Difference between revisions of "Part:BBa K1152012"
m |
|||
| Line 2: | Line 2: | ||
<partinfo>BBa_K1152012 short</partinfo> | <partinfo>BBa_K1152012 short</partinfo> | ||
| − | + | [[File:Heidelberg2013_PPtase_entD.png|220px|thumb|left|'''Figure 1:''' Phenotypes of TOP10 producig indigoidine. As negative control untransformed TOP10 were used.]] | |
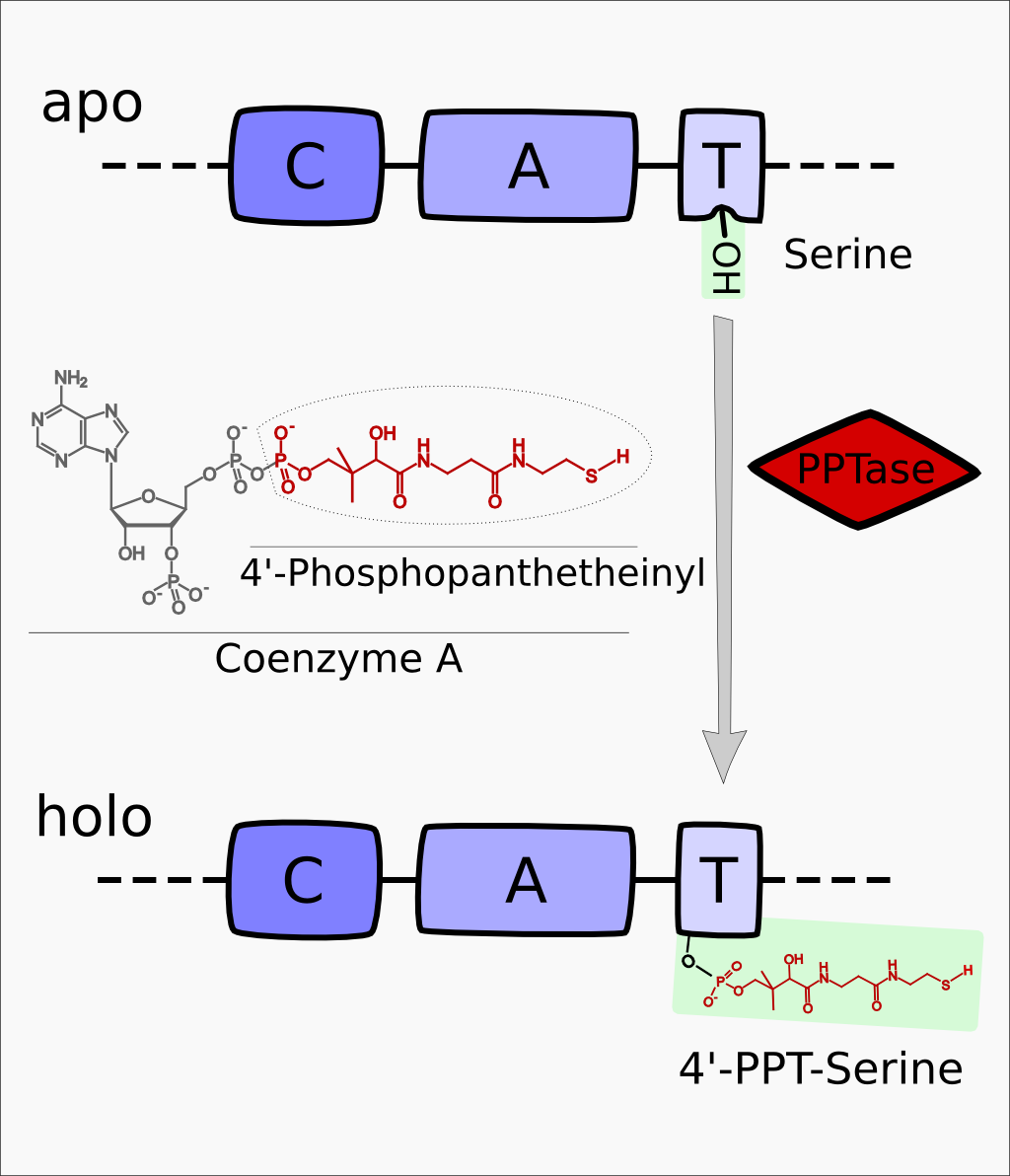
| − | [[File:Heidelberg2013_PPtase_mechanism.png|280px|thumb|'''Figure | + | DelC (Figure 1) encodes a 4'-Phosphopanthetheinyl-transferase (PPTase) which transfers a 4'-PPT residue from CoA to a conserved serine residue in the T-Domain of NRPS modules, thus activating these enzymes (Figure 2). |
| + | |||
| + | [[File:Heidelberg2013_PPtase_mechanism.png|280px|thumb|'''Figure 2:''' General mechanism of activation the activation of NRP Synthetases from apo- to holo-form.]] | ||
| + | |||
| + | |||
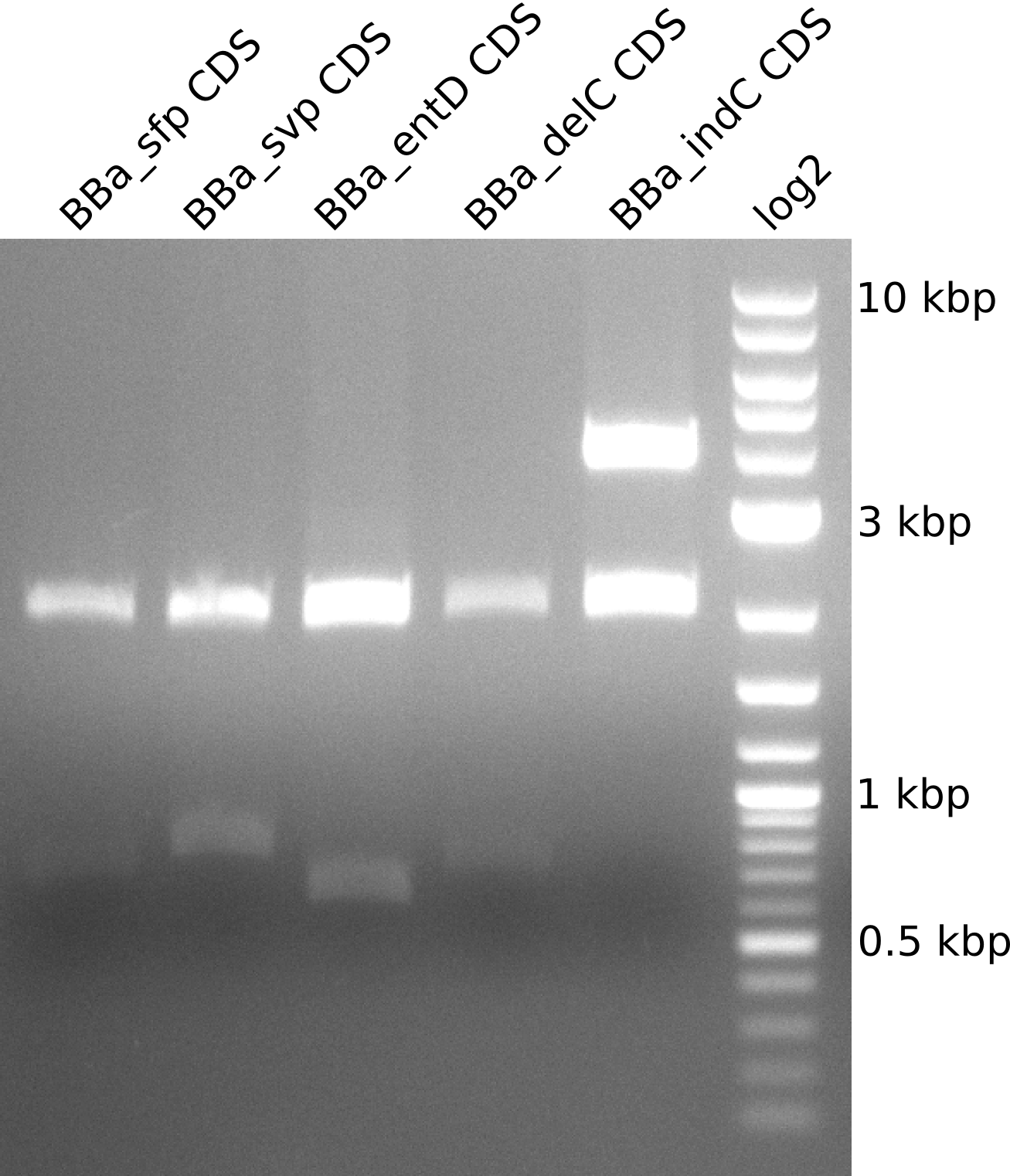
| + | Figure 3 shows the analytical restiction digest we performed for part validation: | ||
| + | [[File:Heidelberg2013_BBa_testdigest_CDS.png|280px|thumb|center|'''Figure 3:''' Restriction digest with EcoRI, XbaI, SpeI, PstI.]] | ||
Revision as of 18:28, 6 October 2013
DelC 4'-Phosphopanthetheinyl-transferase
DelC (Figure 1) encodes a 4'-Phosphopanthetheinyl-transferase (PPTase) which transfers a 4'-PPT residue from CoA to a conserved serine residue in the T-Domain of NRPS modules, thus activating these enzymes (Figure 2).
Figure 3 shows the analytical restiction digest we performed for part validation:
Sequence and Features
Assembly Compatibility:
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000INCOMPATIBLE WITH RFC[1000]Illegal SapI site found at 288
References
- Johnston, C. W., Wyatt, M. a, Li, X., Ibrahim, A., Shuster, J., Southam, G., & Magarvey, N. a. (2013). Gold biomineralization by a metallophore from a gold-associated microbe. Nature chemical biology, 9(4), 241–243. doi:10.1038/nchembio.1179