Part:BBa_R0010
promoter (lacI regulated)
This part is an inverting regulator sensitive to LacI and CAP.
It contains two protein binding sites. The first binds the CAP protein, which is generally present in E.coli and is asocciated with cell health and availability of glucose. The second binds LacI protein.
- In the absence of LacI protein and CAP protein, this part promotes transcription.
- In the presence of LacI protein and CAP protein, this part inhibits transcription.
- LacI can be inhibited by [http://openwetware.org/wiki/IPTG IPTG].
- LacI is coded by BBa_C0010
Intrinsic noise value: 0.0707 (compare with R0011: 0.0040; R0051: 0.0869). See [http://2015.igem.org/Team:William_and_Mary William_and_Mary iGEM 2015]
>Internal Priming Screening Characterization of BBa_R0010: Has 3 possible internal priming site between this BioBrick part and the VR primer.
The 2018 Hawaii iGEM team evaluated the 40 most frequently used BioBricks and ran them through an internal priming screening process that we developed using the BLAST program tool. Out of the 40 BioBricks we evaluated, 10 of them showed possible internal priming of either the VF2 or VR primers and sometime even both. The data set has a range of sequence lengths from as small as 12 bases to as large as 1,210 bases. We experienced the issue of possible internal priming during the sequence verification process of our own BBa_K2574001 BioBrick and in the cloning process to express the part as a fusion protein. BBa_K2574001 is a composite part containing a VLP forming Gag protein sequence attached to a frequently used RFP part (BBa_E1010). We conducted a PCR amplification of the Gag-RFP insert using the VF2 and VR primers on the ligation product (pSB1C3 ligated to the Gag + RFP). This amplicon would serve as template for another PCR where we would add the NcoI and BamHI restriction enzyme sites through new primers for ligation into pET14b and subsequent induced expression. Despite gel confirming a rather large, approximately 2.1 kb insert band, our sequencing results with the VR primer and BamHI RFP reverse primer gave mixed results. Both should have displayed the end of the RFP, but the VR primer revealed the end of the Gag. Analysis of the VR primer on the Gag-RFP sequence revealed several sites where the VR primer could have annealed with ~9 - 12 bp of complementarity. Internal priming of forward and reverse primers can be detrimental to an iGEM project because you can never be sure if the desired construct was correctly inserted into the BioBrick plasmid without a successful sequence verification.
For the BioBrick part BBa_R0010, the first location of the internal priming site is on the 121-113 base number of the BioBrick and on the 12-20 base number of the VR primer. The second location of the internal priming site is on the 11-17 base number of the BioBrick and on the 4-10 base number of the VR primer. The third location of the internal priming site is on the 84-90 base number of the BioBrick and on the 14-20 base number of the VR primer.
Usage and Biology
This is a direct copy of bases 0365739 through 0365540 of the E. coli K-12 MG1655 genome, Genbank NC_000913 in reverse complement form. It is the natural promoter for the LacZYA operon. It includes the tail end of the LacI gene coding region, but no promoter region for that partial gene.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
Usage in Chromobacterium Violaceum
[http://2016.igem.org/Team:Tec-Monterrey Team Tec-Monterrey 2016] tested the Promoter BBa_R0010 in C. Violaceum. We were unable to confirm whether or not C. Violaceum had the lac operon, BLAST analysis of its genome suggested that it did not have this regulating system; our characterization demonstrates that the lac promoter can be used as a constitutive promoter in this chassis. There was no significant difference in the Fluorescence of the group of C. Violaceum with IPTG or without it, but in E. Coli it showed normal induction. Therefore, we can confirm that this promoter is constitutive in C. Violaceum. 

Characterization for usage in genetically recoded organisms and expanded genetic code
Team Aboa 2019used this biobrick in production optimization ofcell strain C321.ΔA.exp using expanded genetic code in expression for modified proteins. This biobrick is completely functional and usable also for production of proteins modified with unnatural amino acids.
