Difference between revisions of "Part:BBa K4192020"
Alexandra L (Talk | contribs) |
Alexandra L (Talk | contribs) |
||
| Line 16: | Line 16: | ||
<p>After sequencing and amplifying, that vector as well as unmodified pET30a were transferred into <i>E.coli</i> BL21 (DE3) separately and cultivated on plates.</p> | <p>After sequencing and amplifying, that vector as well as unmodified pET30a were transferred into <i>E.coli</i> BL21 (DE3) separately and cultivated on plates.</p> | ||
| − | First, we used SDS-PAGE to detect whether the protein can be successfully expressed in <i>E.coli</i> BL21 (DE3). The results are as follows:</p> | + | <p>First, we used SDS-PAGE to detect whether the protein can be successfully expressed in <i>E.coli</i> BL21 (DE3). The results are as follows:</p> |
[[File:Sdsobcapic.png|600px|thumb|center|]] | [[File:Sdsobcapic.png|600px|thumb|center|]] | ||
<p style="text-align: center;"><b>Fig.2 ObcA protein in SDS-PAGE.</b></p> | <p style="text-align: center;"><b>Fig.2 ObcA protein in SDS-PAGE.</b></p> | ||
Latest revision as of 15:19, 12 October 2022
obcA, one of the genes required for oxalic acid synthesis
It is one of the genes required for oxalic acid synthesis in Burkholderia glumae, which can act together with ObcB to catalyze oxaloacetic acid (OAA) and citric acid to form oxalic acid (OA). When there is only ObcA, it has a low ability to form oxalic acid.
Characterization
CAU_China 2022 used this part in their oxalate secretion circuit and the expression product of obcA is one of the main enzyme in this circuit. It can act together with ObcB to catalyze oxaloacetic acid (OAA) and citric acid to form oxalic acid (OA)[1]. When there is only ObcA, it has a low ability to form oxalic acid in our project.
Hence, we aim to verify that the gene obcA, originally from Burkholderia glumae, can be regulated by PT7 and lac operator and properly expressed downstream to produce oxalic acid in Escherichia coli.
Fig.1 Genetic circuit for obcA verification (BBa_K1592150).
We build the gene circuit by ClonExpress II one-step cloning kit (Vazyme Biotech, China). And for the T7 promoter, T7 terminator and lac operator, we just followed the structure that came with pET30a.
After sequencing and amplifying, that vector as well as unmodified pET30a were transferred into E.coli BL21 (DE3) separately and cultivated on plates.
First, we used SDS-PAGE to detect whether the protein can be successfully expressed in E.coli BL21 (DE3). The results are as follows:
Fig.2 ObcA protein in SDS-PAGE.
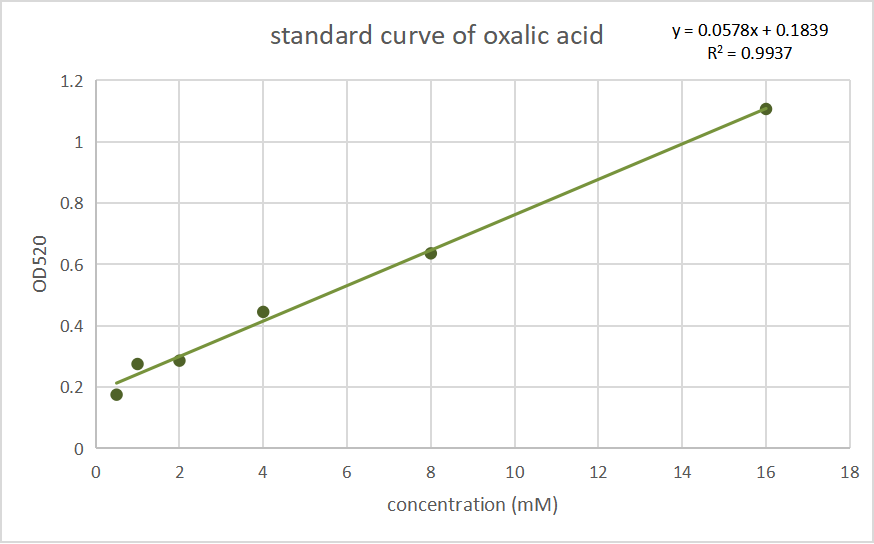
Then we cultivate the recombinant bacteria in the LB liquid culture medium at 200 rpm, 37 ℃ for 12 h, the OD600 of the two bacteria were adjusted to nearly the same (nearly 1.3) using ddH2O, then they were induced by IPTG. Before induction, half of the two bacterial solutions were divided into the control group without induction, which were use as a blank control for colorimetric method. After a cultivation at 120 rpm, 28 ℃ for 8 h, the bacterial solution was collected and then centrifuged at 8000 xg for 1 min. The precipitate was resuspended in a ratio of 200 μl of water per 3 ml of product of the bacterial solution, and ultrasonic crushing for 10 minutes, according to the ultrasonic 3 seconds pause 3 seconds, Amp1 30% parameters. we measured its oxalic acid concentration by Oxalic Acid Content Detection Kit (BOXBIO, Beijing, China), a kit for the determination of oxalic acid concentration by salicylic acid iron colorimetric method. We tested the absorbance at OD520 and convert it into the concentration of oxalic acid inside the thallus. Data are shown below:
Fig.3 Standard curves of oxalate concentration and absorbance at 520 nm.
Fig.4 Oxalic acid concentration results in the verification of obcA.
The control group is negative control, which is an empty pET30 vector. According to the result of t-test(p (0.003) < 0.05), there is significant difference between the two groups of data, indicating that obcA has a great influence on the the oxalic acid produced by the bacteria.
We finally come to the conclusion that obcA can respond to salinity changes in E.coli. Another two parallel experiments were done later, which supported our conclusion. Unfortunately, this concentration is not as high as expected, but it should act with obcB to produce more oxalic acid, as suggested in the literature.[2]
Sequence and Features
- 10INCOMPATIBLE WITH RFC[10]Illegal PstI site found at 184
- 12INCOMPATIBLE WITH RFC[12]Illegal PstI site found at 184
- 21INCOMPATIBLE WITH RFC[21]Illegal BglII site found at 1328
Illegal XhoI site found at 91
Illegal XhoI site found at 1215 - 23INCOMPATIBLE WITH RFC[23]Illegal PstI site found at 184
- 25INCOMPATIBLE WITH RFC[25]Illegal PstI site found at 184
Illegal NgoMIV site found at 176
Illegal NgoMIV site found at 507
Illegal NgoMIV site found at 535
Illegal NgoMIV site found at 571
Illegal NgoMIV site found at 801
Illegal NgoMIV site found at 1578
Illegal AgeI site found at 113 - 1000COMPATIBLE WITH RFC[1000]