Part:BBa_K771305
TesA with Membrane Anchor 2 (without MS2)
BBa_K771305:TesA is linked to C terminal of BBa K771007 by Flexible Linker 3(FL3).TesA is a cytoplasmic mutant of the periplasmic thioesterase. It is capable of releasing free fatty acids, preventing the fatty acids from being directly harnessed for phospholipid biosynthesis.
Fatty acid Biosynthetic Pathway
TesA involves in the biosynthesis of fatty acids in E.coli. The biosynthesis pathway is shown as below:
The E. coli fatty acid synthesis is initiated when holo-ACP, NADPH and NADH, acetyl-CoA and malonyl-CoA undergo condensation and subsequent reduction to form butyryl-ACP. These reactions are catalyzed by the malonyl-CoA:ACP transacylase FabD, the ketosynthase FabH, the NADPH-dependent ketoreductase FabG, either the dual-function dehydratase/isomerase FabA or the monofunctional dehydratase FabZ, and the NADH-dependent enoyl reductase FabI. Then the butyryl-ACP is extended via 5-7 rounds of analogous reactions to produce a C14 to C18-ACP either fully saturated or monounsaturated. These extension cycles are catalyzed by either the ketosynthase FabB or FabF in collaboration with FabD, FabG, FabA or FabZ, and FabI. Finally, the full-length fatty acid is released from the corresponding fatty acyl-ACP via hydrolysis by C16-specific thioesterase,TesA.
Result
The solo introduce of TesA with Membrane Anchor 2 (without MS2)
As Fig.2 A indicates, 20 hours after induction, E.coli with membrane anchored TesA experienced a 50% increase in C-16, C-18 fatty acids content and total fatty acids (Fig.4 B) in supernatant, compared with E.coli with free TesA.Fatty acids yielded from supernatant went up to 0.71mg/(L·OD). The result support that membrane anchored TesA could efficiently transfer fatty acyl-ACP into desirable fatty acids right beneath inner membrane, thus making it much easier for the final products to diffuse into periplasmic space.
Fig.3, on the other hand, shows fatty acids content in sedimentation also went up by 40%, up to 5.02mg/(L·OD). The result is still within our expectation since fatty acyl-ACP has been removed from the reaction system to form fatty acids. As a result, the chemical equilibrium shifts and more fatty acids would be accumulated.
The cooperation of enzymes with membrane anchors
TesA with Membrane Anchor 2 (without MS2), FabG with Membrane Anchor 3, FabZ with Membrane Anchor 4, FabI with Membrane Anchor 4(without VVD) could significantly enhance the production of fatty acid by 9 fold compared with enzymes without membrane anchor.
To optimize the productivity of the system we established, we tended to combine these two privileges together, gathering TesA, FabG, FabI and FabZ through receptor-ligand interaction. Notable increase in both diversity and amount of fatty acids were detected.
Cluster of FabG, FabI and FabZ provides C16- and C18 specific TesA with sufficient amount of fatty acyl-ACP to hydrolyze and release. Therefore, we witnessed a tremendous growth in the turnover of fatty acids with C16 and C18 skeleton in the supernatant.(Fig.3 A)
Moreover, fatty acids with C14 skeleton was first detected in E.coli expressing membrane anchored enzymes compared with ones with free enzymes. It is probably because the productivity of cluster of FabG, FabI and FabZ overloads itself when carbon chain growth slows down as it elongates. Monounsaturated fatty acids also emerge in considerable amount for the first time since TesA is located so closely to the cluster of FabG, FabI and FabZ that it catches intermediate with C16 and C18 skeleton even before they are reduced.(Fig.3 C)
Augment in both diversity and amount of fatty acids led to 24 fold increase in total yield compared with wild type and 9 fold with ones expressing free enzymes. (Fig.6 B) The exciting result convincingly proves that membrane enhances receptor-ligand interaction and cluster of enzymes makes it faster to a surprising extent. Products acclumulates near inner membrane and traval a shorter distance to diffuse through membrane.
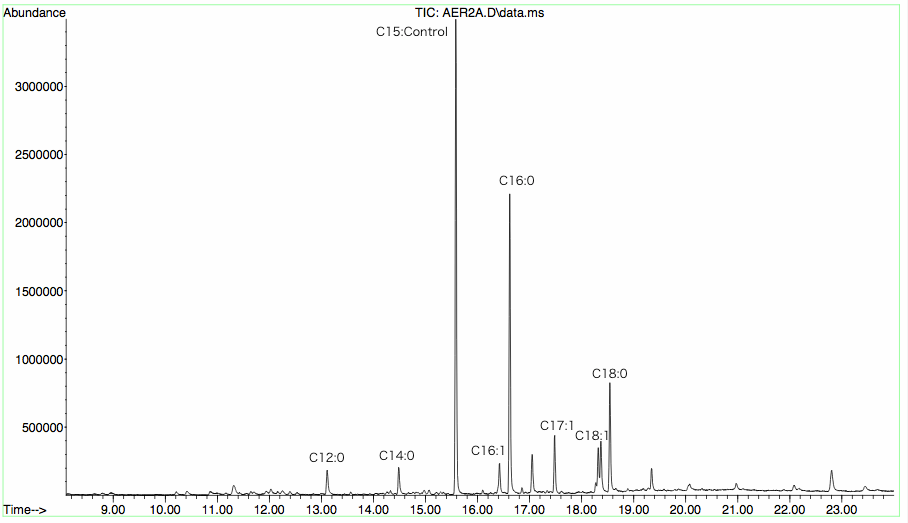
GC-MS
<br\><br\><br\><br\><br\>
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal AgeI site found at 884
- 1000INCOMPATIBLE WITH RFC[1000]Illegal BsaI site found at 505
Illegal SapI.rc site found at 1151
| n/a | TesA with Membrane Anchor 2 (without MS2) |