Difference between revisions of "Part:BBa K3686010"
LeiYingJie (Talk | contribs) |
(→Results) |
||
| (11 intermediate revisions by 2 users not shown) | |||
| Line 3: | Line 3: | ||
<partinfo>BBa_K3686010 short</partinfo> | <partinfo>BBa_K3686010 short</partinfo> | ||
| − | Bt toxins refers to the toxic proteins produced by insect pathogenic bacteria Bacillus thuringiensis. Each Bt toxin display unique conformations of peptide chain thus have a specificity to certain invertebrates. Where Cry7Ca1 is a differentiate of Bt toxins with a molecular mass of 129kDa, recently isolated from Bt strain BHT-13. According to the crystalline analysis, it could be inferred that this toxin displays a similar mode of action, the pre-pore forming model, like its relatives [1][2][3]. Which the lethality of this toxin against locusts have been confirmed at an LC50 (50% lethal concentration) value of 8.98 μg/ml [1].Once ingested by insects, Cry7Ca1 undergoes a series of digestion by protease in insect gut juice. Which cleaves the protective shell and release the core toxin inside. Furthermore, the core toxin binds to the membrane-bonded protease on epithelial cells. The proteolytic degradation removes the N-terminal alpha chain, unreal the core into smaller oligomers that embed onto the cell membrane and result in cell disruption. [4]<br> | + | Bt toxins refers to the toxic proteins produced by insect pathogenic bacteria Bacillus thuringiensis. Each Bt toxin display unique conformations of peptide chain thus have a specificity to certain invertebrates. Where Cry7Ca1 is a differentiate of Bt toxins with a molecular mass of 129kDa, recently isolated from Bt strain BHT-13. According to the crystalline analysis, it could be inferred that this toxin displays a similar mode of action, the pre-pore forming model, like its relatives [1][2][3]. Which the lethality of this toxin against locusts have been confirmed at an LC50 (50% lethal concentration) value of 8.98 μg/ml [1].<br> |
| + | [[File:Cry7ca_structure.png|600px|thumb|center|Jing, X., Yuan, Y., Wu, Y., Wu, D., Gong, P., & Gao, M. (2019). Crystal structure of Bacillus thuringiensis Cry7Ca1 toxin active against Locusta migratoria manilensis. Protein Science, 28(3), 609-619.]] | ||
| + | Once ingested by insects, Cry7Ca1 undergoes a series of digestion by protease in insect gut juice. Which cleaves the protective shell and release the core toxin inside. Furthermore, the core toxin binds to the membrane-bonded protease on epithelial cells. The proteolytic degradation removes the N-terminal alpha chain, unreal the core into smaller oligomers that embed onto the cell membrane and result in cell disruption. [4]<br> | ||
[1]Wu, Y., Lei, C. F., Yi, D., Liu, P. M., & Gao, M. Y. (2011). Novel Bacillus thuringiensis δ-endotoxin active against Locusta migratoria manilensis. Applied and environmental microbiology, 77(10), 3227-3233.<br> | [1]Wu, Y., Lei, C. F., Yi, D., Liu, P. M., & Gao, M. Y. (2011). Novel Bacillus thuringiensis δ-endotoxin active against Locusta migratoria manilensis. Applied and environmental microbiology, 77(10), 3227-3233.<br> | ||