For the measurements we used Cytation5 in overnight incubation and shaking at 37 degrees celsius. Fluorescence was measured every 30 minutes at preset bandwith using GFP and mCherry programs. Relative fluorescence is calculated as fluorescence at the start of induction with IPTG, pAzF and arabinose divided by the end of incubation the next morning.
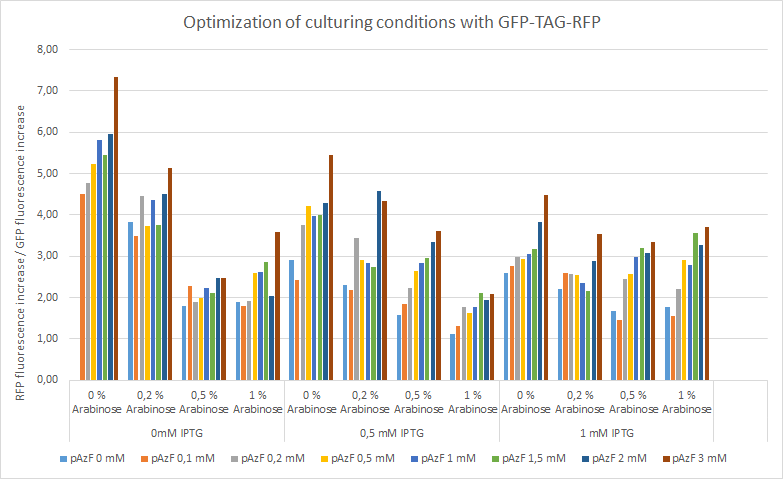
The relative change of RFP fluorescence divided by the change of GFP fluorescence after induction with variable amounts of p-azido-l-phenylalanine, IPTG and arabinose. Relative change of fluorescence is used to normalize signals that are present before induction. The figure shows that IPTG concentrations does not have an effect on the expression of RFP as the promoter is leaky and the arabinose and pAzF are much more critical for producing proteins with unnatural amino acid modifications. High relative expression when little or no pAzF or arabinose is present is most likely a sign of misincorporation of amino acids in avoidance of ribosome stalling.
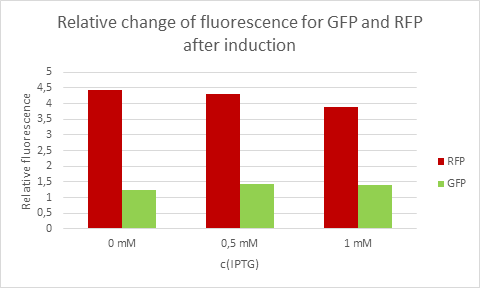
Average eelative change of fluorescence from each concentration of IPTG.
Usage of the lac promoter to express fluorescent markers in two different species
Hong_Kong_HKU tested this promoter (BBa_R0010) in two different species, Escherichia coli and Salmonella Typhi to test whether this is a constitutive promoter in S. Typhi.
E. coli and S. Typhi belong to the same order, Enterobacteriaceae. Lactose fermentation ability is used to differentiate them. S. Typhi does not have the ability to ferment lactose like E. coli does. In this characterisation experiment, this lac promoter is transformed to S. Typhi and E. coli as a control to see the difference in the expression level of the downstream red fluorescent markers.
