Difference between revisions of "Part:BBa K4130000"
(→References) |
(→Characterization) |
||
| Line 17: | Line 17: | ||
We therefore designed a genetic circuit composed of the EibD gene from E. coli strain ECOR-9, a rhamnose-inducible promoter (BBa_K914003), strong ribosome binding site (BBa_B0034), and double-terminator (BBa_B0015). Synthesized parts were assembled into the chloramphenicol-resistance conferring pSB1C3 vector. | We therefore designed a genetic circuit composed of the EibD gene from E. coli strain ECOR-9, a rhamnose-inducible promoter (BBa_K914003), strong ribosome binding site (BBa_B0034), and double-terminator (BBa_B0015). Synthesized parts were assembled into the chloramphenicol-resistance conferring pSB1C3 vector. | ||
| + | |||
===Characterization=== | ===Characterization=== | ||
Revision as of 02:51, 29 September 2022
Contents
IgG F(c) Binding Protein, EibD
Biology
E. coli Ig-binding proteins (Eibs) are a wide-spread class of proteins within intimin-negative, shiga-toxin producing enterohemorrhagic E. coli (EHEC) strains [1]. First discovered in strain ECOR-9 [2, 3], the class has grown to include EibA, C, D, E, F, and G. The class is united in its common characteristic of immunoglobulin (Ig) binding activity.
Immunoglobulins, also known as antibodies, are a family of heterodimeric proteins produced by B cells capable of binding with great specificity to respective antigens. Igs are typically composed of a “variable region”, responsible for binding of the antigen, and a “constant region” with no affinity for the antigen. Igs are further classified according to their “constant regions”, into 5 classes: IgM, IgG, IgA, IgD, and IgE. [4]
E. coli Ig-binding proteins have been shown to be capable of binding the constant region of assorted Ig classes. Affinity and specificity for different classes of Ig proteins varies between Eib family members, with dissociation constants on the 100nM scale [5].
EibD (from E. coli strain ECOR-9) is a 511 amino acid, 210kDa [3] member of the Eib family. The protein exhibits affinity toward both IgG (Kd = ~73nM) and IgA (Kd = ~135nM) [5], with separate binding domains for each Ig [6]. The structure also includes a membrane anchor, left-handed coiled-coil, saddle domain, right-handed coiled-coil, neck, and head domain [6], characteristic of trimeric autotransporter adhesins - a family of secreted proteins in Gram-negative bacteria that are associated with virulence [7]. Besides Ig-binding qualities, heterologous expression of EibD has resulted in self-aggregation phenotypes [6], typical of other trimeric autotransporter adhesins such as YadA [7]. Transmission electron microscopy (TEM) imaging has revealed zipper-like structures forming between neighboring bacteria harboring EibD, explaining the autoaggregation phenotype. Although the biological function of EibD is unknown, its Ig-binding properties and homology to other autotransporter adhesins implicates its role in serum resistance. Binding of IgG by EibD may block binding of immunoglobulins to the adaptive immunity protein C1q, protecting bacteria against innate host defenses [6].
Usage
Given the Ig-binding properties of EibD, we chose to exploit it for whole-cell agglutination/aggregation assays to detect small molecules and proteins. Expression of EibD results in auto-agglutination and biofilm formation that can be observed via the eye. By incubating EibD-expressing bacteria with immunoglobulin antibodies, we hoped to be able to detect various antigens. Introduction of the antibodies would cause visual and quantitative dis-agglutination by molecular competition between self-self interactions and self-antibody interactions. Addition of the antigen would lead to bivalent binding interactions, causing re-agglutination of the cells.
Uniquely, this strategy of detection has the potential to be applied to nearly any antigen with zero need for further genetic engineering. Previous whole-cell bacterial sensors have been focused on expressing antibodies or antibody fragments on the surface of the bacterium, meaning that each strain is specific to an antigen. By expressing a general antibody-binding protein, a single strain can be utilized for the detection of multiple antigens.
We therefore designed a genetic circuit composed of the EibD gene from E. coli strain ECOR-9, a rhamnose-inducible promoter (BBa_K914003), strong ribosome binding site (BBa_B0034), and double-terminator (BBa_B0015). Synthesized parts were assembled into the chloramphenicol-resistance conferring pSB1C3 vector.
Characterization
The EibD biobrick was assembled into the pSB1C3 vector by 3A assembly and cloned into E. coli DH5a for storage and E. coli BL21 for expression experiments.
Response to Induction
Mid-exponential (O.D.600 ~ 0.4 - 0.6) phase bacterial cultures were induced with 0.0% rhamnose, 0.001% rhamnose, and 0.01% rhamnose and grown for 2 hours at 30 C, 200rpm. After induction, the EibD-transformed bacterial cultures appeared markedly different from a BL21 control strain harboring no plasmid. A white precipitate accumulated on the bottom of the EibD cultures induced with 0.001% L-rhamnose, while the uninduced EibD culture exhibited no formation of white precipitate (Figure 1). This white precipitate is likely a result of self-agglutination due to previously reported homophilic interactions between EibD proteins on separate bacteria [6]. We quantified the degree of self-agglutination by measuring the settling rate of bacteria in solution over time. After brief mixing, the optical density at 600 nm (O.D.600) was recorded over a period of 30 minutes. As the agglutinated bacteria settled and precipitated over time, a decrease in optical density was observed. The degree of agglutination was observed to increase with increasing concentrations of L-rhamnose inducer.
