Part:BBa_K4011000
CBM3
CBMs are artificial proteins derived from natural proteins with cellulose-binding functions, such as cellulase. There are three types of CBMs, which are CBMs, CBM1, CBM2, and CBM3. CBM1 is the smallest, whilst CBM3 is the biggest. By fusing CBMs to functionalization proteins, we can achieve modification/functionalization of our bacterial cellulose membrane. This is part in a part collection where we characterize bacterial cellulose modification methods and constructs using CBMs.
The part collection includes: Cellulose binding matrix BBa_K4011000 and BBa_K4011001. CBMs fused with spider silk fibroin BBa_K4011008 and BBa_K4011009. Fused proteins capable of expression and secretion in S. cerevisiae BBa_K4011010 and BBa_K4011011.
This part collection can help and inspire other teams we are trying to achieve modification of cellulose membranes using different modification/functionalization proteins.
Usage and Biology
In nature, CBM3s are expressed as a domain of a protein whose functions require being bound to cellulose, such as cellulase. The structure of CBM3 is displayed in the Characterization section (Protein Data Bank accession: 1NBC).
CBM3 fused with spider silk proteins were first done by Mohammadi et al in 2019, where they tested the changes in physical properties on cellulose fibers after mixing with CBM3-spider silk. (Mohammadi et al, 2019)
Characterization
In order to modify BCM’s physical properties, we designed and expressed spider silk fibroins fused with cellulose binding matrixes (CBMs; learn more on our description page) to bind to BCM (Fig. 1). For our project, we experimented with CBM3 from Ruminiclostridium thermocellum (Protein Data Bank (PDB) accession: 1NBC; Fig. 1D) (2) and CBM2 from Cellulomonas fimi (PDB accession: 1EXG; Fig. 1C). For our spider silk protein, we chose to use the synthetic mini spider silk protein NT2RepCT (2Rep; first characterized by GreatBaySZ_2019). 2Rep is water-soluble due to hydrophilic interactions of protein N-terminal and C-terminal. When 2Rep is submerged in a coagulating bath and subjected to a shear force, the repetitive regions will uncoil, form beta-pleated sheet networks and solidify into silk fiber (Fig. 1A).
For adding CBM3 flanking to 2Rep, we synthesized CBM3-BsaI-CBM3 on a pET28a vector. Primers were then used to add BsaI restriction sites in 2Rep to fuse the respective domains together in the synthesized pET28a vector by Golden Gate assembly (Fig. 2A & 2B). After construction, the plasmids were transformed into E. coli BL21(DE3) for IPTG-inducible expression (Fig. 2C).
Comparing the solubility of CBM2/3 fused with 2Rep
2Rep was shown to possess good water solubility, but we’re uncertain whether our modification will change this desirable property. To test the solubility, we cultured the modified strains, induced the expression of the constructs, collected the cells and performed SDS-PAGE on cell lysate (Fig. 2). Expression of both constructs, CBM3-2Rep-CBM3 (72kDa) and CBM2-2Rep-CBM2 (65kDa), are observed. CBM2-2Rep-CBM2 was present in the whole cell sample, but absent in the cell lysate supernatant, indicating poor water solubility. In contrast, CBM3-2Rep-CBM3 was present in both whole cell and supernatant. Therefore, CBM3-2Rep-CBM3 was chosen for the rest of the project for its superiority in water solubility.

Purification of CBM3-2Rep-CBM3
We purified CBM3-2Rep-CBM3 via the fused his-tag (Fig. 3A), and followed up with BCA assay to measure the yield of our modified spider silk protein. The concentration of CBM3-2Rep-CBM3 is approximately 115.78mg/L (Fig. 3B).
We also constructed a 2Rep plasmid capable of expression in E. coli BL21(DE3) (Fig. 4A). We expressed 2Rep alongside CBM3-2Rep-CBM3 and purified both proteins (Fig. 4B & C). CBM3-2Rep-CBM3 had a concentration of 133.08mg/L, which was higher than 2Rep’s concentration of 98.44mg/L (Fig. 4D). Both proteins demonstrated great water solubility.

