Part:BBa_K2278021
D-NY15 Antimicrobial peptide with Alpha-Factor Secretion Signal
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal XhoI site found at 244
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
Introduction
This DNA biobrick was designed in order to produce an antimicrobial peptide from the crocodile Crocodylus siamensis.
1- Biological background
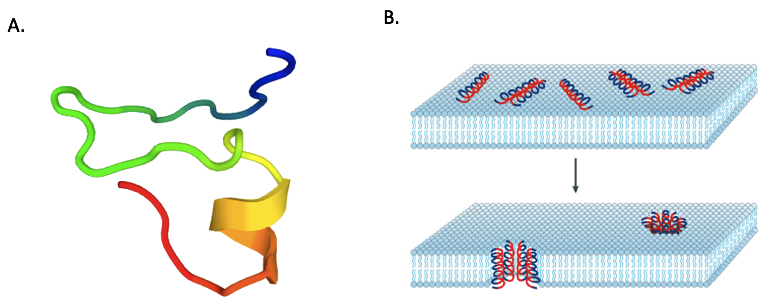
Antimicrobial peptides (AMP) are phylogenetically ancient components of the innate defense of both invertebrates and vertebrates. In the context of growing bacterial antibiotic-resistance, these AMP are considered as potential new therapeutical candidates.The D-NY15 construction was based on the small peptide Leucrocin I sequence (NGVQPKY ; Yaraksa et al., 2013). Leucrocin I
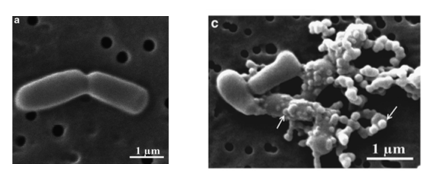
The mechanism of action of D-NY15 has been observed by transmission electron microscopy. The AMP binds and inserts to bacterium membranes to create pores, leading to the lysis of the cells (figure 2).
</html>
2- Usage in iGEM projects
The part was designed during the Croc’n Cholera project (team INSA-UPS-France 2017). It contains the coding sequence to produces the D-NY15 AMP with a yeast promoter. The α-factor (BBa_K1800001) sequence contains a RBS and a signal sequence to secrete the produced peptides.Experiments
1- Molecular biology
The gene was placed under the control of an alpha factor signal. IDT performed the DNA synthesis and delivered the part as gBlock. The construct was cloned by conventional ligation into the pSB1C3 plasmid. The construction was then inserted on plasmid pPICZa and integrated in the yeast genome.
Analysis of the restriction map
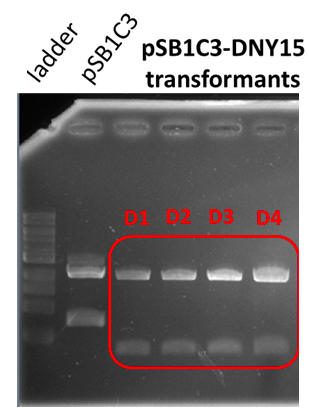
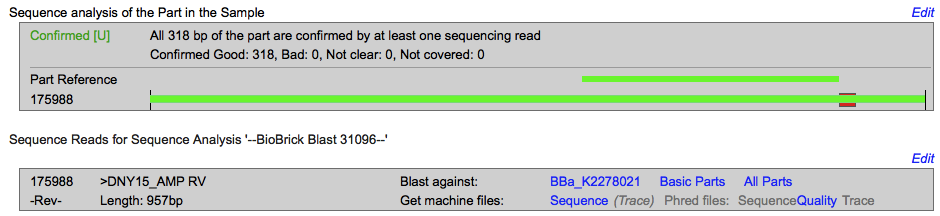
Sequencing
The sequencing successfully validated the sequence of the biobrick.
2- Integration in Pichia pastoris
The biobrick was placed under the control of the constitutive pGAP promoter (BBa_K431009) and was cloned in the pPICZalpha vector, an expression vector for the yeast Pichia pastoris. The plasmid was then linearized and transferred in Pichia pastoris by electroporation. The integration is predicted to be at the pGAP location. Indeed, the pGAP promoter makes genome recombination easier in Pichia pastoris.
