Part:BBa_K3782000
Flavobacterium frigoris strain PS1 ice-binding protein gene
FfIBP
FfIBP is a protein coding region that codes for an ice binding protein (IBP). IBPs, or more specifically antifreeze proteins (AFP), can bind to ice crystals and thereby prevent further ice growth. They are produced by organisms to survive in extremely cold environments. Activities of AFPs can be characterized by their thermal hysteresis (TH) or by their ice recrystallization inhibition (IRI). TH activity corresponds to the lowering of the freezing point without changing the melting point of a solution. IRI activity inhibits the growth of large ice crystals at the expense of smaller ones. The combination of these activities, which vary depending on the protein structure, prevents the freezing of body fluids and cell damage in organisms that live in environments with extremely cold temperatures.
Contents
Profile
| Name | FfIBP |
| Base pairs | 744 |
| Number of amino acids | 247 |
| Molecular weight | 25.35 kDa |
| Origin | Flavobacterium frigoris strain PS1, synthetic |
Usage and Biology
The FfIBP protein coding region was used in the following composite parts (add links). It was expressed in E. coli strain BL21 (DE3) and purified. Various tests and assays were performed to characterize and verify the functionality of this ice-binding protein. FfIBP has moderate TH and IRI activity, and it can bind to ice crystals and inhibit their growth. The aim of our project was to use it on its own or in a mixture of other antifreeze proteins to develop a solution which could be applied on sensitive plant tissues and thereby protect crops from frost damage.
FfIBP is naturally produced by the Gram-negative Antarctic bacterium Flavobacterium frigoris PS1. Its TH activity is around 2.5 K at 50 µM[1].
!!!ADD Biological context and overall mechanism of the part!!!
Characterization
To reduce frost damage during late spring freeze, we wanted to develop a solution containing antifreeze proteins, which bind to ice crystals and inhibit their growth. FfIBP was therefore chosen as one of three AFPs, cloned and expressed in E. coli BL21 (DE3), and finally purified using a His-tag affinity column and gel filtration.
Two different vectors were used for cloning: pET-17b and pColdI, containing a T7 promoter and a cold-shock protein A (cspA) promoter respectively.
Cloning

FfIBP was cloned into the pET-17b plasmid using restriction and ligation via HindIII and XhoI and Gibson Assembly. The vector includes an ampicillin resistance gene and a lac operator, which allows for IPTG induced expression. The final construct was checked with a 1% Agarose gel (Fig.1) and via sequencing.
FfIBP was cloned into the pColdI plasmid using restriction and ligation via NdeI and XhoI and Gibson Assembly. The vector includes an ampicillin resistance gene and a lac operator, which allows for IPTG induced expression. The final construct was checked with a 1% Agarose gel (Fig.1) and via sequencing.
Expression and Purification
After we successfully transformed E. coli strain BL21 (DE3) with our pET-17b-FfIBP and pColdI-FfIBP constructs, we inoculated liquid cultures with LB and ampicillin. Before inducing expression in large volumes of liquid cultures, we first did small-scale test purifications to verify if the antifreeze proteins could be correctly purified. Next, we grew 1-liter liquid cultures at 37°C until an OD600 of 0.6 was reached. We then placed the culture in a cooling incubator at 15°C for 30 minutes. We added IPTG 1mM and induced expression at 15°C for 24 hours. Afterwards, we removed a small volume of culture to run a total cell SDS-PAGE (15%) of E. coli cultures stained with Coomassie blue and to verify that the antifreeze protein was correctly overexpressed. The remaining culture was centrifuged, and the cell pellet was resuspended in Cold Lysis Buffer. We sonicated the cells and centrifuged the cells once more to collect the supernatant, which we loaded into the His-tag affinity column. We eluted the antifreeze proteins which bound to the column and concentrated the solution to a smaller volume. We performed a gel filtration to further purify the protein and finally stored aliquots at -80°C.
