Difference between revisions of "Part:BBa K1033933"
| Line 27: | Line 27: | ||
</ul> | </ul> | ||
<br><br> | <br><br> | ||
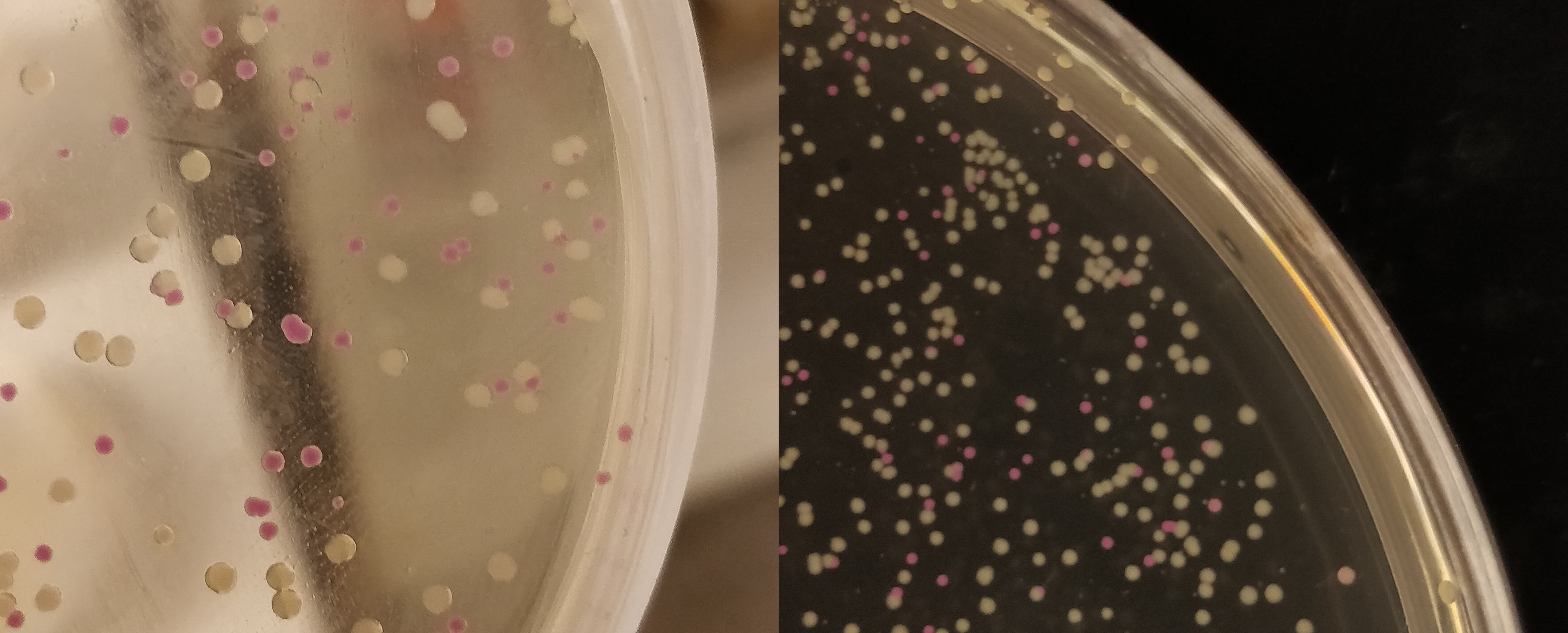
| − | + | To visualize the absorbance of AsPink on a petri dish, BL21 (DE3) colonies containing the pUC19-pCons-AsPink part (<a href="https://parts.igem.org/Part:BBa_K3182100">BBa_K3182100</a>), which was used in the pink-white screening, were incubated for 16 hours in 37 °C. The pCons-AsPink construct was removed and (Magainin 2) and <a href="https://parts.igem.org/Part:BBa_K3182104 ">BBa_K3182104</a> (CHAP) was inserted into the vector. The pink colonies contain AsPink and indicate a religated pCons-AsPink. The white colonies indicate a successful ligation. Below (Figure 7) is a picture of the pink-white screening. | |
</html> | </html> | ||
Revision as of 16:10, 20 October 2019
asPink, pink chromoprotein
This chromoprotein from the coral Anemonia sulcata, asPink (also known as asCP or asFP595), naturally exhibits strong color when expressed. The protein has an absorption maximum at 572 nm giving it a pink/purple color visible to the naked eye. The strong color is readily observed in both LB or on agar plates after less than 24 hours of incubation. The protein asPink has significant sequence homologies with proteins in the GFP family.
