Difference between revisions of "Part:BBa K2273040"
| Line 1: | Line 1: | ||
__NOTOC__ | __NOTOC__ | ||
| + | |||
| + | |||
| + | |||
| + | |||
{| style="color:black; margin: 20px 0px 20px 20px; float: right; text-align: justify;" cellpadding="4" cellspacing="1" border="2" align="right" | {| style="color:black; margin: 20px 0px 20px 20px; float: right; text-align: justify;" cellpadding="4" cellspacing="1" border="2" align="right" | ||
! colspan="2" style="background:#66bbff;"| Part Information | ! colspan="2" style="background:#66bbff;"| Part Information | ||
| Line 8: | Line 12: | ||
|- | |- | ||
|'''Fused Tag''' | |'''Fused Tag''' | ||
| − | |[https://parts.igem.org/Part: | + | |[https://parts.igem.org/Part:BBa_K2273016 BBa_K2273016: SpyTag His-tagged] |
|- | |- | ||
|'''Fluorescent Protein''' | |'''Fluorescent Protein''' | ||
| Line 16: | Line 20: | ||
|[http://2017.igem.org/Team:TU_Dresden TU Dresden] | |[http://2017.igem.org/Team:TU_Dresden TU Dresden] | ||
|} | |} | ||
| − | |||
<partinfo>BBa_K2273040 short</partinfo> | <partinfo>BBa_K2273040 short</partinfo> | ||
| − | This composite part was used for evalutaion in the [http://2017.igem.org/Team:TU_Dresden/Project/Secretion secretion project] of 2017 TU_Dresden iGEM [http://2017.igem.org/Team:TU_Dresden team]. It codes for a fluorescent reporter protein ([https://parts.igem.org/Part: | + | This composite part was used for evalutaion in the [http://2017.igem.org/Team:TU_Dresden/Project/Secretion secretion project] of 2017 TU_Dresden iGEM [http://2017.igem.org/Team:TU_Dresden team]. It codes for a fluorescent reporter protein ([https://parts.igem.org/Part:BBa_K2273034 mCherry]) and a functional tag ([https://parts.igem.org/Part:BBa_K2273015 SpyCatcher]), mediating covalent bonding with its tag partner ([https://parts.igem.org/Part:BBa_K2273014 SpyTag]). It is optimized for usage in <i>Bacillus subtilis</i>. |
===Design=== | ===Design=== | ||
| Line 27: | Line 30: | ||
A signal peptide ([https://parts.igem.org/Part:BBa_K2273023 AmyE SP]) was fused n-terminally to this construct to induce secretion. | A signal peptide ([https://parts.igem.org/Part:BBa_K2273023 AmyE SP]) was fused n-terminally to this construct to induce secretion. | ||
| − | [[File:SfGFP | + | [[File:SfGFP tag.png|thumb|centre|600px|'''Figure 1:''': Genetic constructs with sfGFP. Depicted are translational fusion constructs downstream of the PxylA promotor, that were cloned in the multiple cloning site of the pBS2EPxylA vector. The constructs contain a signal peptide sequence, the gene coding for sfGFP, either c- or n-terminally fused SpyTag or mini. SpyCatcher and a his-tag.]] |
| Line 39: | Line 42: | ||
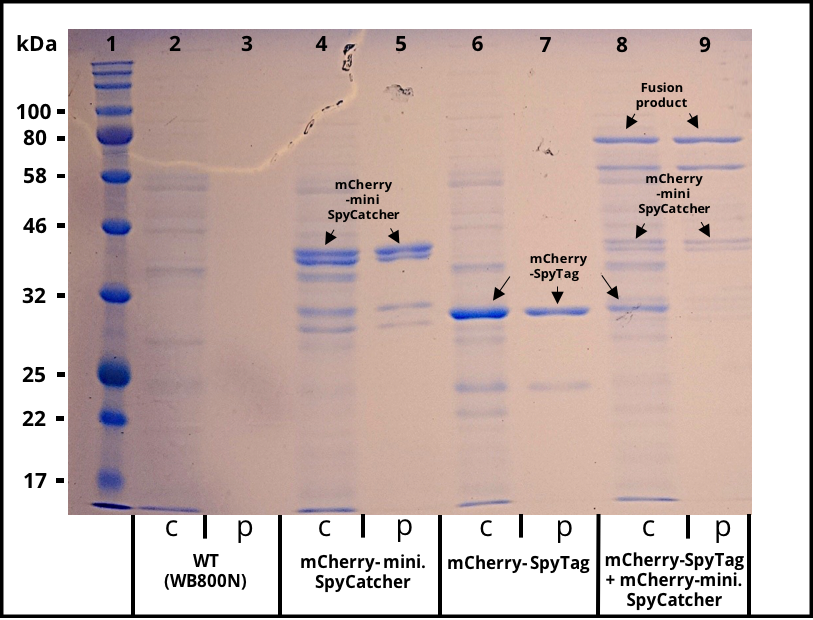
| − | The successful secretion could be proven with a fluorescence assay using the supernatants of <i>B. subtilis </i> (Figure 2 | + | The successful secretion could be proven with a fluorescence assay using the supernatants of <i>B. subtilis </i> (Figure 2). The functionality of the SpyTag/SpyCatcher was proven via SDS-PAGE, using the supernatants (Figure 3). |
