Part:BBa_K1172403
mtrCAB with medium Promoter and medium RBS
Usage and Biology
To enable transfer of electrons from the general metabolism to the outside of the cell, the mtrCAB operon from [http://www.ncbi.nlm.nih.gov/genome/1082?project_id=57949 Shewanella oneidensis MR-1] was heterologously expressed in E. coli. This operon encodes for a minimal set of genes required to build an electron shuttle pathway via different c-type cytochromes. Electrons from the native E. coli protein [http://www.ncbi.nlm.nih.gov/protein/EGT66377.1 NapC] are passed to the periplasmic [http://www.ncbi.nlm.nih.gov/protein/NP_717386.1 MtrA], which transports them to the outer membrane protein [http://www.ncbi.nlm.nih.gov/protein/NP_717385.1 MtrB]. Via the membrane-bound [http://www.ncbi.nlm.nih.gov/protein/NP_717387.1 MtrC] the corresponding electrons can be transferred to extracellular electron acceptors like the anode of a microbial fuel cell.
Part uses
| Device | Regulatory Part | Promoter | RBS | Activity |
|---|---|---|---|---|
| BBa_K1172403 | BBa_K608006 | Anderson 0.33 | medium | medium |
| BBa_K1172404 | BBa_K608002 | Anderson 0.77 | strong | strong |
| BBa_K1172405 | BBa_K525998 | T7 induced | strong | very strong |
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12INCOMPATIBLE WITH RFC[12]Illegal NheI site found at 7
Illegal NheI site found at 30 - 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 2111
- 1000INCOMPATIBLE WITH RFC[1000]Illegal BsaI site found at 297
Results
Genetics
- The amplification of the mtrCAB cluster from the genome of S. oneidensis and the deletion of illegal restriction sites was accomplished.
- The gene cluster was succesfully ligated into the shipping vector pSB1C3 forming the BioBrick BBa_K1172401
- The gene cluster was ligated into three expression vectors with varying promoter strength forming the following devices
Characterisation
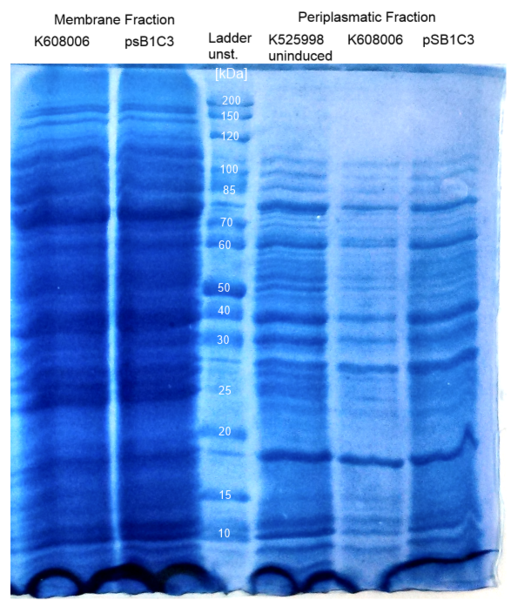
- To prove the expression of the Mtr proteins, E. coli cultures were transformed transformed with BBa_K1172403, BBa_K1172405 (uninduced) and BBa_K1172401 as a control and then cultivated anaerobically. Membrane and periplasmatic fractions of the cells were isolated by [http://2013.igem.org/Team:Bielefeld-Germany/Labjournal/ProtocolsPrograms#Cold_osmotic_shock Cold osmotic shock fractioning.] and analyzed by [http://2013.igem.org/Team:Bielefeld-Germany/Labjournal/ProtocolsPrograms#Sodium_dodecyl_sulfate_polyacrylamide_gel_electrophoresis_.28SDS-PAGE.29 SDS-PAGE]; the results are shown in Figure 5.
- The membrane fraction should contain [http://www.ncbi.nlm.nih.gov/protein/NP_717385.1 MtrB] and [http://www.ncbi.nlm.nih.gov/protein/NP_717387.1 MtrC] of the Mtr complex, with sizes of 72 kDa and 69 kDa, respectively.