[2]Jimenez-Juarez, N., Munoz-Garay, C., Gómez, I., Gill, S. S., Soberón, M., & Bravo, A. (2008). The pre-pore from Bacillus thuringiensis Cry1Ab toxin is necessary to induce insect death in Manduca sexta. Peptides, 29(2), <318-323..<br> | [2]Jimenez-Juarez, N., Munoz-Garay, C., Gómez, I., Gill, S. S., Soberón, M., & Bravo, A. (2008). The pre-pore from Bacillus thuringiensis Cry1Ab toxin is necessary to induce insect death in Manduca sexta. Peptides, 29(2), <318-323..<br> | ||
[3]Vachon, V., Laprade, R., & Schwartz, J. L. (2012). Current models of the mode of action of Bacillus thuringiensis insecticidal crystal proteins: a critical review. Journal of invertebrate pathology, 111(1), 1-12.<br> | [3]Vachon, V., Laprade, R., & Schwartz, J. L. (2012). Current models of the mode of action of Bacillus thuringiensis insecticidal crystal proteins: a critical review. Journal of invertebrate pathology, 111(1), 1-12.<br> | ||
[4]Melo, A. L. D. A., Soccol, V. T., & Soccol, C. R. (2016). Bacillus thuringiensis: mechanism of action, resistance, and new applications: a review. Critical reviews in biotechnology, 36(2), 317-326<br> | [4]Melo, A. L. D. A., Soccol, V. T., & Soccol, C. R. (2016). Bacillus thuringiensis: mechanism of action, resistance, and new applications: a review. Critical reviews in biotechnology, 36(2), 317-326<br> | ||
| + | ===Source of sequence=== | ||
| + | Genome of Bacillus thuringiensis (BTH-3), full amino acid sequence submitted under GenBank accession no. EF486523. | ||
| + | ===Considerations:=== | ||
| + | Codon-optimized for expression in Escherichia coli. | ||
| + | ===Optimization details:=== | ||
| + | [[File:Cry7ca_GC.jpeg|600px|thumb|center|]] | ||
| + | ===Expression in pTac system:=== | ||
| + | Gene fore Cry7Ca1 has been his-tagged and sealed onto vector pET28a and transformed into BL21 and DH10B(E. coli) for pTac regulated expression.<br> | ||
| + | When induced by 1mM IPTG under 37℃ for 6 hours, the cell were harvested and disrupted by ultrasonic homogenizer. Which the Cry7Ca1 in homogenate then purified using Ni-NTA system where samples were isolated from raw lysate and elusion buffer flowthrough.<br> | ||
| + | We have successfully identified the desired band after SDS-PAGE which indicates the presence of Cry7Ca1:<br> | ||
| + | [[File:Cry7ca_express_in_BL21.jpeg|600px|thumb|center|]] | ||
| + | [[File:Cry7ca_express_in_DH10B.jpeg|600px|thumb|center|]] | ||
| + | |||
| + | |||
| + | =SZ-SHD 2021's Improvement= | ||
| + | |||
| + | The part <partinfo>K3895012</partinfo> was improved from SZ-SHD 2020's bt toxin Cry7Ca1 (<partinfo>K3686010</partinfo>) for tobacco wheat instant expression, where Cry7Ca1 is a differentiate of Bt toxins with a molecular mass of 129kDa, recently isolated from Bt strain BHT-13. Cry7Ca1 is connected with GFP (<partinfo>E0040</partinfo>),and constructed into pCAMBIA1301 vector. | ||
| + | |||
| + | [[File:T--SZ SHD--cryjpg.png|center|350px|thumb|'''Figure 1.''' Plasmid construction of pCAMBIA-Cry7Ca1-eGFP.]] | ||
| + | |||
| + | ===Protocol=== | ||
