Difference between revisions of "Part:BBa K2273038"
| (One intermediate revision by the same user not shown) | |||
| Line 47: | Line 47: | ||
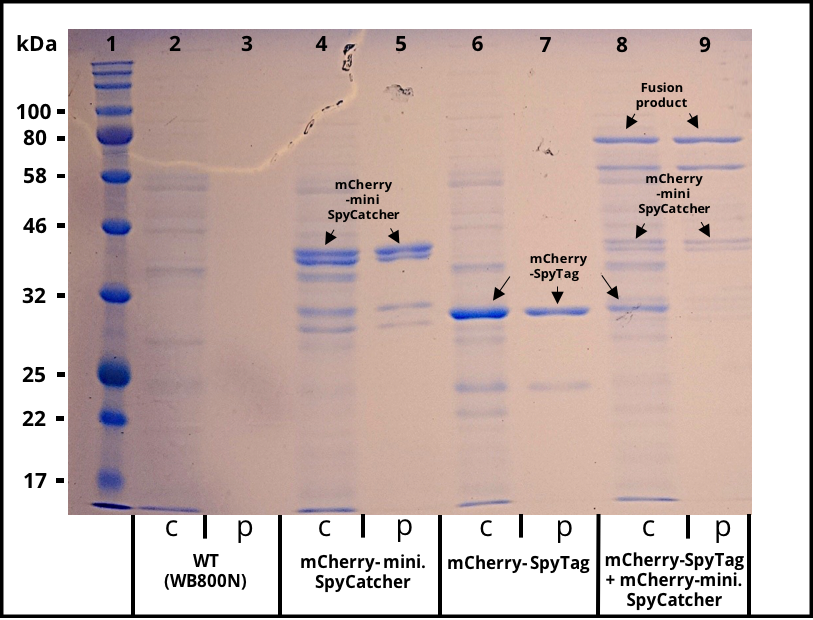
[[File:T--TU_Dresden--secretion---result--sds.png|thumb|left|350px|'''Figure 3:''' SDS gel with crude and purified supernatants. Expression of the multi copy mCherry constructs was induced with 1% Xylose and the supernatants were harvested after 16 h of incubation. The his-tagged proteins were purified with Ni-NTA agarose beads. Lane 1 was loaded with 3 µl of NEB´s “Color Prestained Protein Standard Broad Range” ladder. Crude (c) and purified (p) supernatant of wild-type (WT) are shown as a control in lane 2 and 3. Lane 4 and 5 contain the supernatant of B. subtilis producing mCherry-mini. SpyCatcher fusion protein (36,6 kDa). Lane 4 and 5 contain the supernatant of B. subtilis producing mCherry-SpyTag fusion protein (31,9 kDa). The crude supernatants of the two mCherry producing strains were combined, incubated for 4 h, purified and loaded onto lane 8 and 9. The fusion product of the mCherry constructs is visable in the crude and purified supernatant.]] | [[File:T--TU_Dresden--secretion---result--sds.png|thumb|left|350px|'''Figure 3:''' SDS gel with crude and purified supernatants. Expression of the multi copy mCherry constructs was induced with 1% Xylose and the supernatants were harvested after 16 h of incubation. The his-tagged proteins were purified with Ni-NTA agarose beads. Lane 1 was loaded with 3 µl of NEB´s “Color Prestained Protein Standard Broad Range” ladder. Crude (c) and purified (p) supernatant of wild-type (WT) are shown as a control in lane 2 and 3. Lane 4 and 5 contain the supernatant of B. subtilis producing mCherry-mini. SpyCatcher fusion protein (36,6 kDa). Lane 4 and 5 contain the supernatant of B. subtilis producing mCherry-SpyTag fusion protein (31,9 kDa). The crude supernatants of the two mCherry producing strains were combined, incubated for 4 h, purified and loaded onto lane 8 and 9. The fusion product of the mCherry constructs is visable in the crude and purified supernatant.]] | ||
| + | |||
| + | |||
| + | |||
| + | <partinfo>BBa_K2273038 SequenceAndFeatures</partinfo> | ||
Latest revision as of 19:06, 1 November 2017
| Part Information | |
|---|---|
| RFC standard | RFC 25 |
| Fused Tag | BBa_K2273015: SpyCatcher |
| Fluorescent Protein | BBa_K2273022: mCherry His-tagged |
| Submitted by | [http://2017.igem.org/Team:TU_Dresden TU Dresden] |
mCherry with N-Terminal SpyCatcher and C-Terminal His Tag
This composite part was used for evalutaion in the [http://2017.igem.org/Team:TU_Dresden/Project/Secretion secretion project] of 2017 TU_Dresden iGEM [http://2017.igem.org/Team:TU_Dresden team]. It codes for a fluorescent reporter protein (mCherry) and a functional tag (SpyCatcher), mediating covalent bonding with its tag partner (SpyTag). It is optimized for usage in Bacillus subtilis.