Measuring physical properties of BCM mixed with 2Rep and CBM3-2Rep-CBM3
After purification of both 2Rep and CBM3-2Rep-CBM3, we went on to test whether the binding of additional spider silk layer can enhance the ability of BCM enduring pulling and the softness of our material. To bind our protein to BCM and to prevent rehydration of BCM, we added protein elute to dried BCM, soaked BCM in ethanol and subsequently soup water, and dried the BCM.
We compared the maximal force of BCM added with 2Rep or CBM3-2Rep-CBM3 We found statistical significant increase in maximal force endured when BCM mixed with CBM3-2Rep-CBM3.We then compared the effect of adding different quantities of 2Rep and CBM3-2Rep-CBM3 on maximal force of BCM (Fig. 5C & D), and found a one-fold increase from raw BCM to BCM with 5mg 2Rep and 1.5-fold increase from raw BCM to BCM with 5mg CBM3-2Rep-CBM3. We noticed that a statistically significant increase in maximal force endured will only be observed after adding at least 3.4mg of CBM3-2Rep-CBM3.

We then compared the softness of BCM mixed with different quantities of 2Rep and CBM3-2Rep-CBM3 (Fig. 6B & 6C). Both comparisons showed around 35% increase between raw BCM and BCM with 3.4mg of protein. Same amounts of 2Rep and CBM3-2Rep-CBM3 generally achieved similar results, indicating that softness is not affected by the addition of CBM3.
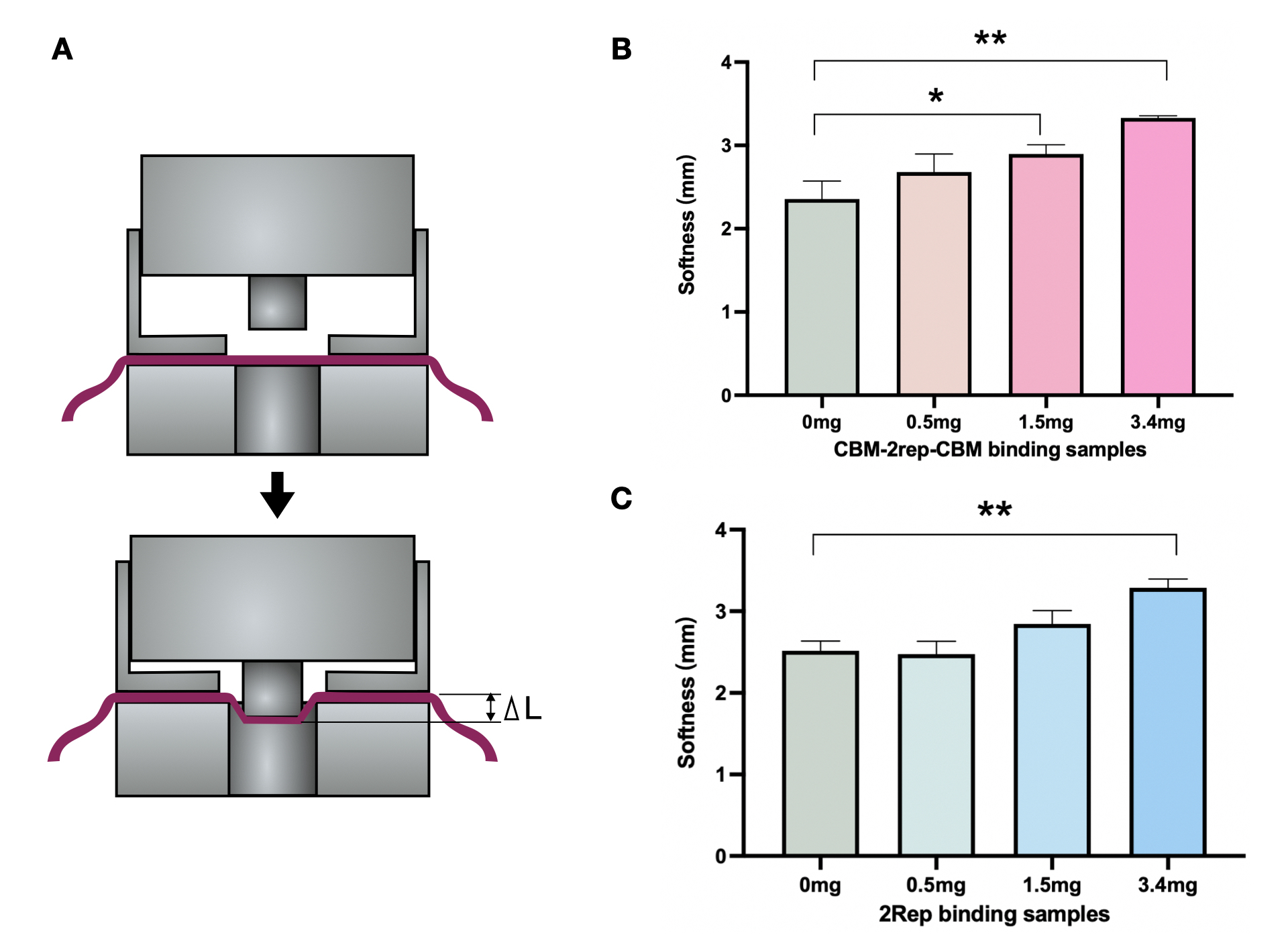
Overall, we have demonstrated that adding 2Rep and CBM3-2Rep-CBM3 can significantly improve the physical characteristics of BCM, with CBM3-2Rep-CBM3 outperforming 2Rep in improved maximal force. This demonstrates the viability of spider-silk-BCM composite material to be used as a leather substitute.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal AgeI site found at 91
Illegal AgeI site found at 301 - 1000COMPATIBLE WITH RFC[1000]
CONTRIBUTIONS: LINKS-China 2024 | CBM3-sfGFP
Authors iGEM Links China 2024, Tong Ding.
Carbohydrate-Binding Module 3 (CBM3 BBa_K4011000) is a protein domain found in carbohydrate-active enzymes, particularly those involved in cellulose degradation. CBM3 enhances the ability of these enzymes to bind to their polysaccharide substrates, improving catalytic efficiency. By fusing CBM3 to functional proteins, we can target cellulose-containing materials for modification or degradation. We chose the chromoprotein sfGFP BBa_K592011 to be fused with CBM3 to enhance visualization of CBM3's protein binding abilities.
