Difference between revisions of "Part:BBa K1150003"
| Line 60: | Line 60: | ||
| − | |||
| − | |||
<span class='h3bb'>==Sequence and Features==</span> | <span class='h3bb'>==Sequence and Features==</span> | ||
<partinfo>BBa_K1150003 SequenceAndFeatures</partinfo> | <partinfo>BBa_K1150003 SequenceAndFeatures</partinfo> | ||
| + | |||
===References=== | ===References=== | ||
Revision as of 21:42, 3 October 2013
G9a
| G9a | |
|---|---|
| Function | Histone Methyltransferase |
| Use in | Mammalian cells |
| RFC standard | RFC 25 |
| Backbone | pSB1C3 |
| Organism | Mus musculus |
| Source | Albert Jeltsch, Stuttgart |
| Submitted by | [http://2013.igem.org/Team:Freiburg Freiburg 2013] |
This part contains the catalytically active set domain of G9a on the murine EHMT2 gene. G9a is able to transfer methyl groups to lysine 9 of histone 3 (H3K9), which is part of every nucleosome. In regions of open chromatin structures H3K9 methylation can lead to chromatin condensation and thereby inactivation of the concerned locus. This G9a part alone contains the histone methyltransferase domain needed for transcriptional repression and can be combined with dCas9 Part:BBa_K1150000 to target different loci for transcriptional repression of gene expression.
Usage and Biology
This part belongs to the uniCAS toolkit of iGEM Team Freiburg 2013. In fusion with dCas9 Part:BBa_K1150024 this part can be used to effectively repress target genes by di- or tri-methylation of H3K9. We offer four devices using this part:
- the uniCAS Histone Modifier (CMV promoter) (Part:BBa_K1150024)
- the uniCAS Histone Modifier (SV40 promoter) (Part:BBa_K1150023) uses a medium strong SV40 promoter to optimize this device for means were a strong expression is suboptimal.
- the uniCAS Red Light Switch Part II - Histone Modifier (Part:BBa_K1150028) is a result of combining G9a with part of our red light switch, Phytochrome BPart:BBa_K1150004. PhyB will dimerize with PIF6 upon red light stimulus. PIF6 is fused to dCas9 in the uniCAS Red Light Switch Part I - Stimulator (Part:BBa_K1150025) . Using these two devices G9a is brought into proximity of the DNA targeting dCas9 and can therefore site specifically transfer methyl groups and repress gene expression.
- the uniCAS UV Light Switch Part II - Histone Modifier (Part:BBa_K1150032)
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BglII site found at 597
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
Functional tests
Expression
To ensure protein expression of the fusion protein HEK-293T cells were seeded into 6-wells at a density of 150,000 cells per well. 24 hours later they were transfected with this device.
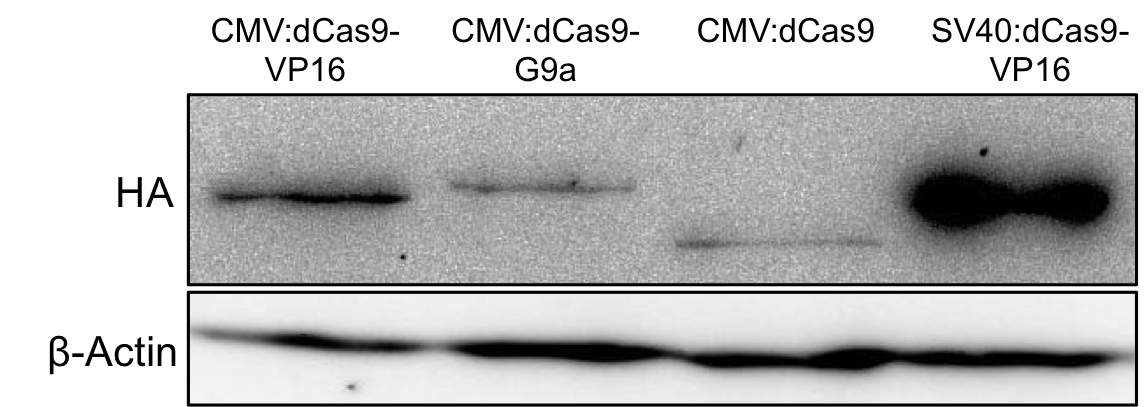
2 days post transfection the protein expression was evaluated by Western blot analysis of total cell lysates. Figure 2 demonstrates successful expression of the dCas9-G9a fusion protein by detection of the HA-tag.