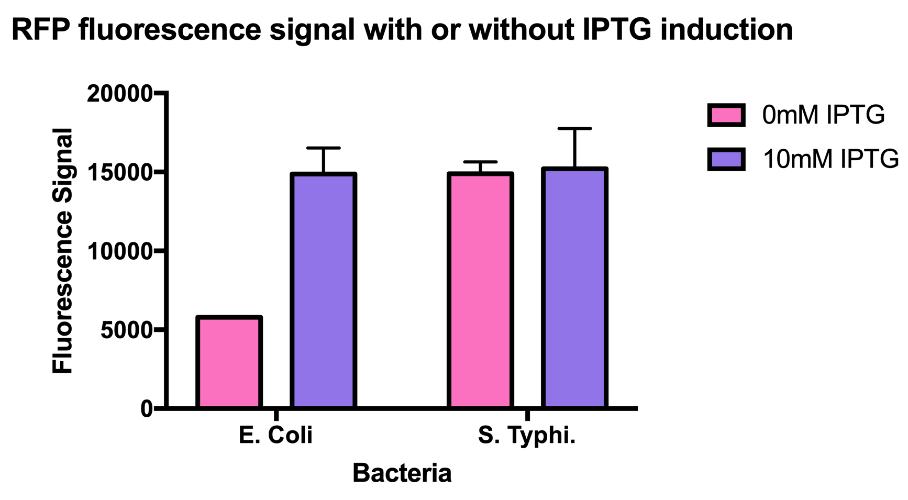
As the data suggests, this promoter is a constitutive promoter in S. Typhi as there is no difference in signals with or without IPTG. As expected, under IPTG induction, there is a 3-fold increase in RFP signals compared to those without IPTG.
Tec-Chihuahua 2019: Usage of lacI promoter to express RFP into E.coli SHuffle T7 Express
Team Tec-Chihuahuaevaluated the functionality of this promoter by testing it in E. coli expression strain SHuffle T7 Express®️. SHuffle was chosen as chassis for having a cytoplasmic environment that favors the production of disulfide bonds. A designed was made for the composite parts under the regulation of part BBa_R0010, a lacI regulated promoter inducible by IPTG. However,characterization of promoters in the iGEM Parts Registry for their use in SHuffle strains is nonexistent.
To evaluate protein production by part BBa_R0010, part BBa_J04450 was transformed, an RFP coding device under the regulation of this promoter, into E. coli SHuffle T7 Express®️. Transformed cells were grown on LB agar with chloramphenicol, for BBa_J04450 was located in the pSB1C3 backbone.
RFP expression was induced at different temperature conditions and IPTG concentration to determine the ideal conditions for the proper functionality of the promoter. Protein expression was analysed through an SDS-PAGE.
The functionality of promoter BBa_R0010 in E. coli SHuffle T7 Express®️ was demonstrated as well as ideal conditions for protein production by characterizing part BBa_J04450, an RFP construct. E. coli SHuffle T7 Express®️ produces higher amounts of protein at 30°C, and concentrations of 0.2 and 0.4 mM IPTG are enough to induce expression when using this lacI regulated promoter.
