Difference between revisions of "Part:BBa K3605010"
| Line 3: | Line 3: | ||
<partinfo>BBa_K3605010 short</partinfo> | <partinfo>BBa_K3605010 short</partinfo> | ||
| − | Since the existing part J04450, a RFP generator, has some leaking expression. We created this modified RFP generator,which only expresses RFP in the presence of IPTG. | + | Since the existing part J04450, a RFP generator, has some leaking expression. We created this modified RFP generator, which only expresses RFP in the presence of IPTG. |
| Line 12: | Line 12: | ||
<br/> | <br/> | ||
This year, we constructed a new RFP generator (BBa_K3605010) also driven by Plac promoter. It contains LacI gene, which can lower significantly the RFP leakage expression. Both LacI and RFP are linked to the downstream of Plac, which is regulated by both LacI repressor and IPTG inducer. Using RFP as a reporter, its fluorescence intensity appears a lower leakage expression. This new RFP generator could be self-regulated because LacI protein can inhibit its self-expression, so that no excessive LacI expression which is considered as waste of resources. We detected the response of this generator to different concentration of IPTG, indicating that it could be inhibited by LacI, and induced well by IPTG. | This year, we constructed a new RFP generator (BBa_K3605010) also driven by Plac promoter. It contains LacI gene, which can lower significantly the RFP leakage expression. Both LacI and RFP are linked to the downstream of Plac, which is regulated by both LacI repressor and IPTG inducer. Using RFP as a reporter, its fluorescence intensity appears a lower leakage expression. This new RFP generator could be self-regulated because LacI protein can inhibit its self-expression, so that no excessive LacI expression which is considered as waste of resources. We detected the response of this generator to different concentration of IPTG, indicating that it could be inhibited by LacI, and induced well by IPTG. | ||
| + | <br/> | ||
| + | <br/> | ||
| + | ==Results:== | ||
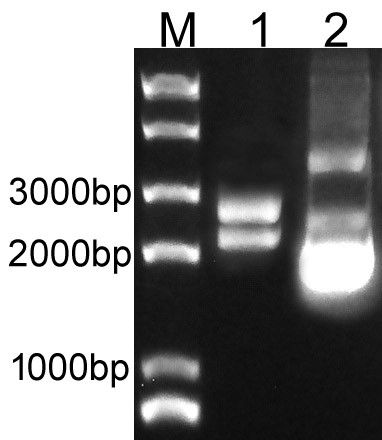
| + | Overlapping PCR was used to clone the LacI and RFP genes. Then the PCR product was inserted to plasmid pSB1C3 to create the new part BBa_K3605010. The identification result is showed in Fig.1. | ||
| + | <br/> | ||
| + | [[File:K3605010-1.jpg|center]] | ||
| + | Fig.1. The result of BBa_K3605010 construction. | ||
