Difference between revisions of "Part:BBa K404243"
| Line 7: | Line 7: | ||
===Usage and Biology=== | ===Usage and Biology=== | ||
Integrins are transmembrane proteins that, among other functions, mediate cell attachment to surrounding tissues. They bind to a motif consisting of the amino acids arginine, glycine and aspartic acid (RGD in one-letter code). Because Integrin is highly expressed in many tumor cell lines (Albelda et al., 1990), (Damjanovich, Albelda, Mette, & Buck, 1992), (Lessey et al., 1995), (Smythe, LeBel, Bavaria, Kaiser, & Albelda, 1995), (Gladson & Cheresh, 1991), AAV particles displaying the RGD motif on various positions in their capsid proteins have been created by (Shi et al., 2003). Particles displaying RGD at amino acid positions 584 & 588 as well as 453 or 587 (Boucas et al., 2009) showed transduction efficiencies similar to wt AAV, even when the cells’ HSPG receptors were blocked by heparin sulfate or when the natural HSPG binding motif on the capsid surface was knocked out. To further broaden the area of therapeutic application, we created a ViralBrick containing the RGD motive to specifically target cells with low HSPG-/high Integrin expression. | Integrins are transmembrane proteins that, among other functions, mediate cell attachment to surrounding tissues. They bind to a motif consisting of the amino acids arginine, glycine and aspartic acid (RGD in one-letter code). Because Integrin is highly expressed in many tumor cell lines (Albelda et al., 1990), (Damjanovich, Albelda, Mette, & Buck, 1992), (Lessey et al., 1995), (Smythe, LeBel, Bavaria, Kaiser, & Albelda, 1995), (Gladson & Cheresh, 1991), AAV particles displaying the RGD motif on various positions in their capsid proteins have been created by (Shi et al., 2003). Particles displaying RGD at amino acid positions 584 & 588 as well as 453 or 587 (Boucas et al., 2009) showed transduction efficiencies similar to wt AAV, even when the cells’ HSPG receptors were blocked by heparin sulfate or when the natural HSPG binding motif on the capsid surface was knocked out. To further broaden the area of therapeutic application, we created a ViralBrick containing the RGD motive to specifically target cells with low HSPG-/high Integrin expression. | ||
| + | |||
| + | https://static.igem.org/mediawiki/parts/f/f6/Freiburg10_ViralBrick_motif_RGD.png | ||
| + | |||
<!-- --> | <!-- --> | ||
Revision as of 23:13, 25 October 2010
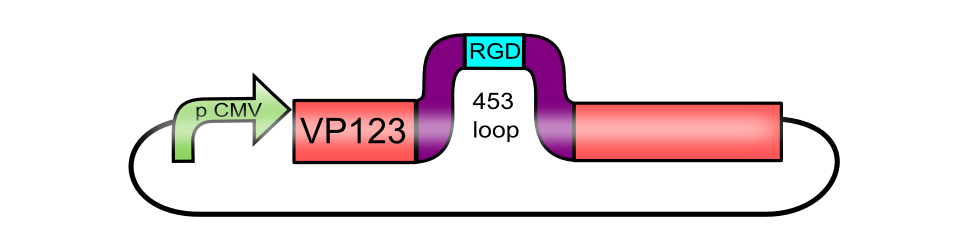
pCMV_[AAV2]VP123 (ViralBrick-453-RGD)
The RGD integrin binding motif, inserted into the 453 loop of pCMV_[AAV2]-VP123
Usage and Biology
Integrins are transmembrane proteins that, among other functions, mediate cell attachment to surrounding tissues. They bind to a motif consisting of the amino acids arginine, glycine and aspartic acid (RGD in one-letter code). Because Integrin is highly expressed in many tumor cell lines (Albelda et al., 1990), (Damjanovich, Albelda, Mette, & Buck, 1992), (Lessey et al., 1995), (Smythe, LeBel, Bavaria, Kaiser, & Albelda, 1995), (Gladson & Cheresh, 1991), AAV particles displaying the RGD motif on various positions in their capsid proteins have been created by (Shi et al., 2003). Particles displaying RGD at amino acid positions 584 & 588 as well as 453 or 587 (Boucas et al., 2009) showed transduction efficiencies similar to wt AAV, even when the cells’ HSPG receptors were blocked by heparin sulfate or when the natural HSPG binding motif on the capsid surface was knocked out. To further broaden the area of therapeutic application, we created a ViralBrick containing the RGD motive to specifically target cells with low HSPG-/high Integrin expression.

Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BamHI site found at 2429
Illegal XhoI site found at 698
Illegal XhoI site found at 884 - 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 665
- 1000INCOMPATIBLE WITH RFC[1000]Illegal BsaI site found at 2955
Illegal SapI site found at 1833