Part:BBa_K1022101
pT7: RBS: His6 - SUMO: MaximinH5
This part codes for the peptide 'Maximin H5' tagged with 'His-6-SUMO' molecule. Its production is triggered by IPTG (through pT7 promoter).
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12INCOMPATIBLE WITH RFC[12]Illegal NheI site found at 46
- 21INCOMPATIBLE WITH RFC[21]Illegal BglII site found at 170
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal AgeI site found at 355
- 1000COMPATIBLE WITH RFC[1000]
Characterization
For more info, visit [http://2013.igem.org/Team:TU-Delft/Peptides#SUMO Peptide production] on TU Delft iGEM13 Wiki
Peptide production
Introduction:
In the project by iGEM 13 Team TU Delft, a SUMO-peptide fusion was opted as a suitable expression system as they make the fusion proteins more soluble. The peptide by itself is not soluble in the cytoplasm but making a fusion of peptide with Small Ubiquitin like Modifiers (SUMO) will increase the solubility of the peptide, thus increasing the cytoplasmic fraction of the peptide.
A gene was constructed in such a way that the SUMO-peptide production was driven by the strong T7 phage promoter. This gene containing plasmid was harboured in a BL21(DE3) strain that has lac promoter driven T7 polymerase. Upon induction by IPTG the SUMO peptide fusion is produced as a soluble protein fraction.
The protocol can be seen [http://2013.igem.org/Team:TU-Delft/Protocol_10#protocol_10 here].
Result:
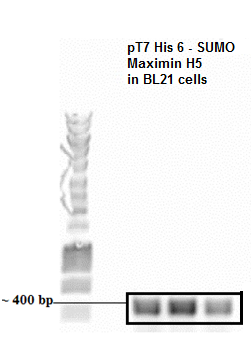
- Colony PCR SUMO Peptide
The presence of plasmid with gene inserts encoding the SUMO-peptide (BBa_K1022101) was confirmed by a colony pcr. The expected size of the insert with the T7 promoter was approximately around 400 bp which was clearly evident from the agarose gel picture. An Eurogentec Smartladder MW-1700-10 (https://secure.eurogentec.com/uploads/TDS-MW-1700-10.pdf) was used to identify the size of the fragments.
Discussion:
Maximin H5 did not give convincing MIC results(seen below), thus making it less interesting to characterize further. Nevertheless, the peptide production of Signiferin by Biobrick BBa_K1022102 is representative of the peptide production of Maximin H5 tagged with SUMO protein.
Highlights:
The SUMO domain that was used in our project was synthesized as a gene from DNA 2.0. The SUMO was designed to be bio-brick compatible without the restriction sites that are present in the prefix and suffix. The restriction sites where removed without disturbing the codons.
The Biobrick was designed in such a way that the ease of cloning SUMO fusion protein to be expressed has been improved. An Age I restriction site was introduced at the end of SUMO which when translated to amino acids Tyrosine Glycine Glycine. This is the recognition sequence of the Ulp-1 [2]protease that can cleave the peptides from the SUMO without any scar. So, one can use Age I and any one of the suffix enzyme to make a SUMO fusion.
Peptide Characterization (MIC)
Introduction:
An important part of the project TU Delft iGEM13 is inhibition of growth or killing of bacteria with the use of antimicrobial peptides (AMPs). In order to get an idea of the toxicity of our peptides we conducted several minimal inhibiting concentration (MIC) experiments with synthetic peptides ordered from DNA 2.0. These MIC measurements where done on E. coli, B. subtilis and S. delphini, with the first as representative of our Gram-negative expression host, the second for the Gram-positive targets and the last for our specific target.
MIC determination:
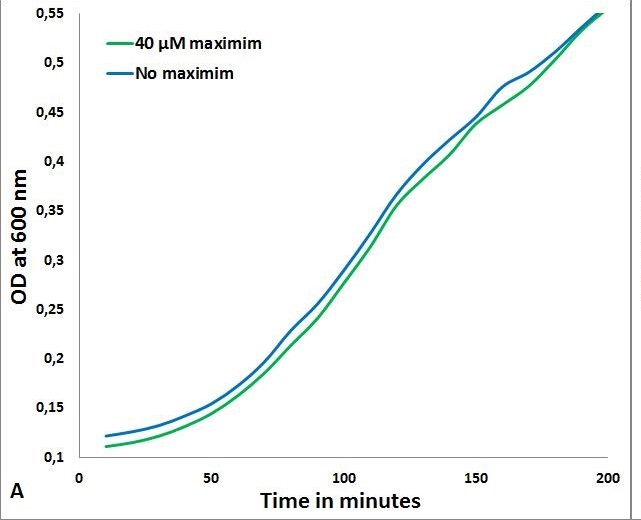
The MIC of Maximin H5 on S. delphini, B. subtilis and E. coli were done according to the protocol described [http://2013.igem.org/Team:TU-Delft/PeptideCharacterization here]. Maximin H5 did not give a measurable reduction in growth below 40µM, making us decide not to proceed testing, as modeling showed it was not possible to reach these concentrations through expression in E.coli.
- MIC of Maximin H5 on S. delphini
For Other peptides and their characterization please visit [http://2013.igem.org/Team:TU-Delft/PeptideCharacterization Peptide characterization] on TU Delft iGEM13 Wiki
Comparison with two peptides:
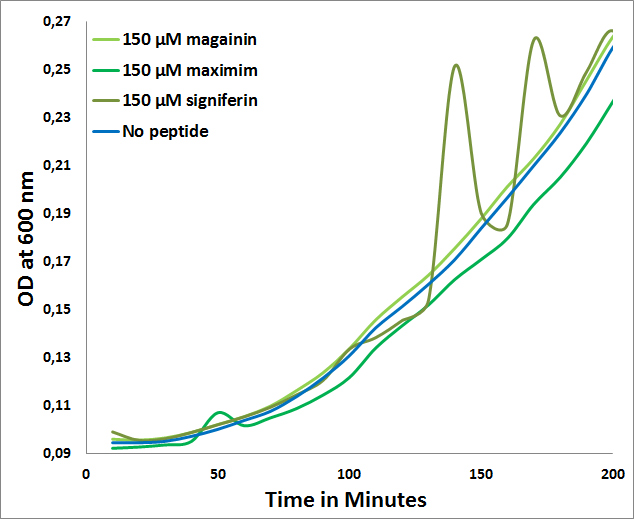
The fact that for none of the peptides a MIC could be determined for E. coli (>150µM) further confirms the expected selectivity towards Gram-positives which is observed in the following graph:
- MICs of Maximim-H5, Signiferin and Magainin on E. coli
For more info, visit [http://2013.igem.org/Team:TU-Delft/PeptideCharacterization Peptide characterization] on TU Delft iGEM13 Wiki
SUMO-Peptide cleavage by Ulp1 protease
For more info, visit [http://2013.igem.org/Team:TU-Delft/Peptides#cleavage SUMO Cleaved by Ulp 1] on TU Delft iGEM13 Wiki
The construct used for testing this is: BBa_K1022116
References
1. Panavas T, Sanders C, Butt TR.. (2009). SUMO fusion technology for enhanced protein production in prokaryotic and eukaryotic expression systems. Methods in Molecular Biology. 10 (8), p4-20.
2. Lee, C., Sun, H., Hu, S., Chiu, C., Homhuan, A., Liang, S., Leng, C. and Wang, T. 2008. An improved SUMO fusion protein system for effective production of native proteins. Protein Science, 17 (7), pp. 1241--1248.
| n/a | pT7: RBS: His6 - SUMO: MaximinH5 |