| + | M: Marker; 1: Digestion of BBa_K3605010; 2: Plasmid of BBa_K3605010. | ||
| + | <br/> | ||
| + | <br/> | ||
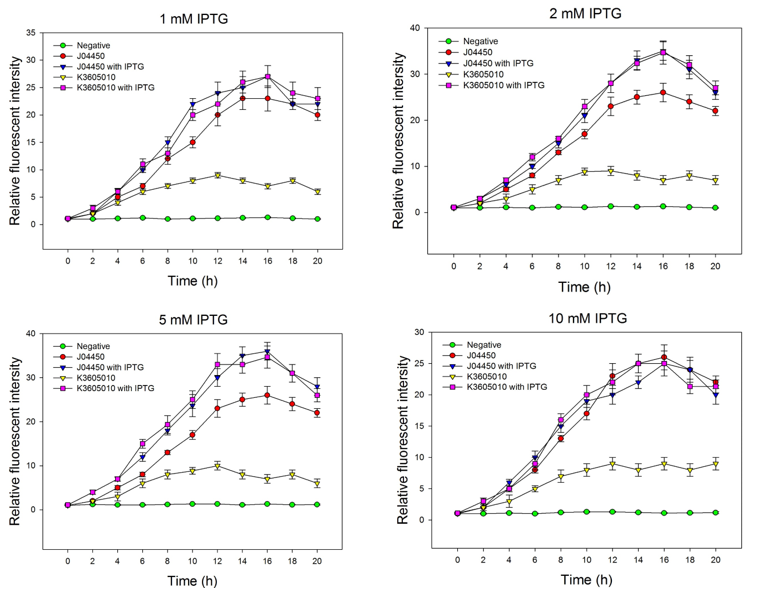
| + | We compared the inducing effect of IPTG on the two RFP generators, using different concentration of IPTG. We set 5 groups: 4 experimental groups of the old RFP generator (BBa_Jo4450) and the new RFP generator (BBa_K3605010) with or without IPTG, one negative control without RFP expression. At 0h, all groups’ OD600 approximately reaches to 0.6, then certain concentration of IPTG was added to the culture medium, incubated cells at 37℃ for 20h. Measure the fluorometric value at 610 nm and OD600 value for each group every 2h, using an automatic microplate reader. The results are showed as follows (Fig.2-Fig.4). | ||
| + | <br/> | ||
| + | [[File:K3605010-2.jpg|center]] | ||
| + | Fig.2. The inducing effect of IPTG on the two RFP generators. Relative fluorescent intensity is fluorescence per OD600 standardized with fluorescence per OD600 value of each test group at time = 0, IPTG=0. The figure indicated that both BBa_J04450 and BBa_K3605010 expressed RFP induced by IPTG, and different concentration of IPTG had same inducing trend. | ||
| + | <br/> | ||
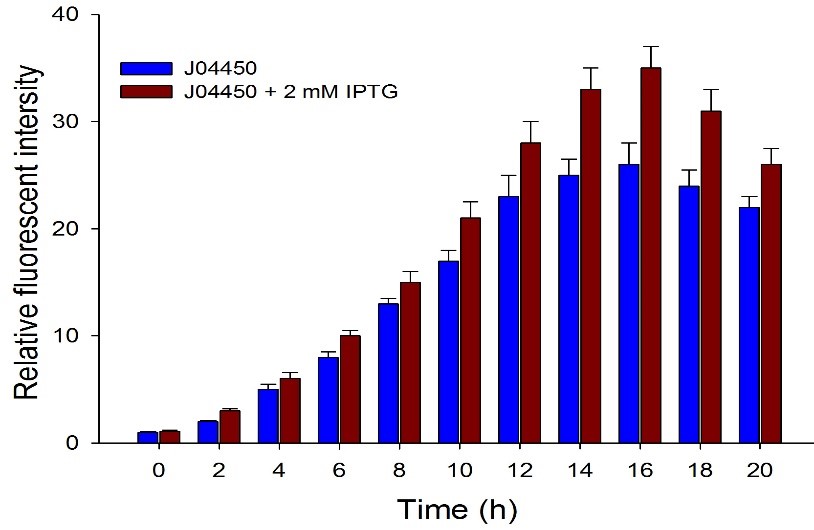
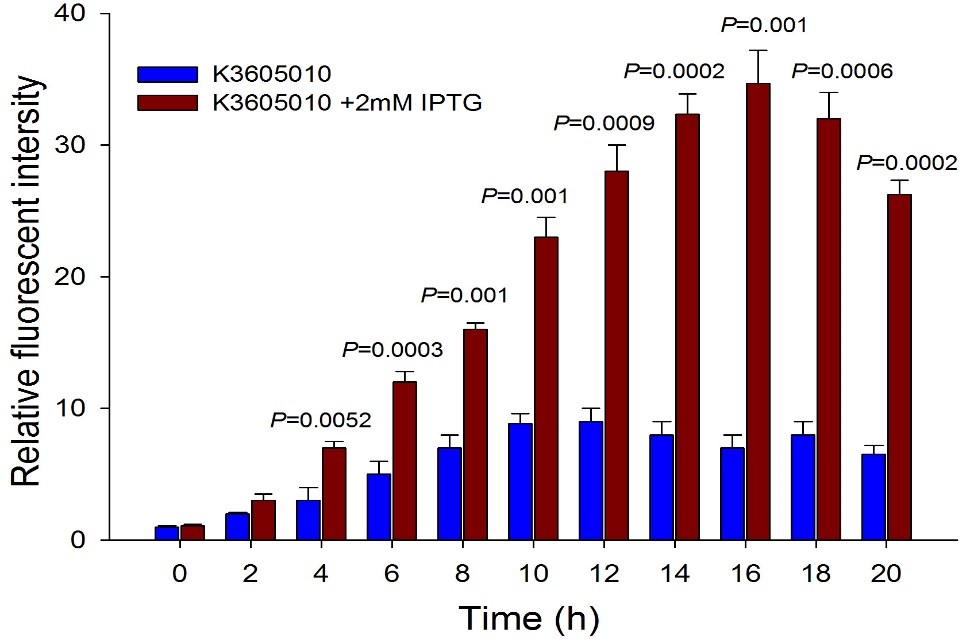
| + | Then we detected the RFP expression with IPTG presence or absence, using BBa_J04450 and BBa_K3605010, respectively. The results showed that BBa_J04450 expressed a mountain of RFP whether IPTG is present or not, which means that BBa_J04450 had a high leakage expression and lower sensitivity to the IPTG induction. However, BBa_K3605010 expressed a high level RFP only with IPTG presence. The leakage expression is very low, and it is sensitive to the IPTG induction. | ||
| + | <br/> | ||
| + | <br/> | ||
| + | [[File:K3605010-3.jpg|center]] | ||
| + | Fig.3. The comparison of RFP expression using BBa_J04450 with and without IPTG induction. Relative fluorescent intensity is fluorescence per OD600 standardized with fluorescence per OD600 value of each test group at time = 0, IPTG=0. This figure indicated that the RFP generator (BBa_J04450) had a high leakage expression, and low sensitivity to the IPTG induction. | ||
| + | <br/> | ||
| + | <br/> | ||
| + | [[File:K3605010-4.jpg|center]] | ||
