Difference between revisions of "Part:BBa K338001"
| Line 1: | Line 1: | ||
| − | |||
__NOTOC__ | __NOTOC__ | ||
<partinfo>BBa_K338001 short</partinfo> | <partinfo>BBa_K338001 short</partinfo> | ||
| Line 5: | Line 4: | ||
This is a modified K112400 heat shock promoter in BBa standard. | This is a modified K112400 heat shock promoter in BBa standard. | ||
| − | + | ||
===Usage and Biology=== | ===Usage and Biology=== | ||
| − | + | ====BioBrick Characterization==== | |
| + | =====Effect of Heat Shock Temperature===== | ||
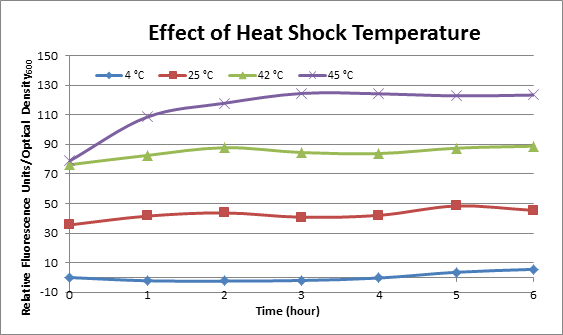
| + | DH5α cells containing HSP-GFP were incubated at 4°C, 25°C, 42°C, and 45°C separately for two hours. Measurements of fluorescence level of GFP were taken using the plate reader at 20 minutes interval and were averaged over 1 hour interval. The vertical axis is raw fluorescence units normalized by OD600. Normalized value of 4°C at t=0 was subtracted from all the values to show a relative difference. | ||
| + | |||
| + | [[Image:Effect of Heat Shock Temperature.png]] | ||
| + | |||
| + | |||
| + | This experiment data suggested that heat shock promoter activity increased with heat shock temperature | ||
| + | |||
| + | =====Effect of Heat Shock Duration===== | ||
| + | |||
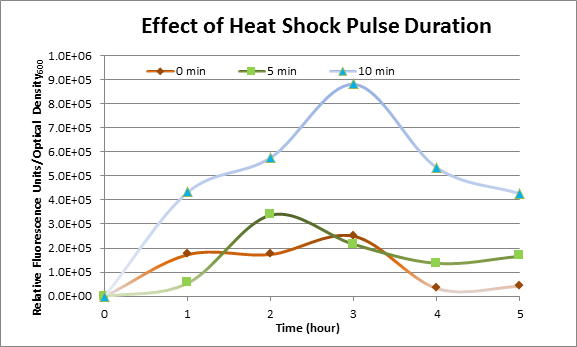
| + | DH5α cells containing HSP-GFP were heat shocked at 42°C for 0 min (kept at room temperature), 5 min and 10 min separately. Measurements of fluorescence level of GFP were taken using the plate reader at 15 minutes interval and were averaged over 1 hour interval. The vertical axis is raw fluorescence units normalized by OD<sub>600</sub>. | ||
| + | |||
| + | [[Image:Effect of Heat Shock Pulse Duration.png]] | ||
| + | |||
| + | The measurements suggest that a heat shock period of 10 minutes significantly increase the production of GFP, while a heat shock period of 5 minutes leads to a similar GFP production level as the not heat shocked cells. Furthermore, the production of GFP shows a decreasing trend after 3 hours suggesting that the heat shock promoter was only activated for a certain time period. | ||
| + | DH5α Cells contained in the images below were incubated at 42°C for 10 minutes, and 6 hours later the fluorescence visualized by an optical microscope with a GFP-specific filter. | ||
| + | |||
| + | [[Image:HSPGFP(1).png]][[Image:HSPGFP(2).png]] | ||
| + | |||
| + | |||
<span class='h3bb'>Sequence and Features</span> | <span class='h3bb'>Sequence and Features</span> | ||
<partinfo>BBa_K338001 SequenceAndFeatures</partinfo> | <partinfo>BBa_K338001 SequenceAndFeatures</partinfo> | ||
Revision as of 23:54, 24 October 2010
Heat Shock Promoter (HSP)
This is a modified K112400 heat shock promoter in BBa standard.
Usage and Biology
BioBrick Characterization
Effect of Heat Shock Temperature
DH5α cells containing HSP-GFP were incubated at 4°C, 25°C, 42°C, and 45°C separately for two hours. Measurements of fluorescence level of GFP were taken using the plate reader at 20 minutes interval and were averaged over 1 hour interval. The vertical axis is raw fluorescence units normalized by OD600. Normalized value of 4°C at t=0 was subtracted from all the values to show a relative difference.
This experiment data suggested that heat shock promoter activity increased with heat shock temperature
Effect of Heat Shock Duration
DH5α cells containing HSP-GFP were heat shocked at 42°C for 0 min (kept at room temperature), 5 min and 10 min separately. Measurements of fluorescence level of GFP were taken using the plate reader at 15 minutes interval and were averaged over 1 hour interval. The vertical axis is raw fluorescence units normalized by OD600.
The measurements suggest that a heat shock period of 10 minutes significantly increase the production of GFP, while a heat shock period of 5 minutes leads to a similar GFP production level as the not heat shocked cells. Furthermore, the production of GFP shows a decreasing trend after 3 hours suggesting that the heat shock promoter was only activated for a certain time period. DH5α Cells contained in the images below were incubated at 42°C for 10 minutes, and 6 hours later the fluorescence visualized by an optical microscope with a GFP-specific filter.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]