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| + | [[File:T--TU Dresden--secretion--Figure1.png|thumb|right|450px|'''Figure 2:''': Endpoint measurement of the fluorescence from supernatants carrying our constructs and the wild type. Expression of the single copy mCherry or sfGFP fusion SpyTag/SpyCather constructs (purple) was induced with 1% xylose and the supernatants were harvested after 16 h of incubation. Wild type supernatant is shown as a control (pink). Excitation wavelength for sfGFP was set to 480 nm and emission was recorded at 510 nm and for mCherry excitation wavelength was set to 585 nm and emission was recorded at 615 nm . The fluorescence was normalized by the optical density (OD600). Graph shows mean values and standard deviations of at least two biological and three technical replicates.]] | ||
| − | [[File:T--TU_Dresden--secretion---result--sds.png|thumb| | + | [[File:T--TU_Dresden--secretion---result--sds.png|thumb|left|350px|'''Figure 3:''' SDS gel with crude and purified supernatants. Expression of the multi copy mCherry constructs was induced with 1% Xylose and the supernatants were harvested after 16 h of incubation. The his-tagged proteins were purified with Ni-NTA agarose beads. Lane 1 was loaded with 3 µl of NEB´s “Color Prestained Protein Standard Broad Range” ladder. Crude (c) and purified (p) supernatant of wild-type (WT) are shown as a control in lane 2 and 3. Lane 4 and 5 contain the supernatant of B. subtilis producing mCherry-mini. SpyCatcher fusion protein (36,6 kDa). Lane 4 and 5 contain the supernatant of B. subtilis producing mCherry-SpyTag fusion protein (31,9 kDa). The crude supernatants of the two mCherry producing strains were combined, incubated for 4 h, purified and loaded onto lane 8 and 9. The fusion product of the mCherry constructs is visable in the crude and purified supernatant.]] |
Revision as of 16:02, 1 November 2017
| Part Information | |
|---|---|
| RFC standard | RFC 25 |
| Fused Tag | BBa_K2273016: SpyTag His-tagged |
| Fluorescent Protein | BBa_K2273033: sfGFP |
| Submitted by | [http://2017.igem.org/Team:TU_Dresden TU Dresden] |
sfGFP with C-Terminal SpyCatcher and His Tag
This composite part was used for evalutaion in the [http://2017.igem.org/Team:TU_Dresden/Project/Secretion secretion project] of 2017 TU_Dresden iGEM [http://2017.igem.org/Team:TU_Dresden team]. It codes for a fluorescent reporter protein (mCherry) and a functional tag (SpyCatcher), mediating covalent bonding with its tag partner (SpyTag). It is optimized for usage in Bacillus subtilis.
Design
A signal peptide (AmyE SP) was fused n-terminally to this construct to induce secretion.

Application
The successful secretion could be proven with a fluorescence assay using the supernatants of B. subtilis (Figure 2). The functionality of the SpyTag/SpyCatcher was proven via SDS-PAGE, using the supernatants (Figure 3).
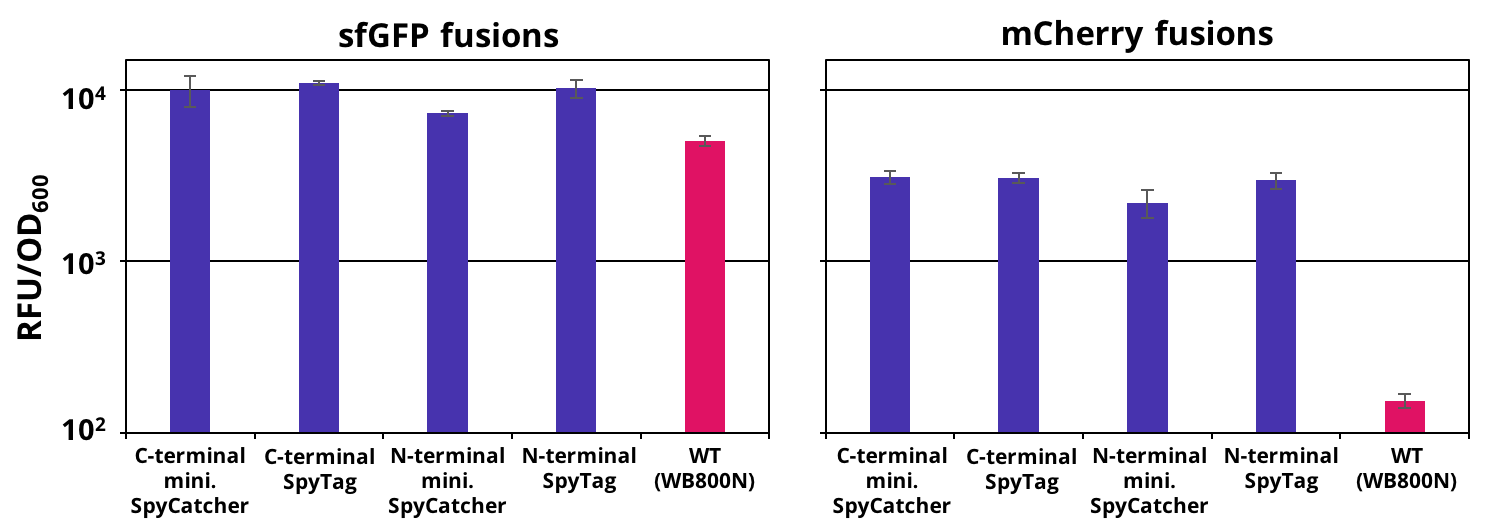
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