| + | Fig.4. The comparison of RFP expression using BBa_K3605010 with and without IPTG induction. Relative fluorescent intensity is fluorescence per OD600 standardized with fluorescence per OD600 value of each test group at time = 0, IPTG=0. This figure indicated that the new RFP generator (BBa_K3605010) is very sensitive to the IPTG induction, and the RFP leakage expression is very low. | ||
| + | <br/> | ||
| + | ==Protocol:== | ||
| + | 1.Transform the plasmids into BL21 strain. | ||
| + | 2.Pick up a single colony by a sterile tip from each of the plates for all the experimental and control groups. And put the colony into 5ml M9 medium with 100 µg/ml ampicillin. Incubate at 37℃ in a shaker for 20h. | ||
| + | 3. Detect OD600value of the culture medium with spectrophotometer, and dilute the culture medium to OD600= 0.6. | ||
| + | 4.Add 100 µl bacteria culture medium into a sterile 96-well plate. IPTG is added to final concentrations of 1, 2, 5, 10mM. M9 medium is the blank control. The colony without RFP expression is the negative control. | ||
| + | 5. Incubate at 37℃, and measure the fluorometric value at 610 nm and OD600value for each well every 2h, using an automatic microplate reader. | ||
| + | 6. The experiment should be repeated at least 3 times. | ||
| + | <br/> | ||
| + | <br/> | ||
<!-- Add more about the biology of this part here | <!-- Add more about the biology of this part here | ||
Revision as of 12:57, 26 October 2020
Modified RFP generator
Since the existing part J04450, a RFP generator, has some leaking expression. We created this modified RFP generator, which only expresses RFP in the presence of IPTG.
Overview:
BBa_J04450 is a RFP generator driven by Plac promoter, which is regulated by LacI and induced by IPGT. Plac is one of the most common promoter in life science research field. It is mainly composed of Lac operon containing LacO site. LacI repressor, encoded by LacI gene, can bind to LacO site, inhibiting the binding of RNA polymerase to the promoter. So the expression of downstream genes is blocked. Serving as inducer, IPTG can bind with LacI repressor, making the latter’s conformation changes, so LacI is detached from LacO site, which enables the transcription of downstream genes. BBa_J04450 is a RFP generator driven by Plac promoter, however there is no LacI gene in it. Although the E. coli could express some LacI, it is not enough for inhibition RFP expression. So this RFP generator has some leakage expression, like the designer stated that some RFP express even without IPTG inducer presence.