| + | 1. Transfection of pCAMBIA-Cry7Ca1-eGFP vector through Carbon dots nanocomposite (CDP) to Nicotiana benthamiana | ||
| + | |||
| + | Coupling CDP with plant expressing vector | ||
| + | Mix the following ingredients: | ||
| + | |||
| + | {|border=1 width="90%" align="center" | ||
| + | |- | ||
| + | !width="20%" style="background:#CCCCFF"|Ingredients | ||
| + | !width="20%"|Volumn | ||
| + | |||
| + | |-align="center" | ||
| + | |style="background:#EEEEFF"|MES buffer(50X) | ||
| + | |10ul | ||
| + | |-align="center" | ||
| + | |style="background:#EEEEFF"|CDP(50X) | ||
| + | |10ul | ||
| + | |-align="center" | ||
| + | |style="background:#EEEEFF"|DNA(237ng/ul >10ng/ul final con) | ||
| + | |22ul | ||
| + | |-align="center" | ||
| + | |style="background:#EEEEFF"|10% glycerol | ||
| + | |25ul | ||
| + | |-align="center" | ||
| + | |style="background:#EEEEFF"|ddH2O | ||
| + | |433ul | ||
| + | |-align="center" | ||
| + | |style="background:#EEEEFF"|total | ||
| + | |500ul | ||
| + | |} | ||
| + | Gently mix and incubate at 37℃ for 30min. | ||
| + | |||
| + | 2. Brush the mixture gently on the leaf of Nicotiana benthamiana(leaf length>10cm,Growing well) ,mark the area of brushing | ||
| + | |||
| + | 3. Put the plant back in the light incubator(28℃,12h light 12h dark), repeat the process for four days at 3:00pm each day(2/4) | ||
| + | |||
| + | |||
| + | ===Results=== | ||
| + | Fluorescence microscope to observe the GFP in leave | ||
| + | {|border=0 width="90%" align="center" | ||
| + | |-align="center" | ||
| + | |[[File:T--SZ SHD--cry1.jpg|400px|thumb|center|'''Figure 2(a). Positive results showed successful expression of pCAMBIA-Cry7Ca1-eGFP in Nicotiana''' ]] | ||
| + | |[[File:T--SZ SHD--cry2.jpg|400px|thumb|center|'''Figure 2(b). Negative results without Cry7Ca1-eGFP in Nicotiana benthamiana.''' ]] | ||
| + | |} | ||
| + | |||
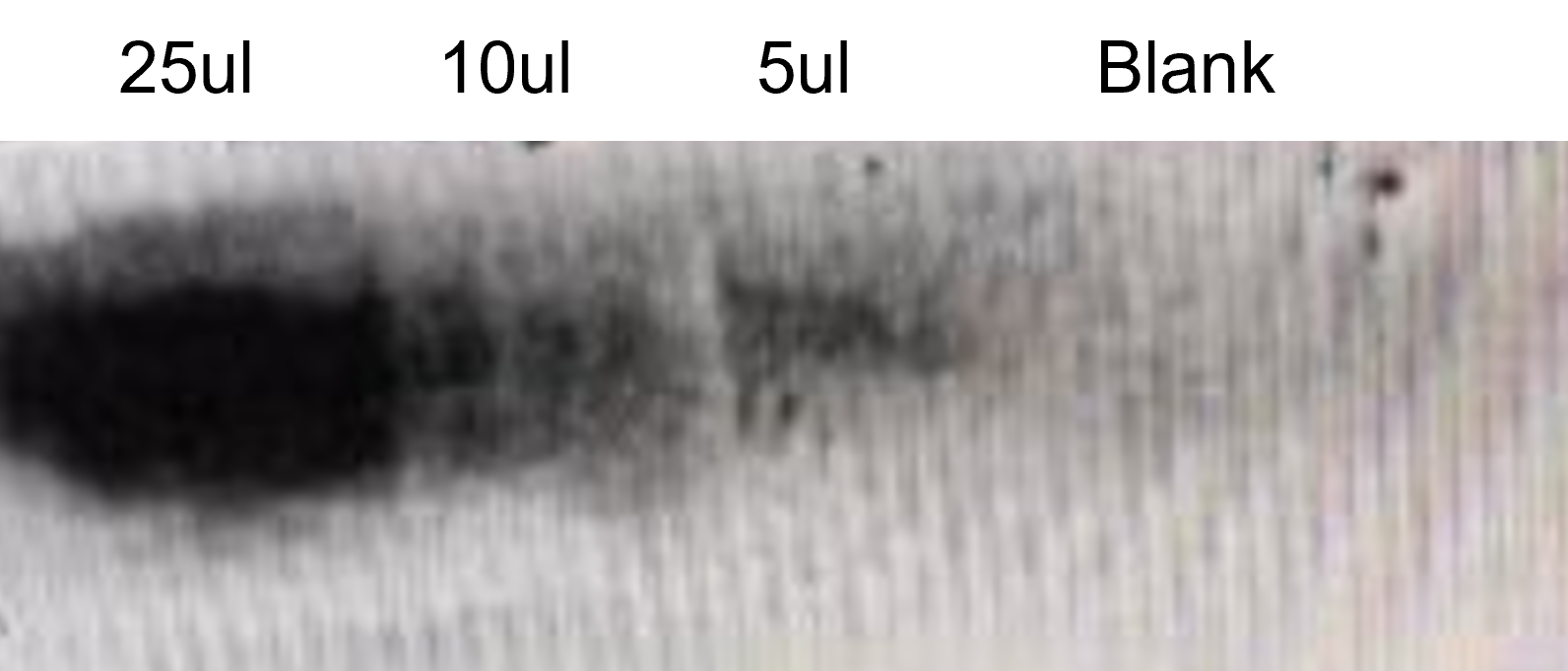
| + | [[File:T--SZ SHD--lyj2.jpg|300px|center]] | ||
| + | '''Figure 3.''' Western Blot result of Cry7Ca1-eGFP protein express in wheat leaf. | ||
| + | [[File:T--SZ_SHD--lyj1.jpg|300px|center]] | ||
| + | '''Figure 4.''' Fluorescence microscopy images of the CDP transfected on plant leaf. 1/3. Blank control group with CDP only; 2/4. CDP with 10ng/ul plant expressing vector. | ||
| + | |||
| + | |||
Latest revision as of 17:16, 21 October 2021
Insecticidal protein from Bacillus thuringiensis (BTH-3), complete CDs, codon-optimized
Bt toxins refers to the toxic proteins produced by insect pathogenic bacteria Bacillus thuringiensis. Each Bt toxin display unique conformations of peptide chain thus have a specificity to certain invertebrates. Where Cry7Ca1 is a differentiate of Bt toxins with a molecular mass of 129kDa, recently isolated from Bt strain BHT-13. According to the crystalline analysis, it could be inferred that this toxin displays a similar mode of action, the pre-pore forming model, like its relatives [1][2][3]. Which the lethality of this toxin against locusts have been confirmed at an LC50 (50% lethal concentration) value of 8.98 μg/ml [1].