- The periplasmatic fraction should only contain the [http://www.ncbi.nlm.nih.gov/protein/NP_717386.1 MtrA] protein of the Mtr complex (32 kDa).
- It was not possible to confirm the expression of the Mtr proteins by [http://2013.igem.org/Team:Bielefeld-Germany/Labjournal/ProtocolsPrograms#Sodium_dodecyl_sulfate_polyacrylamide_gel_electrophoresis_.28SDS-PAGE.29 SDS-PAGE] analysis, as no significant differences between control and experimental condition were visible at the relevant heights of the SDS-PAGE.
- The redox activity of [http://www.ncbi.nlm.nih.gov/protein/NP_717386.1 MtrA] and [http://www.ncbi.nlm.nih.gov/protein/NP_717387.1 MtrC] was probed via absorption spectroscopy and did not show significant indications for a positive result as the expected shift of the Soret peak from 410 nm to 420 nm could not be observed.
Conclusion
- The electron transfer system from Shewanella oneidensis MR-1 seems to be suitable for the usage in our Microbial Fuel Cell in principle. However, expression regulation, heme-loading, and correct folding as well as localization of the cytochromes are very complex and therefore our team was not able to produce the functional system in the available time.
References
- Beliaev AS, Saffarini DA (1998)Shewanella putrefaciens mtrB encodes an outer membrane protein required for Fe(III) and Mn(IV) reduction. [http://jb.asm.org/content/180/23/6292.short J Bacteriol 180:6292–6297]
- Beliaev, A. S., D. A. Saffarini, J. L. McLaughlin, and D. Hunnicutt. 2001. MtrC, an outer membrane decahaem c cytochrome required for metal reduction in Shewanella putrefaciens MR-1. [http://onlinelibrary.wiley.com/doi/10.1046/j.1365-2958.2001.02257.x/full Mol. Microbiol. 39:722-730]
- Grove, J., Tanapongpipat, S., Thomas, G., Griffiths, L., Crooke, H., and Cole, J. (1996)Escherichia coliK-12 genes essential for the synthesis of c-type cytochromes and a third nitrate reductase located in the periplasm. [http://onlinelibrary.wiley.com/doi/10.1046/j.1365-2958.1996.383914.x/abstract Mol. Microbiol. 19, 467−481]
- Jensen HM, Albers AE, Malley KR, Londer YY, Cohen BE, et al. (2010) Engineering of a synthetic electron conduit in living cells. [http://www.pnas.org/content/107/45/19213.short Proc Natl Acad Sci USA. 10.1073/pnas.1009645107 Proc. Natl Acad. Sci. USA 107, 19213–19218 (2010).]
- Myers CR, Myers JM (2002) MtrB is required for proper incorporation of the cytochromes OmcA and OmcB into the outer membrane of Shewanella putrefaciens MR-1. [http://aem.asm.org/content/68/11/5585.short Appl Environ Microbiol68:5585–5594]
- Sanders, C., Turkarslan, S., Lee, D.-W., and Daldal, F. (2010) Cytochromecbiogenesis: the Ccm system. [http://www.sciencedirect.com/science/article/pii/S0966842X10000442 Trends Microbiol. 18, 266−274]
- Schicklberger, M., Bucking, C., Schuetz, B., Heide, H., and Gescher, J. (2011) Involvement of the Shewanella oneidensis decaheme cytochrome MtrA in the periplasmic stability of the beta-barrel protein MtrB. [http://aem.asm.org/content/77/4/1520.short Appl. Environ. Microbiol. 77, 1520−1523]
- Thony-Meyer L, Fischer F, Kunzler P, Ritz D, Hennecke H (1995)Escherichia coligenes required for cytochrome c maturation. [http://jb.asm.org/content/177/15/4321.short J Bacteriol 177:4321–4326]
| None |