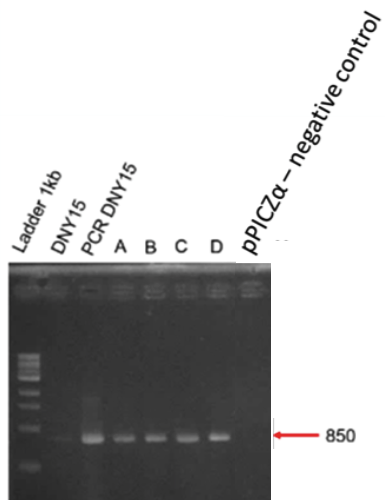
Correct amplifications were observed for the 4 colonies tested (A to D) and the positive controls with the D-NY15 fragment as matrix (DNY15) or the pPICZalpha-D-NY15 plasmid (PCR DNY15). Negative control with pPICZalpha presented no band, as expected.
Characterization
D-NY15 production was performed with Pichia pastoris in YPD 40 g/L glucose grown for 4 days at 30 °C with shacking. Cells and supernatants were sampled.
1- Validation of D-NY15 expression in Pichia pastoris by qRT-PCR
RNA from the Pichia pastoris cells were extracted and qRT-PCR was performed (figure 6).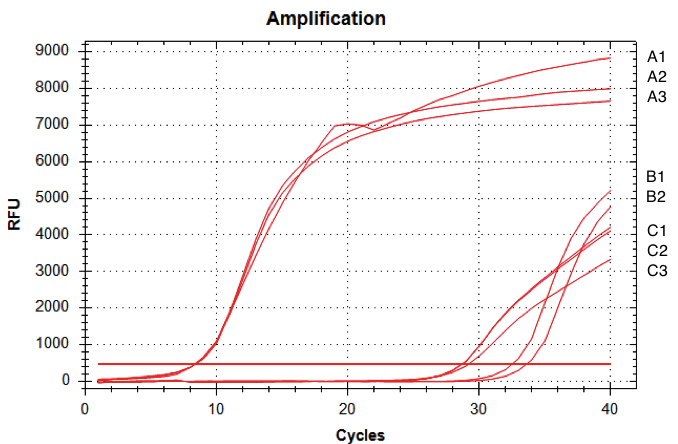
The amount of fluorescence provided by the qRT-PCR with the D-NY15 primers rose after 8 cycles whereas the negative control (pPICZalpha only) started to be amplified at over 29 cycles (non specific amplification). This means that the D-NY15 encoding gene is expressed in Pichia pastoris.
2. Toxicity assay
The supernatants of Pichia pastoris strains with or without the D-NY15 encoding gene were used in a halo assay against V. harveyi as the target of AMPs. Briefly, 35mL of supernatants were freeze-dried and then resuspended in 3.5mL of water. A paper cut was soaked with one of these solutions and placed on a Petri plate inoculated with V. harveyi (figure 7).
Conclusion :
Halo are formed around the positive control and the patch containing supernatants from a D-NY15 expressing strain. This nicely demonstrated the capacity of D-NY15 to inhibit the growth of Vibrio harveyi. Here, we show both the capacity of Pichia pastoris to produce antimicrobial peptides and the efficiency of the crocodile peptide D-NY15 to inhibit Vibrio harveyi growth.Perspectives:
To go further, inhibition test should be now performed in liquid assay to be closer to the conditions in which we want to use these strains.Design Notes
Part:BBa_K1800001: Alpha-Factor Secretion Signal
Source
The peptides DNA sequence has been obtained by reverse translation of the amino acid sequence of the D-NY15 proposed by Yaraksa et al., 2013, which was determined by mass spectrometry analysis.
References
Yaraksa, N., Anunthawan, T., Theansungnoen, T., Daduang, S., Araki, T., Dhiravisit, A. and Thammasirirak, S. (2013). Design and synthesis of cationic antibacterial peptide based on Leucrocin I sequence, antibacterial peptide from crocodile (Crocodylus siamensis) white blood cell extracts. The Journal of Antibiotics, 67(3), pp.205-212.
Brogden KA (2005) Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria. Nat Rev Microbiol. 3:238-50.| None |