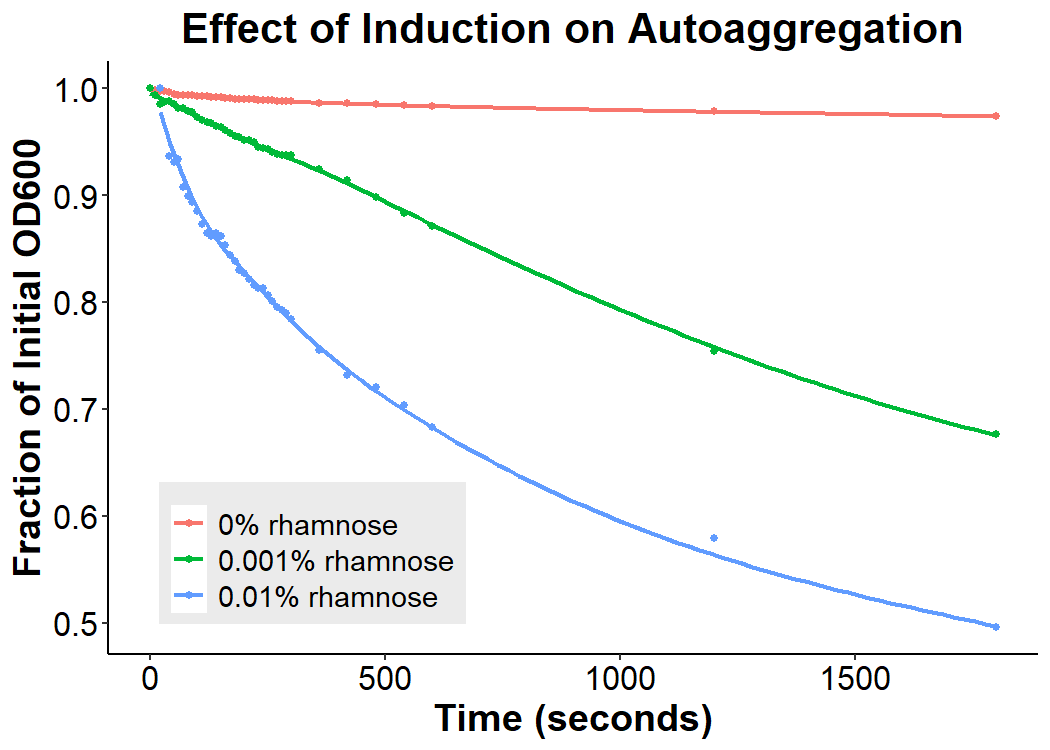
Figure 2: Effect of Induction Concentration on Agglutination. Mid-exponential phase BL21 EibD-transformed cultures were induced with 0%, 0.001%, and 0.01% L-rhamnose and grown for 2 hours. 2mL of pelleted bacteria was resuspended in PBS, briefly mixed, and the optical density at 600nm was monitored for 30 minutes.
References
Merkel V, Ohder B, Bielaszewska M, Zhang W, Fruth A, Menge C, Borrmann E, Middendorf B, Müthing J, Karch H, Mellmann A. Distribution and phylogeny of immunoglobulin-binding protein G in Shiga toxin-producing Escherichia coli and its association with adherence phenotypes. Infect Immun. 2010 Aug;78(8):3625-36. doi: 10.1128/IAI.00006-10. Epub 2010 Jun 14. PMID: 20547747; PMCID: PMC2916290.
Sandt, C. H., Y. D. Wang, R. A. Wilson, and C. W. Hill. 1997. Escherichia coli strains with nonimmune immunoglobulin-binding activity. Infect. Immun. 65:4572-4579.
Sandt, C. H., and C. W. Hill. 2000. Four different genes responsible for nonimmune immunoglobulin-binding activities within a single strain of Escherichia coli. Infect. Immun. 68:2205-2214.
Schroeder HW Jr, Cavacini L. Structure and function of immunoglobulins. J Allergy Clin Immunol. 2010 Feb;125(2 Suppl 2):S41-52. doi: 10.1016/j.jaci.2009.09.046. PMID: 20176268; PMCID: PMC3670108.
Leo, J. C., Goldman, A. 2009. The immunoglobulin-binding Eib proteins from Escheria coli are receptors for IgG Fc. Molecular Immunology 46(8-9):1860-1866.
Leo, J. C., Lyskowski, A., Hattula, K., Hartmann, M. D., Schwarz, H., Butcher, S. J., Linke, D., Lupus, A. N., Goldman, A. 2011. The Structure of E. coli IgG-Binding Protein D Suggests a General Model for Bending and Binding in Trimeric Autotransporter Adhesins. Structure 19(7):1021-1030.
D. Linke, T. Riess, I.B. Autenrieth, A. Lupas, V.A. Kempf. Trimeric autotransporter adhesins: variable structure, common function. Trends Microbiol., 14 (2006), pp. 264-270
