Part:BBa_K404004
[AAV2]-Rep-VP13(ViralBrick-587KO-empty)_p5-TATAless
Replication
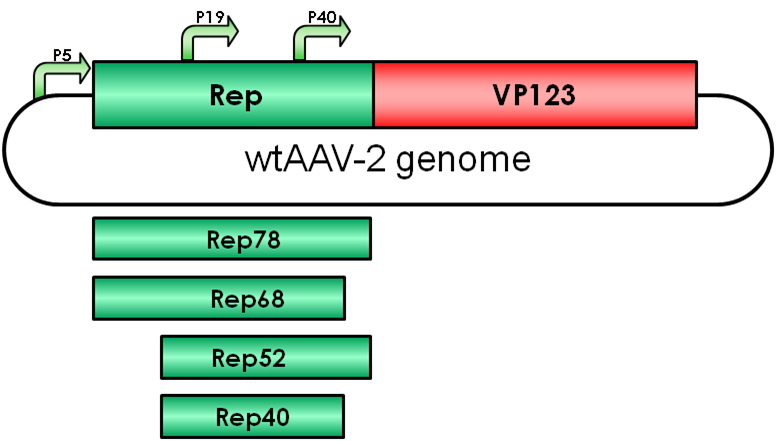
The Adeno-associated virus (AAV) consists of two open reading frames (ORF), rep and cap ORF. The four non-structural rep genes are driven by two promoters located at map units 5 (p5 promoter) and 19 (p19 promoter). Rep proteins are involved in genome encapsidation (reference), regulation of gene expression (reference) and replication of the viral genome (reference).
The two larger proteins Rep78/68 play an essential role in viral genome integration and regulation of AAV gene expression, whereas the smaller Rep proteins are involved in viral genome encapsidation. Rep proteins act both as repressors and activators of AAV transcription in respect to the absence and presence of helper viruses such as adenoviruses (Ad) or herpes simplex viruses (HSV) by interacting with several cellular proteins (Nash, Chen, Salganik, & Muzyczka, 2009).
Furthermore, in the absence of Rep proteins, as it is the case in recombinant AAVs, integration of the viral genome into the human genome is rare and random. There are several hotspots for integration of wtAAV genomes such as the human chromosome 19q13.42, known as the AAVSI site, but as well some other accessible chromatin regions for preferred integration have been found (5p13.3 and 3p24.3).
Integration into the human genome is mediated by the two regulatory proteins Rep68 and Rep78 driven by the AAV p5 promoter. The proteins bind to the Rep binding site (RBS) which is located within the inverted terminal repeats (ITRs). The minimal consensus Rep binding site (RBS) GAGT GAGC is found within the ITRs and in the p5 integration-efficient element (p5IEE) of the p5 promoter (Hüser et al., 2010). Rep78/68 proteins possess DNA-binding (reference), helicase (reference) and site-specific endonuclease activity located within the first 200 amino acids (Davis, Wu, & Owens, 2000). Since the N-terminal region is unique to the larger Rep proteins, the two smaller Rep proteins possess other biological functions.
Rep52/40 gene expression is driven by the p19 promoter which is located within rep ORF and the proteins are involved in encapsidating the viral genome into the preformed capsids.
Gene expression of these proteins is suppressed in absence of adenovirus infection by binding of Rep78/68 to the p5 promoter. Gene expression of p19 and p40 is transacvtivated by the Rep proteins Rep78/68 during coinfection.
Rep78
Regulated by the p5 promoter, Rep78 is the largest non-structural protein found in the wtAAV. Besides regulation of gene expression and viral genome replication, Rep78 has been found to play a functional role in AAV site-specific integration into the human genome (Hüser et al., 2010). In absence of Ad helper viruses, overexpression of Rep78 leads to cell cycle arrest by interacting with cell-cycle regulating phosphatases causing DNA damage by its intrinsic endonuclease activity (Berthet, Raj, Saudan, & Beard, 2005) and induces apoptosis. Due to its ability to bind to the Rep binding site (RBS) in the p5 integration-efficient element (p5IEE) of the p5 promoter, Rep78 mediates gene expression and retain a constant level of Rep proteins by suppressing transcriptional activity of the p5 promoter in absence of Ad viruses (Yue et al., 2010). Interaction of Rep78 with cellular factors such as transcription factors (Lackner & Muzyczka, 2002) provides the basis for gene regulation by Rep78 in associated with endogenous molecules.
