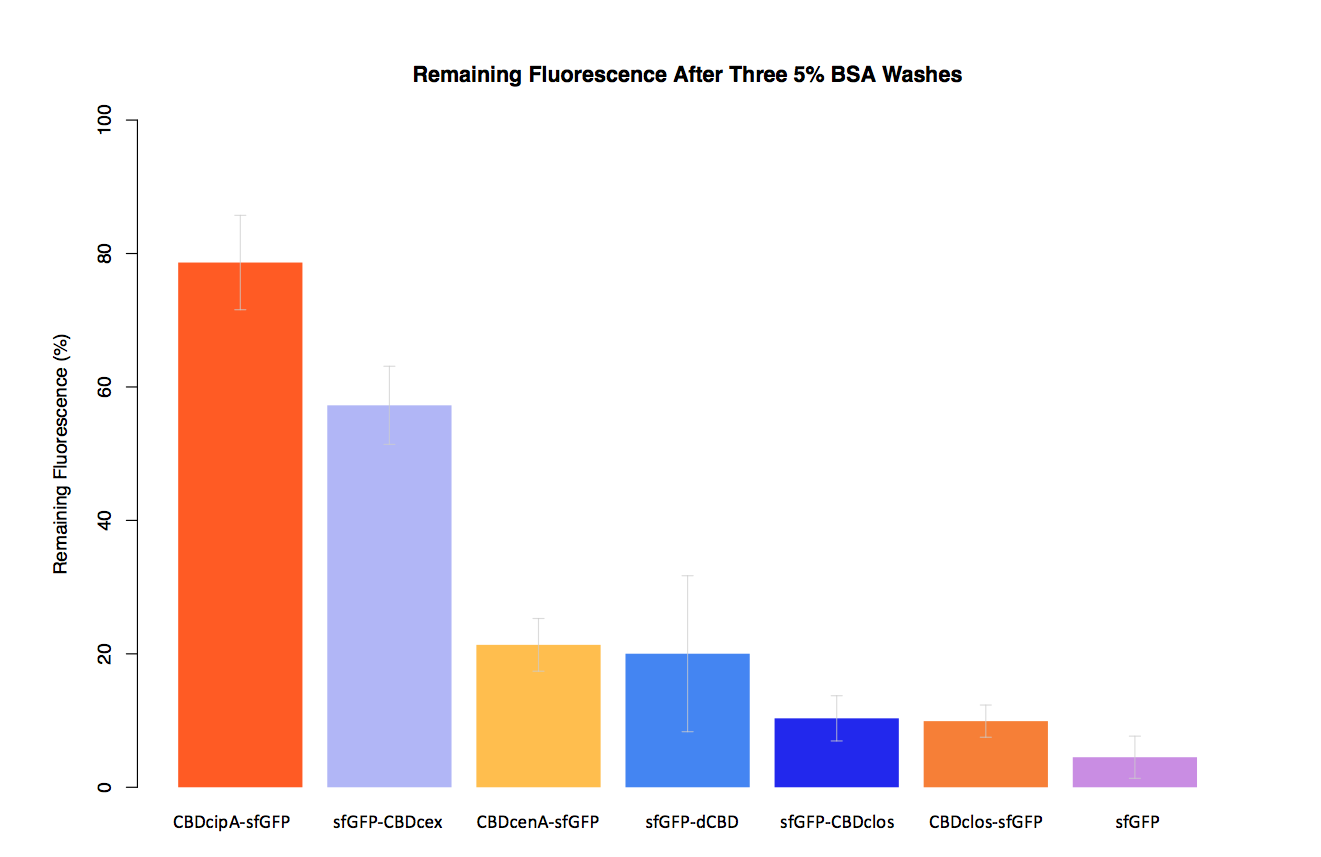
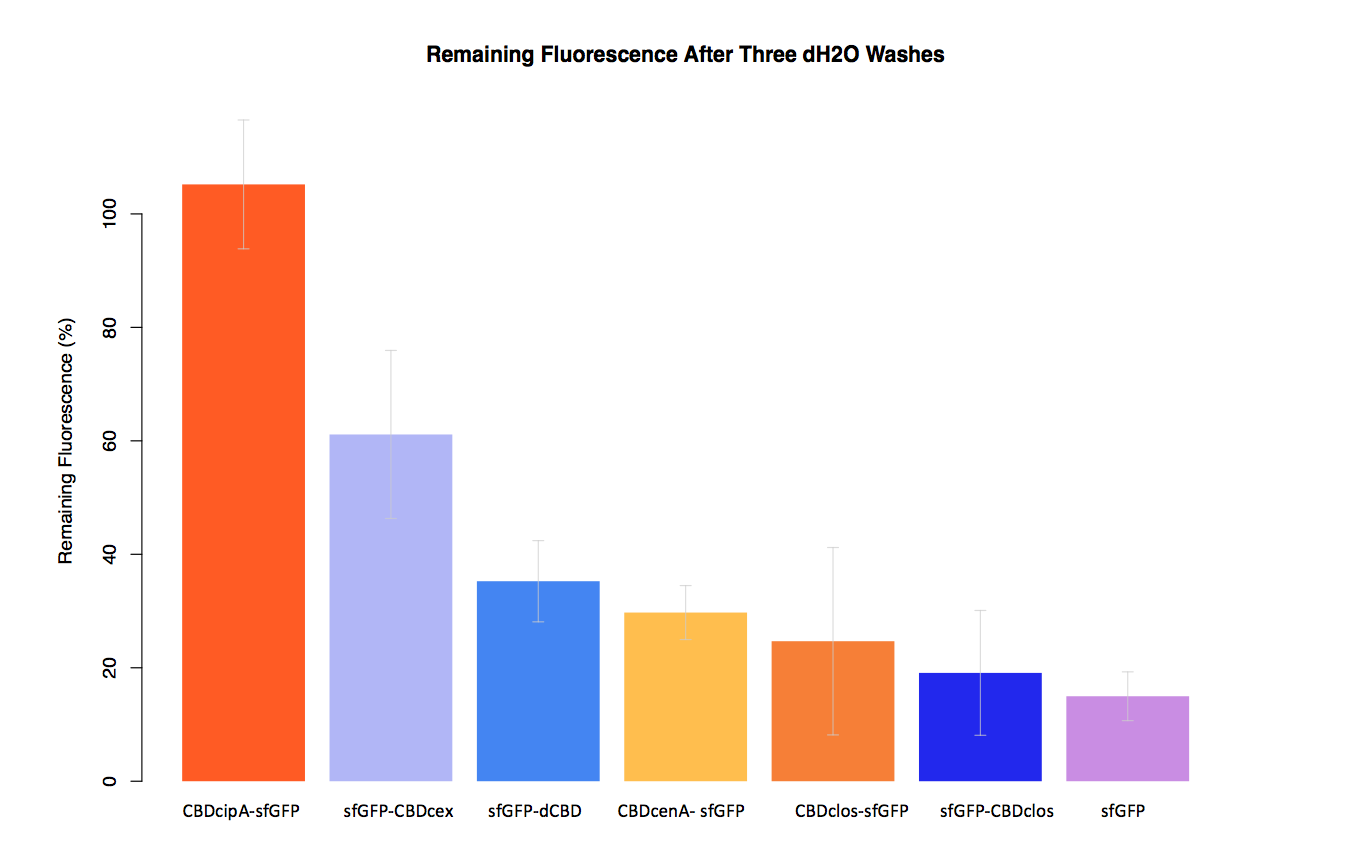
As part of our project we carried out an assay to determine the relative binding ability of CBD-sfGFP fusions to bacterial cellulose.
In our second assay performed to determine the relative strengths of various CBDs’ binding to bacterial cellulose – represented by the percentage fluorescence left from CBDs fused to (sfGFP (RFC25)) bound to bacterial cellulose discs, when subjected to various washes (protocol [http://2014.igem.org/Team:Imperial/Protocols here]) - it was determined that the fusion with CBDcenA had moderate binding ability in comparison to five other CBDs fused to sfGFP, having the third and fourth highest fluorescence remaining after three washes with 5% BSA and dH2O.
Please note, in the second graph (with dH2O washes) the unexpectedly high fluorescence readings were most likely caused by a calibration error in the plate reader used, as it was the first instance in which it had carried out measurements in our assays.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000INCOMPATIBLE WITH RFC[1000]Illegal SapI.rc site found at 382
| None |