Usage and Biology
In nature, CBM3 is expressed as a domain within cellulases and other glycoside hydrolases that degrade cellulose, a major component of plant cell walls. The binding of CBM3 to cellulose increases catalytic efficiency by creating a better fit between the enzyme and substrate surfaces. The structure of CBM3 is documented in the Protein Data Bank (accession: 2VZC). sfGFP is a chromoprotein that emits bright green fluorescence, commonly used in molecular biology applications. This protein serves as a visual marker, enabling researchers to track and confirm the expression of fusion proteins in various experimental setups. By incorporating sfGFP into our constructs, we can enhance the visualization of protein localization and interactions, thereby improving the overall effectiveness of our assays. CBM3 is applicable in biotechnological processes such as biomass conversion, biofuel production, and the recycling of plant materials.
Source
CBM3-sfGFP: This wiki.
Results

We obtained a fusion protein composed of a binding domain protein linked to a fluorescent protein, naming it CBM3-sfGFP. Figure A shows the results of the SDS-PAGE analysis of the target fusion proteins (binding domains linked to chromoproteins). TheCBM3-sfGFP fusion protein was expressed in E. coli BL21 (DE3). CBM3-sfGFP has a molecular weight of 46 kDa and is successfully expressed. The color of CBM3-sfGFP suspended in 20mM Tris-HCl under bluelight is shown in Figure B, expressing the correct color green. CBM3's ability to bind with cellulose is assessed, as shown in Figure C. The results indicate that CBM3 can successfully bind to cellulose material, as shown by the comparative brightness in color under bluelight.
References
Pezhman Mohammadi, A. Sesilja Aranko, Christopher P. Landowski, Olli Ikkala, Kristaps Jaudzems, Wolfgang Wagermaier, & Markus B. Linder (2019). Biomimetic composites with enhanced toughening using silk-inspired triblock proteins and aligned nanocellulose reinforcements. Science Advances, 5(9), eaaw2541.
| None |