Design
A signal peptide (AmyE SP) was fused n-terminally to this construct to induce secretion.
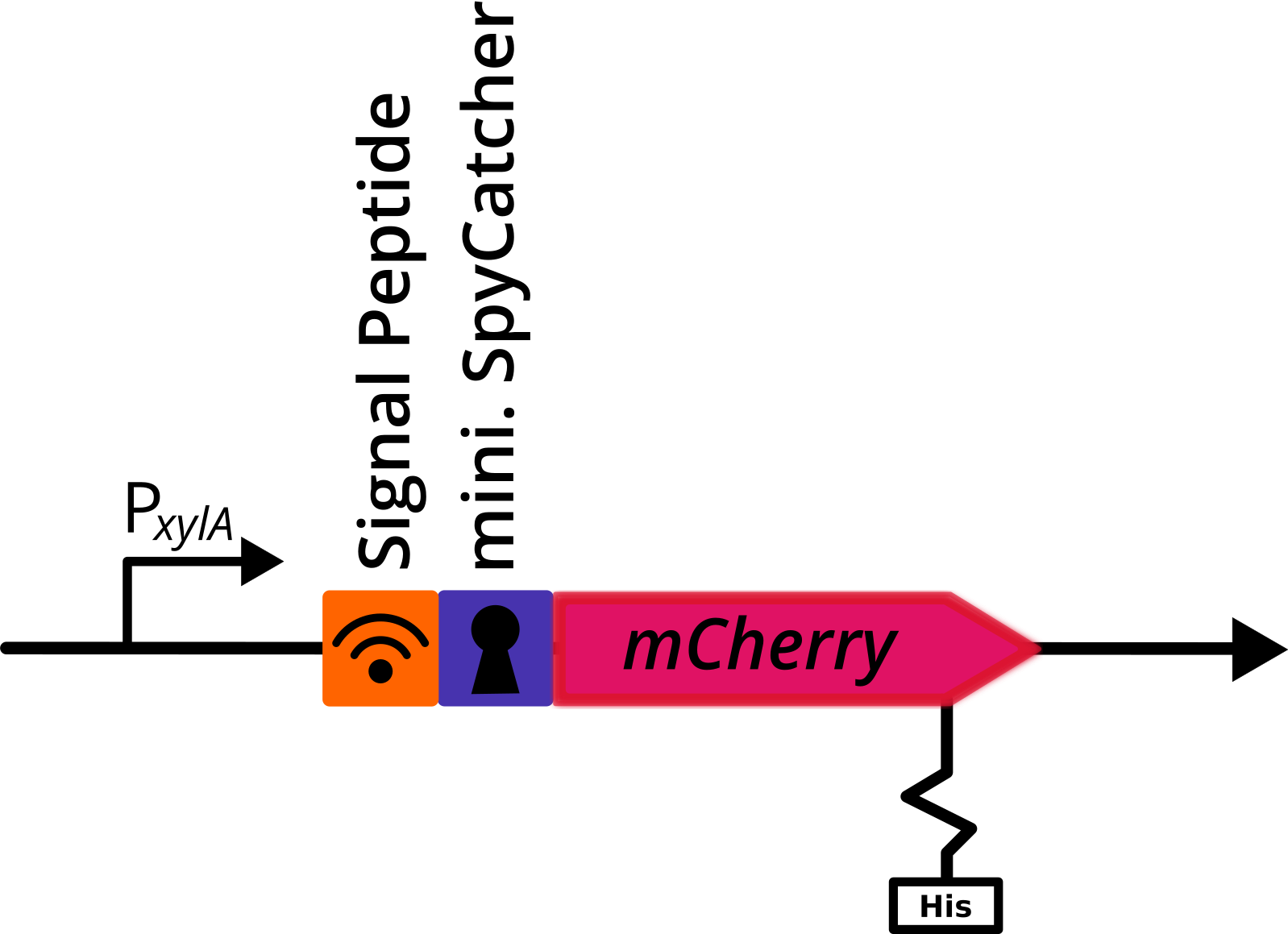
Application
The successful secretion could be proven with a fluorescence assay using the