| Well | 37°C | Well | 30°C |
|---|---|---|---|
| Negative control | Untransformed control | Negative control | Untransformed control |
| Uninduced control (without IPTG) | Uninduced control (without IPTG) | ||
| |
Transformed cells, 0.2 mM IPTG | |
Transformed cells, 0.2 mM IPTG |
| |
Transformed cells, 0.4 mM IPTG | |
Transformed cells, 0.4 mM IPTG |
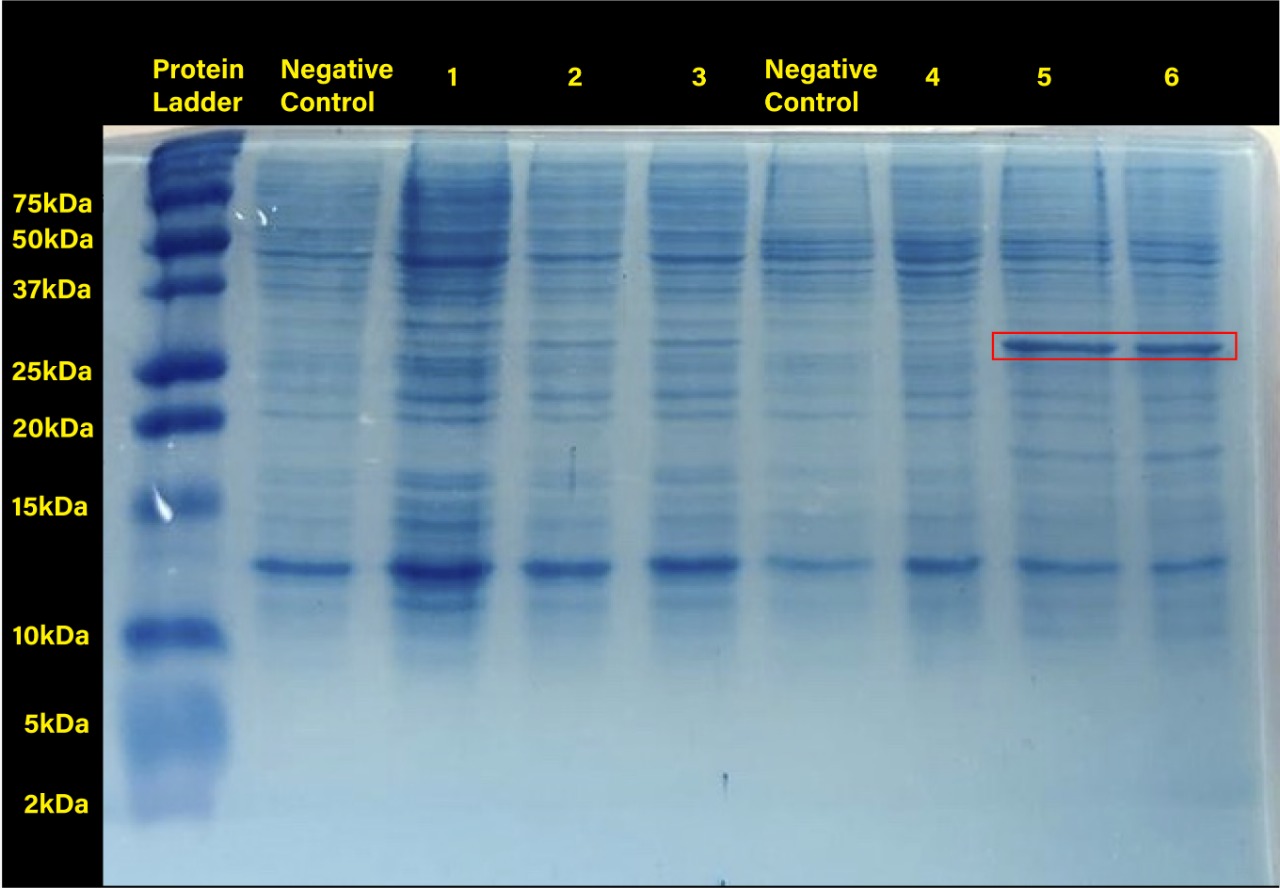
Two prominent bands can be appreciated in the last two columns, corresponding to induction conditions of 0.2 and 0.4 mM IPTG at 30°C, close to the 25 kDa band of the Precision Plus ProteinTM Dual Xtra Prestained Protein Standards. It was concluded that they correspond to RFP, a 26 kDa protein.
When taking a closer look, the band corresponding to RFP can be seen in each column with exception of the untransformed cells controls (negative controls). The uninduced controls (column 1 for 37°C and column 4 for 30°C) without IPTG were expected not to show bands if the promoter lacked leakiness; nevertheless, a faint band can be seen for both controls. Bands corresponding to 0.2 and 0.4 mM IPTG for both temperatures (columns 2 and 3 for 37°C and columns 5 and 6 for 30°C) show stronger bands compared to the uninduced control. Thus, the promoter is slightly leaky, but the addition of proper amounts of IPTG increases protein production. When comparing temperatures, incubation at 30°C clearly yields a higher amount of RFP, but yields similar amounts of expression between both concentrations.
//direction/forward
//chassis/prokaryote/ecoli
//promoter
//regulation/negative
//classic/regulatory/uncategorized
| biology | |
| control | IPTG, lacI |
| direction | Forward |
| negative_regulators | 1 |
| o_h | |
| o_l | |
| positive_regulators |

 1 Registry Star
1 Registry Star