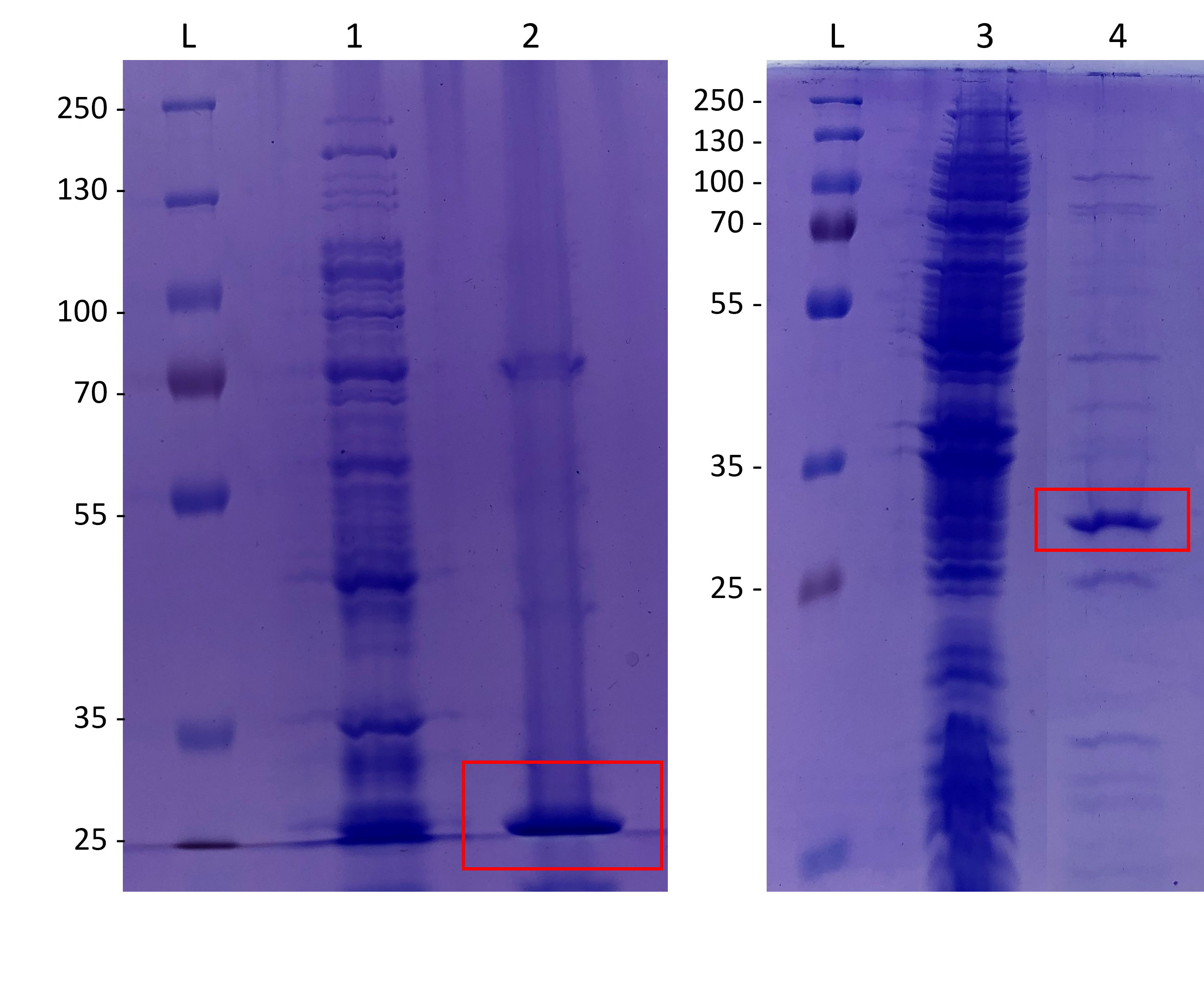
We induced expression of FfIBP with IPTG and ran a total cell SDS-PAGE (15%) of the E. coli BL21 (DE3) cultures stained with Coomassie blue. We used two different vectors, pET-17b and pColdI, to compare how the promoters influenced expression efficiency. The overexpression was successful for FfIBP. We observed thicker bands that matched the weight of the protein. The pColdI vector worked better than the pET-17b vector, because the band appeared to be stronger for this first plasmid on the gel (Fig.2).
ISF Assay
FDT Assay
Sequence and Features
- 10INCOMPATIBLE WITH RFC[10]Illegal PstI site found at 269
Illegal PstI site found at 350
Illegal PstI site found at 530 - 12INCOMPATIBLE WITH RFC[12]Illegal PstI site found at 269
Illegal PstI site found at 350
Illegal PstI site found at 530 - 21COMPATIBLE WITH RFC[21]
- 23INCOMPATIBLE WITH RFC[23]Illegal PstI site found at 269
Illegal PstI site found at 350
Illegal PstI site found at 530 - 25INCOMPATIBLE WITH RFC[25]Illegal PstI site found at 269
Illegal PstI site found at 350
Illegal PstI site found at 530 - 1000COMPATIBLE WITH RFC[1000]
References
Template:Reflist- ↑ Do H, Lee JH, Lee SG, Kim HJ. Crystallization and preliminary X-ray crystallographic analysis of an ice-binding protein (FfIBP) from Flavobacterium frigoris PS1. Acta Crystallogr Sect F Struct Biol Cryst Commun [Internet]. 2012 Jul [cited 2021 Jul 7];68(Pt 7):806. Available from: /pmc/articles/PMC3388927/
//cds
//chassis/prokaryote/ecoli
//collections/antifreeze
| biology | Flavobacterium frigoris strain PS1 |
| protein | FfIBP |
