Difference between revisions of "Part:BBa K3717006"
EastonLiaw (Talk | contribs) |
|||
| Line 3: | Line 3: | ||
<partinfo>BBa_K3717006 short</partinfo> | <partinfo>BBa_K3717006 short</partinfo> | ||
| − | + | α-Galactosidase catalyzes the cleavage of the galactose off of B type blood antigens such that the remaining sugar can be classified as a H antigen, which the anti-A and anti-B antibodies are unable to recognize and thus does not elicit an immune response in the human body. Therefore, the enzyme converts B blood types to universal O type. | |
https://static.igem.org/mediawiki/parts/f/f4/T--TAS_Taipei--hisagal.jpg | https://static.igem.org/mediawiki/parts/f/f4/T--TAS_Taipei--hisagal.jpg | ||
| − | + | <b> Figure 1: α-Galactosidase with His-Tag and GS linker </b> | |
| + | |||
| + | |||
| + | <b><font size="+1.2"> Construct Design </font></b> | ||
| + | |||
| + | We attached a 6x histidine tag (6x His-Tag) upstream of the α-Galactosidase sequence followed by a glycine-serine linker (GS linker) to form our open reading frame (ORF) (BBa_K3717006) for purification purposes. We flanked our open reading frame with a T7 promoter + RBS (BBa_K525998) upstream of the open reading frame and a double terminator(BBa_B0015) downstream of the sequence. The composite part was assembled through DNA synthesis by IDT. | ||
| + | |||
| + | |||
| + | <b><font size="+1.2"> Characterization </font></b> | ||
| + | |||
| + | <b><font size="+0.5"> Protein Expression and Purification </font></b> | ||
| + | |||
| + | We tested protein expression of the composite parts by transforming our plasmids into BL21 E. Coli cells. We grew an overnight culture of the BL21 cells with our plasmids then diluted our cells to a standardized OD600 of ~0.1 and let it grow until an OD600 of 0.5~0.6. The diluted cultures of OD600 0.5~0.6 were then induced for expression with 0.5 M IPTG stock (to a final concentration of 0.5mM in the culture) and allowed to grow and induce overnight at room temperature. | ||
| + | |||
| + | Cells were then harvested after the overnight induction and lysed them either through sonication or with xTractor Lysis Buffer (XTractorTM Buffer & XTractor Buffer Kit User Manual, n.d.) spiked with 500mM Imidazole stock (to a final concentration of 20mM in the lysate solution). The Histidine tagged proteins were then purified using Ni sepharose affinity chromatography. SDS-PAGE was then utilized to confirm sizes of purified proteins. | ||
| + | |||
| + | Our results indicate a protein band at roughly 69.7 kDa, which is the molecular weight of our α-Galactosidase ligase enzyme with the 6x His tag and GS linker attached, proving that our α-Galactosidase (Part: BBa_K3717009) was expressed and purified. | ||
| + | |||
| + | https://2021.igem.org/wiki/images/3/3a/T--TAS_Taipei--almostthere.png | ||
| + | |||
| + | <b> Figure 2: SDS-PAGE of purified proteins with the T7 promoter α-Galactosidase expressing construct (BBa_K3717009). Red triangles indicate expected size for the part. </b> | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
<!-- Add more about the biology of this part here | <!-- Add more about the biology of this part here | ||
Revision as of 09:04, 18 October 2021
α-Galactosidase with N-Terminal 6x Histidine tag
α-Galactosidase catalyzes the cleavage of the galactose off of B type blood antigens such that the remaining sugar can be classified as a H antigen, which the anti-A and anti-B antibodies are unable to recognize and thus does not elicit an immune response in the human body. Therefore, the enzyme converts B blood types to universal O type.

Figure 1: α-Galactosidase with His-Tag and GS linker
Construct Design
We attached a 6x histidine tag (6x His-Tag) upstream of the α-Galactosidase sequence followed by a glycine-serine linker (GS linker) to form our open reading frame (ORF) (BBa_K3717006) for purification purposes. We flanked our open reading frame with a T7 promoter + RBS (BBa_K525998) upstream of the open reading frame and a double terminator(BBa_B0015) downstream of the sequence. The composite part was assembled through DNA synthesis by IDT.
Characterization
Protein Expression and Purification
We tested protein expression of the composite parts by transforming our plasmids into BL21 E. Coli cells. We grew an overnight culture of the BL21 cells with our plasmids then diluted our cells to a standardized OD600 of ~0.1 and let it grow until an OD600 of 0.5~0.6. The diluted cultures of OD600 0.5~0.6 were then induced for expression with 0.5 M IPTG stock (to a final concentration of 0.5mM in the culture) and allowed to grow and induce overnight at room temperature.
Cells were then harvested after the overnight induction and lysed them either through sonication or with xTractor Lysis Buffer (XTractorTM Buffer & XTractor Buffer Kit User Manual, n.d.) spiked with 500mM Imidazole stock (to a final concentration of 20mM in the lysate solution). The Histidine tagged proteins were then purified using Ni sepharose affinity chromatography. SDS-PAGE was then utilized to confirm sizes of purified proteins.
Our results indicate a protein band at roughly 69.7 kDa, which is the molecular weight of our α-Galactosidase ligase enzyme with the 6x His tag and GS linker attached, proving that our α-Galactosidase (Part: BBa_K3717009) was expressed and purified.
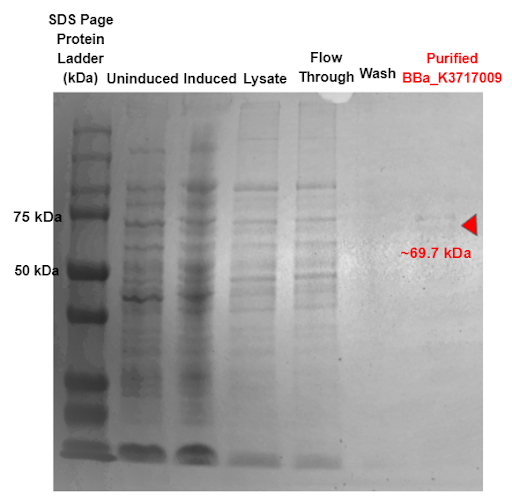
Figure 2: SDS-PAGE of purified proteins with the T7 promoter α-Galactosidase expressing construct (BBa_K3717009). Red triangles indicate expected size for the part.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal AgeI site found at 208
Illegal AgeI site found at 511 - 1000COMPATIBLE WITH RFC[1000]