Figure 2.: HA-tag fused to different dCas9-fusion proteins encoded on the RFC25 pSB1C3 backbone.
Proof of Function
Targeting of dCas9-G9a to an open, endogenous locus should lead to changes in the epigenetic state and repression. To test this, the endogenous VEGF-a locus was chosen, as it is well characterized and open in HEK-293T cells. Additionally, VEGF production can easily be measured by ELISA analysis.
Cells were seeded into 24-well format at a density of 65,000 cells per well and were co-transfected with the desired crRNAs, based on the RNAimer. These plasmids are derivates of the RNAimer, the only difference was the insertion of the target sites. These were: -8, -573, +434, -475 and -573/+434. As an off-target control crRNA targeting EMX was used. Every site was targeted with CMV:dCas9 wthout G9a, to differentiate the action of G9a from the CRISPRi effect. As an internal standard a constitutive SEAP reporter was used. 24 hours post transfection, VEGF levels and SEAP levels were measured and the ratio was calculated. Error bars represent standard deviations.
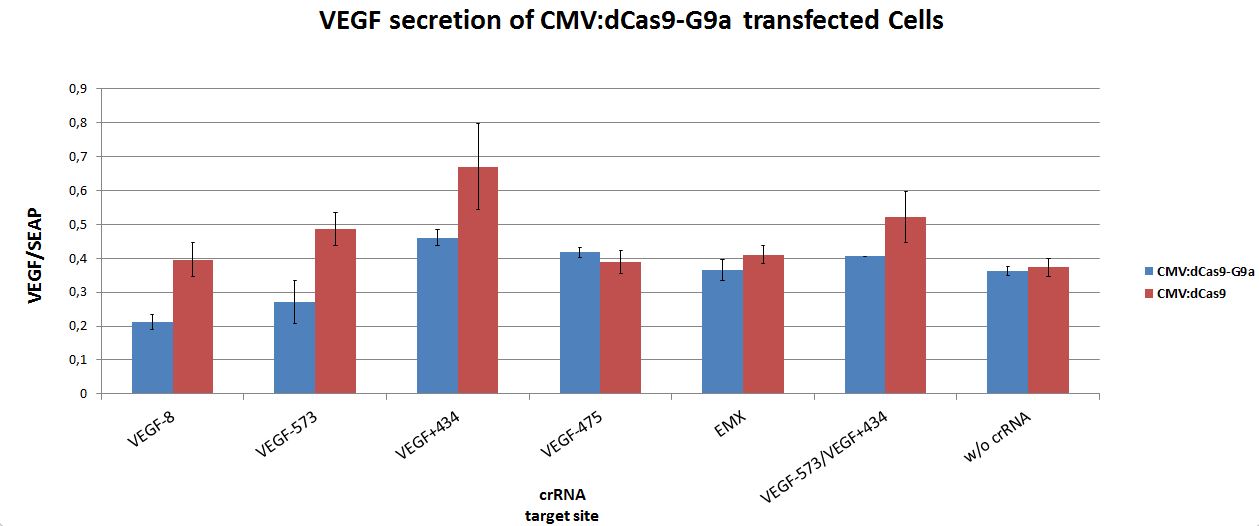
Figure 3.: dCas9-G9a targeting of endogenous VEGF-A locus in HEK-293T cells.
ELISA values, normalized to internal SEAP standard. For some loci clear repressive effects up to 50% were detectable. Controls did not display any off-target effects. Combining two target sites did not show any stronger repression.
Different VEGF target sites led to different repression efficiencies and simulatenous targeting of two sites did not further enhance the observed effects.
In summary, the CMV:dCas9-G9a is an effective repressor of endogenous gene expression, by modulating the chromatin structure of the targeted locus. This offers application in fundamental research as well as in medical science.
==Sequence and Features==
References
[1]Wolffe, A., et al. (1999). Epigenetics: Regulation Through Repression. Science 286169, 481.
[2]Snowden, A., et al. (2002). Gene-Specific Targeting of H3K9 Methylation Is Sufficient for Initiating Repression In Vivo. Current Biology 12, 2159-2166.
[3] Lee, D., et al. (2006). Histone 3 Lysine 9 Methyltransferase G9a Is a Transcriptional Coactivator for Nuclear Receptors. Journal of Biological Chemistry 281, 8476-8485.