Part:BBa_K3398001
Enhanced T7 promoter 2
This part is a T7 promoter derived from BBa_I712074 with some point mutation according to previous research [1], and as a higher expression efficiency relative to BBa_I712074 by the fluorescence intensity when expressing EGFP. To express BioBricks under the control of this part, a T7 polymerase gene is needed in the chassis.
Usage and Biology
T7 promoter is very specific promoter which is transcribed only by specific T7 RNA polymerase. Usually this promoter is used in expression systems where T7 promoter is cotransfected with T7 RNA polymerase. That ensures strong transcription of desired genes.
Characterization
Result
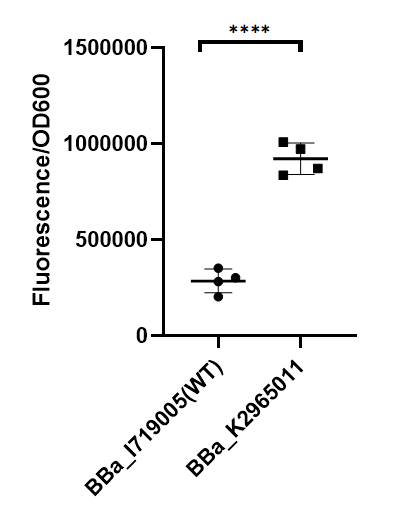
To test the function of mutant promoters, we chose EGFP as our reporter gene.When the E.coli BL21 was cultured at the OD600 between 0.6 to 0.8, we added 1 mM IPTG to induce the expression of EGFP for 20 hours. By assessing the absolute fluorescence and OD600, we can conclude the relative strength of all promoters. Finally we screened out 3 mutants with higher intensity.(BBa_K3398000, BBa_K3398001, BBa_K3398002).
As we can see from the figure, our mutant promoter showed largely increased strength compared with wild type T7 promoter. Therefore, our mutant promoter offer users more opportunity to control the expression of protein using T7 promoter and permit higher levels of target protein expression to be obtained.
Protocol
1. Use BamHI and XhoI to insert eGFP fragment into pET28 plasmid.
2. Linearize pET28-eGFP plasmid by PCR. (WT: F-CCCGCTGCAGTAATACGACTCACTATAGGGAGAGGAATTGTGAGCGGATAACAA / R- GCGGTGGACTGCAGCAACTCAGCTTCCTTTCGGGCT ; Mu: F-CCCGCTGCAGTAATACGACTCACWNNNGSRRNNGGAATTGTGAGCGGATAACAA / R- GCGGTGGACTGCAGCAACTCAGCTTCCTTTCGGGCT )
3. Cyclization the plasmid by PstI and ligase.
4. Transform the plasmids into E. coli BL21(DE3), add 20μl 100mM IPTG to the plate before coating the plate.
5. Incubate at 37℃ and observe colony color to screen for strong mutants preliminarily.
6. Pick greener single colonies, add the colonies into 3ml Lb, incubate at 37℃ in a shaker for 6-8h to dilute the IPTG introduced when picking single colonies.
7. Add 10μl germ solution from last step into 3ml Lb, incubate at 37℃ in a shaker until OD600 reaches 0.6.
8. Add IPTG to final concentrations 0.5mM, and induce for 4h at 18℃.
9. Measure the fluorescence (excitation wavelength: 485 nm; detection wavelength: 528 nm) and OD600.
Reference
[1] Ikeda R A, Ligman C M, Warshamana S, et al. T7 promoter contacts essential for promoter activity in vivo[J]. Nucleic Acids Research, 1992, 20(10): 2517-2524. [2] Paul S, Stang A, Lennartz K, Tenbusch M, Uberla K. Selection of a T7 promoter mutant with enhanced in vitro activity by a novel multi-copy bead display approach for in vitro evolution[J]. Nucleic Acids Research, 2013, 41(1):e29.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
| None |