This year, we constructed a new RFP generator (BBa_K3605010) also driven by Plac promoter. It contains LacI gene, which can lower significantly the RFP leakage expression. Both LacI and RFP are linked to the downstream of Plac, which is regulated by both LacI repressor and IPTG inducer. Using RFP as a reporter, its fluorescence intensity appears a lower leakage expression. This new RFP generator could be self-regulated because LacI protein can inhibit its self-expression, so that no excessive LacI expression which is considered as waste of resources. We detected the response of this generator to different concentration of IPTG, indicating that it could be inhibited by LacI, and induced well by IPTG.
Results:
Overlapping PCR was used to clone the LacI and RFP genes. Then the PCR product was inserted to plasmid pSB1C3 to create the new part BBa_K3605010. The identification result is showed in Fig.1.
Fig.1. The result of BBa_K3605010 construction.
M: Marker; 1: Digestion of BBa_K3605010; 2: Plasmid of BBa_K3605010.
We compared the inducing effect of IPTG on the two RFP generators, using different concentration of IPTG. We set 5 groups: 4 experimental groups of the old RFP generator (BBa_Jo4450) and the new RFP generator (BBa_K3605010) with or without IPTG, one negative control without RFP expression. At 0h, all groups’ OD600 approximately reaches to 0.6, then certain concentration of IPTG was added to the culture medium, incubated cells at 37℃ for 20h. Measure the fluorometric value at 610 nm and OD600 value for each group every 2h, using an automatic microplate reader. The results are showed as follows (Fig.2-Fig.4).
Fig.2. The inducing effect of IPTG on the two RFP generators. Relative fluorescent intensity is fluorescence per OD600 standardized with fluorescence per OD600 value of each test group at time = 0, IPTG=0. The figure indicated that both BBa_J04450 and BBa_K3605010 expressed RFP induced by IPTG, and different concentration of IPTG had same inducing trend.
Then we detected the RFP expression with IPTG presence or absence, using BBa_J04450 and BBa_K3605010, respectively. The results showed that BBa_J04450 expressed a mountain of RFP whether IPTG is present or not, which means that BBa_J04450 had a high leakage expression and lower sensitivity to the IPTG induction. However, BBa_K3605010 expressed a high level RFP only with IPTG presence. The leakage expression is very low, and it is sensitive to the IPTG induction.
Fig.3. The comparison of RFP expression using BBa_J04450 with and without IPTG induction. Relative fluorescent intensity is fluorescence per OD600 standardized with fluorescence per OD600 value of each test group at time = 0, IPTG=0. This figure indicated that the RFP generator (BBa_J04450) had a high leakage expression, and low sensitivity to the IPTG induction.
Fig.4. The comparison of RFP expression using BBa_K3605010 with and without IPTG induction. Relative fluorescent intensity is fluorescence per OD600 standardized with fluorescence per OD600 value of each test group at time = 0, IPTG=0. This figure indicated that the new RFP generator (BBa_K3605010) is very sensitive to the IPTG induction, and the RFP leakage expression is very low.
Protocol:
1.Transform the plasmids into BL21 strain.
2.Pick up a single colony by a sterile tip from each of the plates for all the experimental and control groups. And put the colony into 5ml M9 medium with 100 µg/ml ampicillin. Incubate at 37℃ in a shaker for 20h.
3. Detect OD600value of the culture medium with spectrophotometer, and dilute the culture medium to OD600= 0.6.
4.Add 100 µl bacteria culture medium into a sterile 96-well plate. IPTG is added to final concentrations of 1, 2, 5, 10mM. M9 medium is the blank control. The colony without RFP expression is the negative control.
5. Incubate at 37℃, and measure the fluorometric value at 610 nm and OD600value for each well every 2h, using an automatic microplate reader.
6. The experiment should be repeated at least 3 times.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BglII site found at 1373
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal AgeI site found at 1940
Illegal AgeI site found at 2052 - 1000COMPATIBLE WITH RFC[1000]