Rep68
Rep68 is a regulatory protein driven by the p5 promoter with an apparent molecular weight of 68 kDa lacking 92 amino acids from the carboxy terminus due to splicing of mRNA coding for the two larger Rep proteins.
The non-structural protein Rep68 belongs to the superfamily 3 (SF3) helicase found in other small DNA and RNA viruses such as simian virus 40 (SV40) and bovine papillomavirus (Mansilla-Soto et al., 2009). Formation of oligomeric complexes of Rep proteins provides the basis for the functional versatility of the two larger regulatory proteins. The AAA+ motor domain is known to function as an initiator for oligomerization of the Rep proteins. The cooperative effect of both domains appears to be further regulated by ATP binding as well as different DNA substrates such as dsDNA and ssDNA. Assembly of different nucleoprotein structures suggest that viral replication and genome integration is regulated and controlled by distinct Rep complexes which means that in presence of dsDNA Rep68 assembles to smaller complexes than in presence of ssDNA resulting in octamers.
Rep52
Involved in genome encapsidation
Rep 52 is under the control of the p19 promoter and shares the same N-terminus with Rep78. It was shown that Rep52 possesses helicase and ATPase activity with 3´-5´polarity (Smith & Kotin, 1998). Despite the helicase activity, Rep52 and Rep78 share a putative zinc-finger domain, which suggest interactions with diverse cellular factors (Nash, Chen, Salganik, & Muzyczka, 2009) such as transcription factors (Lackner & Muzyczka, 2002) and TATA-binding proteins (Hermonat, Santin, Batchu, & Zhan, 1998).
Rep40
The smallest Rep protein (Rep40) possesses helicase and ATPase activity as well, but does not have strict requirements for DNA duplexes containing a 3´single-stranded end. Rep40 helicase activity requires bivalent ions such as Mg2+ or Mn2+ and is most active using ATP as substrate. Lacking the zinc finger domain, present in Rep52, Rep40 requires dimerization for functional helicase activity (Collaco, Kalman-Maltese, Smith, Dignam, & Trempe, 2003). Rep40/52 proteins are required for translocation of the single-stranded, viral genomes into the preformed capsids proceeding with the 3´end of the DNA (King, Dubielzig, Grimm, & Kleinschmidt, 2001).
Capsid
p5TATAless promoter
In contrast to the natural location of the p5 promoter, the iGEM team Freiburg 2010 provides the RepCap plasmid with a relocated p5 promoter downstream of the RepCap genes (Figure 6). Additionally the p5 promoter lacks the TATA box element (AVIGEN, 1997). Those modifications result in an attenuated expression of the larger Rep proteins therefore leading to normal transcription of the Rep proteins driven by p19 promoter and enhanced expression of the Cap proteins, which are under the control of the p40 promoter. Additionally, removing the p5 promoter downstream of the RepCap genes and deletion of the TATA box eliminates contamination with wtAAVs. Hence, alteration of the p5 promoter is useful for enhanced production of recombinant viral particles attenuating repression of Rep78/68 and improving gene transcription of the capsid proteins and Rep proteins involved in genome packaging.
HSPG-KO
The primary receptor of AAV-2 is the heparan sulfate proteoglycan (HSPG) receptor (Perabo et al. 2006). Its binding motif consists of five amino-acids located on the capsid surface (Trepel et al. 2009).
HSPG belongs to the glycosaminoglycanes as well as heparin and consists of heparan sulfate glycosaminoglycan attached to a core protein and can be found on every human cell surface.
Its acid residues bear negative charges and are therefore prone to electrostatic interactions with e.g. the positively charged HSPG binding motif of AAV-2. Other interactions with polar residues are possible, too.
Regarding AAV-2, two point mutations in AAV-2 (R585A and R588A) are sufficient to eliminate heparin binding. (Opie et al. 2003). The biobricks with this knockout are annotated with „HSPG-ko“.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BamHI site found at 3611
Illegal XhoI site found at 1913
Illegal XhoI site found at 2099 - 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000INCOMPATIBLE WITH RFC[1000]Illegal BsaI site found at 4137
Illegal BsaI site found at 4335
Illegal BsaI site found at 4372
Illegal SapI site found at 3048
//viral_vectors
//viral_vectors/aav
//viral_vectors/aav/capsid_coding
| None |