Once ingested by insects, Cry7Ca1 undergoes a series of digestion by protease in insect gut juice. Which cleaves the protective shell and release the core toxin inside. Furthermore, the core toxin binds to the membrane-bonded protease on epithelial cells. The proteolytic degradation removes the N-terminal alpha chain, unreal the core into smaller oligomers that embed onto the cell membrane and result in cell disruption. [4]
[1]Wu, Y., Lei, C. F., Yi, D., Liu, P. M., & Gao, M. Y. (2011). Novel Bacillus thuringiensis δ-endotoxin active against Locusta migratoria manilensis. Applied and environmental microbiology, 77(10), 3227-3233.
[2]Jimenez-Juarez, N., Munoz-Garay, C., Gómez, I., Gill, S. S., Soberón, M., & Bravo, A. (2008). The pre-pore from Bacillus thuringiensis Cry1Ab toxin is necessary to induce insect death in Manduca sexta. Peptides, 29(2), <318-323..
[3]Vachon, V., Laprade, R., & Schwartz, J. L. (2012). Current models of the mode of action of Bacillus thuringiensis insecticidal crystal proteins: a critical review. Journal of invertebrate pathology, 111(1), 1-12.
[4]Melo, A. L. D. A., Soccol, V. T., & Soccol, C. R. (2016). Bacillus thuringiensis: mechanism of action, resistance, and new applications: a review. Critical reviews in biotechnology, 36(2), 317-326
Source of sequence
Genome of Bacillus thuringiensis (BTH-3), full amino acid sequence submitted under GenBank accession no. EF486523.
Considerations:
Codon-optimized for expression in Escherichia coli.
Optimization details:
Expression in pTac system:
Gene fore Cry7Ca1 has been his-tagged and sealed onto vector pET28a and transformed into BL21 and DH10B(E. coli) for pTac regulated expression.
When induced by 1mM IPTG under 37℃ for 6 hours, the cell were harvested and disrupted by ultrasonic homogenizer. Which the Cry7Ca1 in homogenate then purified using Ni-NTA system where samples were isolated from raw lysate and elusion buffer flowthrough.
We have successfully identified the desired band after SDS-PAGE which indicates the presence of Cry7Ca1:
SZ-SHD 2021's Improvement
The part BBa_K3895012 was improved from SZ-SHD 2020's bt toxin Cry7Ca1 (BBa_K3686010) for tobacco wheat instant expression, where Cry7Ca1 is a differentiate of Bt toxins with a molecular mass of 129kDa, recently isolated from Bt strain BHT-13. Cry7Ca1 is connected with GFP (BBa_E0040),and constructed into pCAMBIA1301 vector.
Protocol
1. Transfection of pCAMBIA-Cry7Ca1-eGFP vector through Carbon dots nanocomposite (CDP) to Nicotiana benthamiana
Coupling CDP with plant expressing vector Mix the following ingredients:
| Ingredients | Volumn |
|---|---|
| MES buffer(50X) | 10ul |
| CDP(50X) | 10ul |
| DNA(237ng/ul >10ng/ul final con) | 22ul |
| 10% glycerol | 25ul |
| ddH2O | 433ul |
| total | 500ul |
Gently mix and incubate at 37℃ for 30min.
2. Brush the mixture gently on the leaf of Nicotiana benthamiana(leaf length>10cm,Growing well) ,mark the area of brushing
3. Put the plant back in the light incubator(28℃,12h light 12h dark), repeat the process for four days at 3:00pm each day(2/4)
Results
Fluorescence microscope to observe the GFP in leave
Figure 3. Western Blot result of Cry7Ca1-eGFP protein express in wheat leaf.
Figure 4. Fluorescence microscopy images of the CDP transfected on plant leaf. 1/3. Blank control group with CDP only; 2/4. CDP with 10ng/ul plant expressing vector.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 205
Illegal NgoMIV site found at 220
Illegal NgoMIV site found at 1567
Illegal NgoMIV site found at 1828
Illegal NgoMIV site found at 1990 - 1000COMPATIBLE WITH RFC[1000]