Important: This part is not available in the registry yet, however, the same part is available from the registry with the standard RBS B0034: BBa_K1033927.
Usage and Biology
This part is useful as a reporter.
Contribution
Group: Linkoping_Sweden iGEM 2019
Author: Andreas Holmqvist, Leo Juhlin and Emmanuel Berlin
Summary: We studied the expression in of E.coli BL21(DE3) containing CBD-asPink (BBa_K3182100) protein with a constitutive promotor to see if the protein could be used for a pink/white screening. We also tested the bindning capacity of the CBD-asPink protein to a micro cellulose bandage and then studied the release mechanism.
Documentation:
The expression system used contained the following parts:
- the BBa_B0034 Ribosome binding site
- the BBa_J23110 Constitutive promotor
To visualize the absorbance of AsPink on a petri dish, BL21 (DE3) colonies containing the pUC19-pCons-AsPink part (BBa_K3182100), which was used in the pink-white screening, were incubated for 16 hours in 37 °C. The pCons-AsPink construct was removed and (Magainin 2) and BBa_K3182104 (CHAP) was inserted into the vector. The pink colonies contain AsPink and indicate a religated pCons-AsPink. The white colonies indicate a successful ligation. Below (Figure 7) is a picture of the pink-white screening.

CBD-AsPink (K3182000) were expressed in BL21 (DE3) Gold 16 hours at 16°C at 80 rpm in 1L of LB-miller in Figure 1 above. Colonies containing a CBD-pCons-asPink part (
CBD-asPink bindning capacity
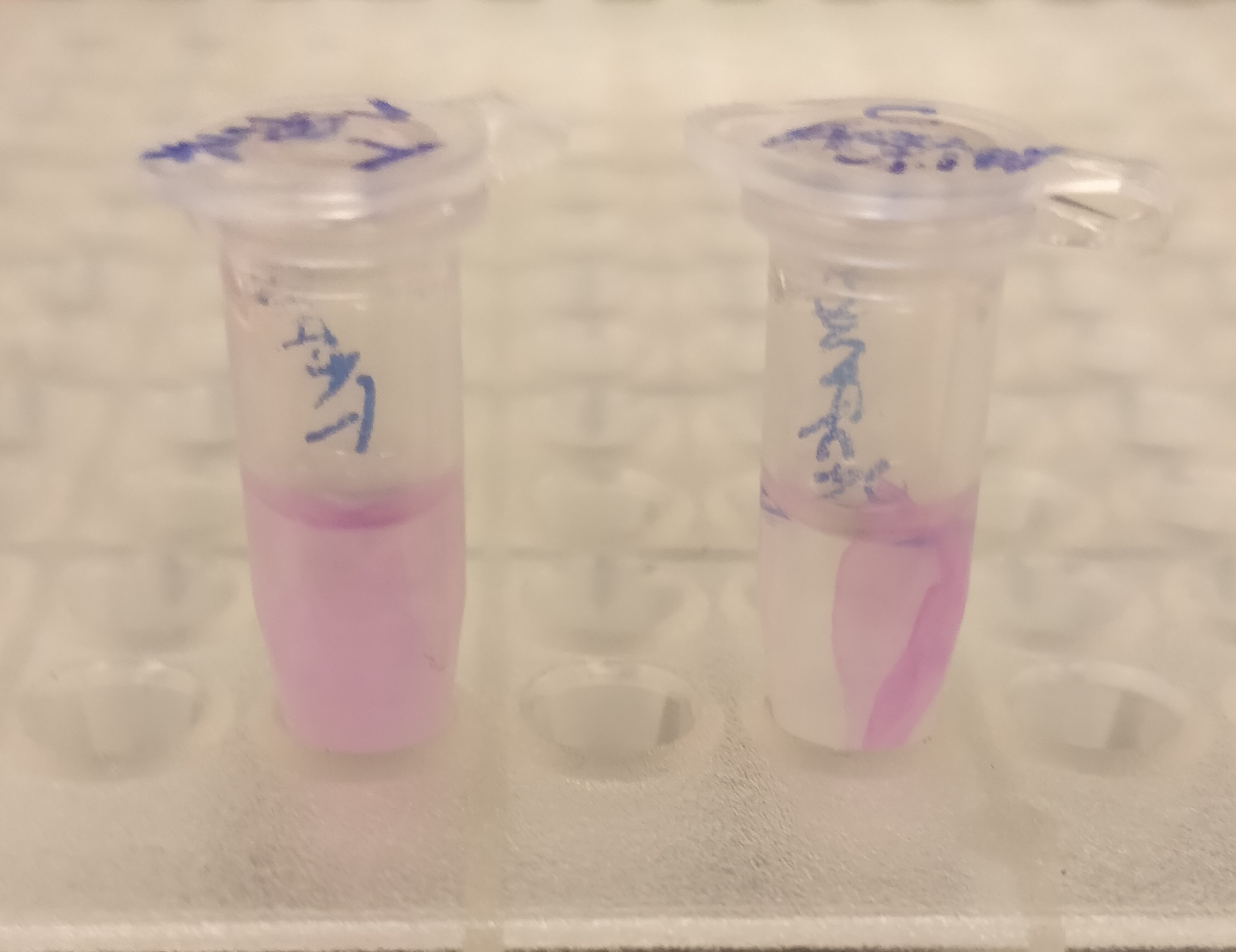
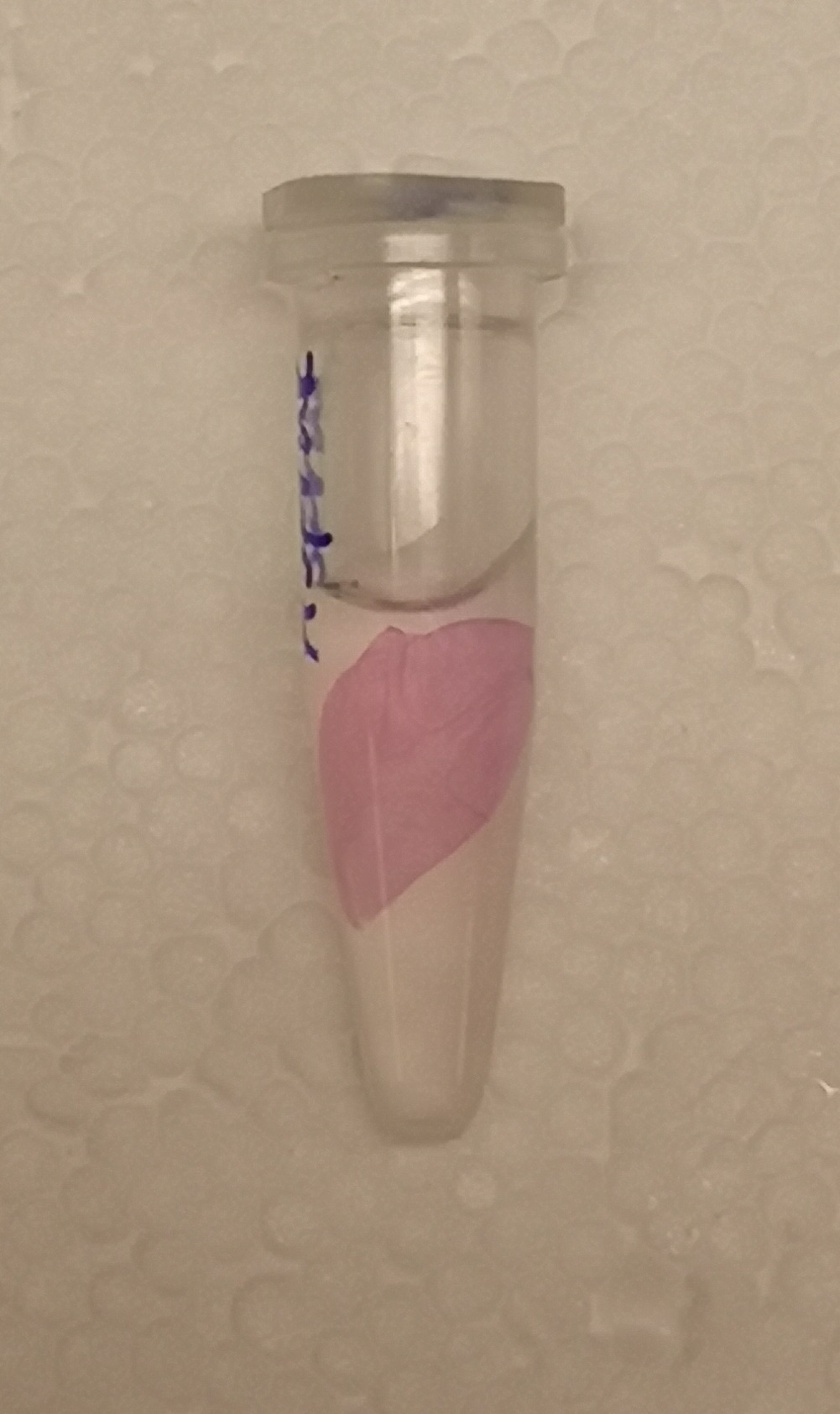
After the washes, human thrombin and cleavage buffer was added to the bandage with bound CBD-asPink to test the release mechanism of the fusion protein. The bandage was then incubated with the solution for 16 hours on an end to end rotator together with an negative control containing cellulose bandage and only cleavage buffer. After the incubation, the supernatant containing thrombin was pink while the negative control containing only cleavage buffer was transparent with a clear pink cellulose bandage (Figure 4). This indicates that the release mechanism work and the AsPink protein has successfully been released from the bandage into the supernatant.
asPink does not exist as CDS only but only as RBS-asPink (BBa_K1033927) and J23110-RBS-spisPink (BBa_K1033926).
iGEM2013 Uppsala: Expression of asPink in E. coli DH5alpha by promoter J23110 from medium copy plasmid pSB3K3 (left) or high copy plasmid pSB1K3 (right).
Source
The protein was first extracted and characterized by Lukyanov et. al. 2000 under the name asFP595 (GenBank: AAG02385.1). This version is codon optimized for E coli by Genscript.
References
[http://www.jbc.org/content/275/34/25879.short]Lukyanov, Konstantin A., et al. "Natural animal coloration can be determined by a nonfluorescent green fluorescent protein homolog." Journal of Biological Chemistry 275.34 (2000): 25879-25882.
[http://www.jbc.org/content/280/4/2401.short]Wilmann, Pascal G., et al. "Variations on the GFP Chromophore - A Polypeptide Fragmentation Within The Chromophore Revealed In The 2.1-Å Crystal Structure Of A Nonfluorescent Chromoprotein From Anemonia Sulcata." Journal of Biological Chemistry 280.4 (2005): 2401-2404.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]