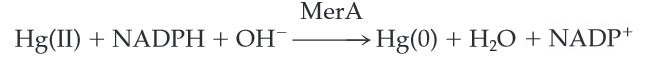Part:BBa_K1420001
merA, mercuric reductase from Serratia marcescens
Overview
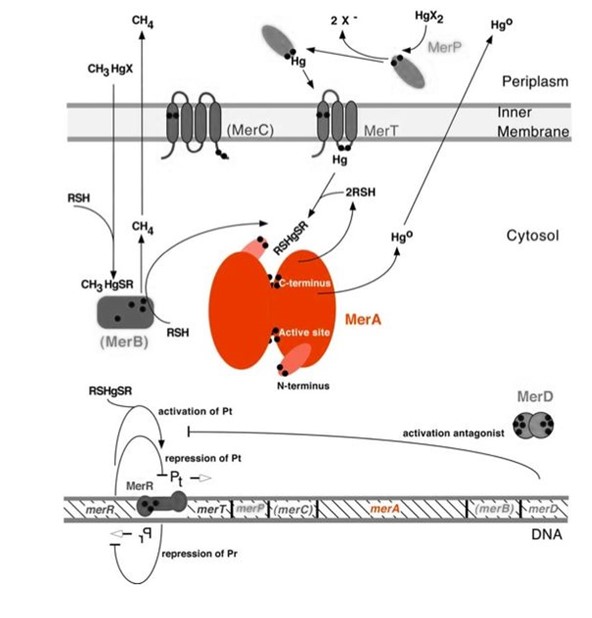
Mercury resistance gene merA (1.6Kb) encodes MerA, a mercuric ion reductase (cytosolic, flavin disulfide oxidoreductase, ~120kDa), that is essential to bacterial mercury resistance. MerA and gene merA are highlighted in orange in Figure 1. The lower part of the Figure 1 shows the arrangement of mer genes in the operon, and merA is located downstream of merT and merP, the genes that encode mercury transport proteins. As shown in Scheme 1, the essential mechanism of mercury resistance is carried out by MerB and MerA. MerB transforms methylmercury to less toxic, non-biomagnified ionic mercury Hg(II), and MerA reduces Hg(II) to the least toxic metallic mercury, Hg(0).
Figure 1. Model of mercury resistance operon. The symbol • indicates a cysteine residue. RSH indicates cytosolic thiol redox buffers such as glutathione. Figure 1 shows the interactions of MerA with mercury compounds and other gene products of mer operon. (This figure is adapted from "Bacterial mercury resistance from atoms to ecosystems". Reference: T. Barkay et al. FEMS Microbiology Reviews 27 (2003) 355-384.)
The nature evolves different mercury detoxification strategies. Here we describe MerA and MerB confer mercury resistance by coupling electrochemical reductions of thiol-avid organic mercury species to relatively inert, uncharged, monoatonic mercury. Since organic mercuric species have high affinity to tiol groups and commomly form strong but reversible bonds,cytosolic thiol redox buffers and cysteine residues of mercury binding proteins compete with thiol groups in other protein and therefore prevents cationic mercury from inhibiting cytosolic machineries. Once the organomercury is converted to ionic mercury by MerB, MerA reduces ionic mercury to volatile, elemental mercury which diffuses through cell membrane without any active transport systems. While MerA and MerB are the key mercury detoxification enzymes, they orchestrate with mercury transport proteins, MerT and MerP, to confer bacterial mercury resistance.
Molecular Function
MerA catalyzes the reduction of mercuric ion to the relative inert, volatile monoatomic mercury in a NADPH dependent reaction. Ionic mercury Hg(II) is released from protonlysis of organomercury catalyzed by MerB.
Scheme 1. MerA Catalyzed Reaction
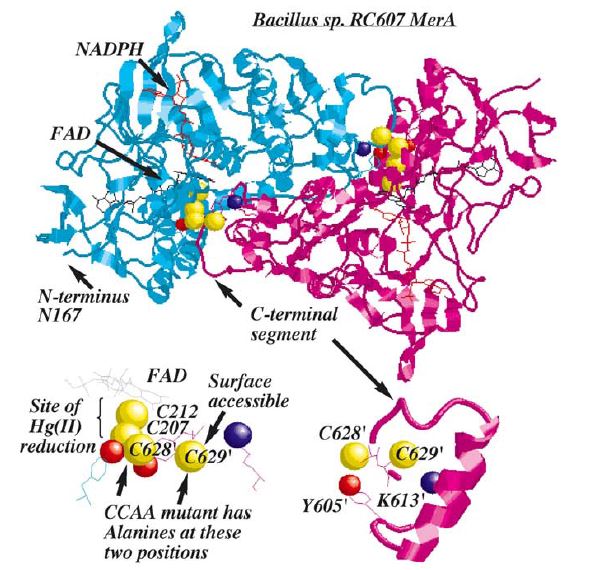
Structure and Mechanism
MerA is a 125kDA homodimer.
Characterization of merA
To test the effect of MerA to mercury resistance, we generated a merA deletion construct from pBBRBB::mer and characterized it by zone of inhibition test.
Zone of Inhibition Results
Figure 2. Zones of Inhibition Test For Mercury Resistance Activity. In this assay, three different constructs were expressed in Escherichia coli strain K12 to compare level of mercury resistance. Each plate had 10µL of 0.1M HgCl2 spotted on a filter disk in the middle of an agar plate. (A)Plates of ZOI test. Left, K12 conntaining pBBRBB::mer; center,K12 containing pBBRBB::gfp; right, K12 strain containing pBBRBB::merΔmerA, or the merA deletion mutant. (b) Graphical representation of the size of zones of inhibition diameter. The diameter of the Zone of Inhibition was measured in triplicate. Green corresponds to pBBRBB::gfp, blue to pBBRBB::mer, and red to pBBRBB::merΔmerA.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 1201
Illegal NgoMIV site found at 1249
Illegal NgoMIV site found at 1311
Illegal NgoMIV site found at 1522 - 1000COMPATIBLE WITH RFC[1000]
//cds
| None |