supernatants of B. subtilis (Figure 2). The functionality of the SpyTag/SpyCatcher was proven via SDS-PAGE, using the supernatants (Figure 3).
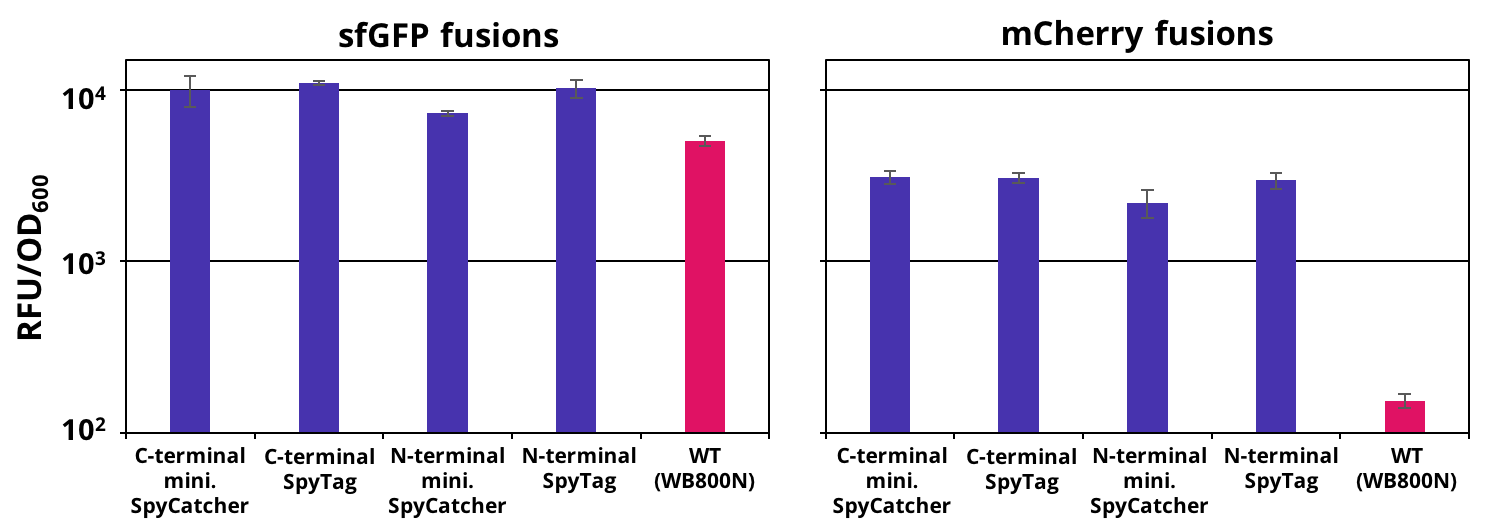
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
