Difference between revisions of "Part:BBa K4035003"
| Line 30: | Line 30: | ||
'''Immunostaining''' | '''Immunostaining''' | ||
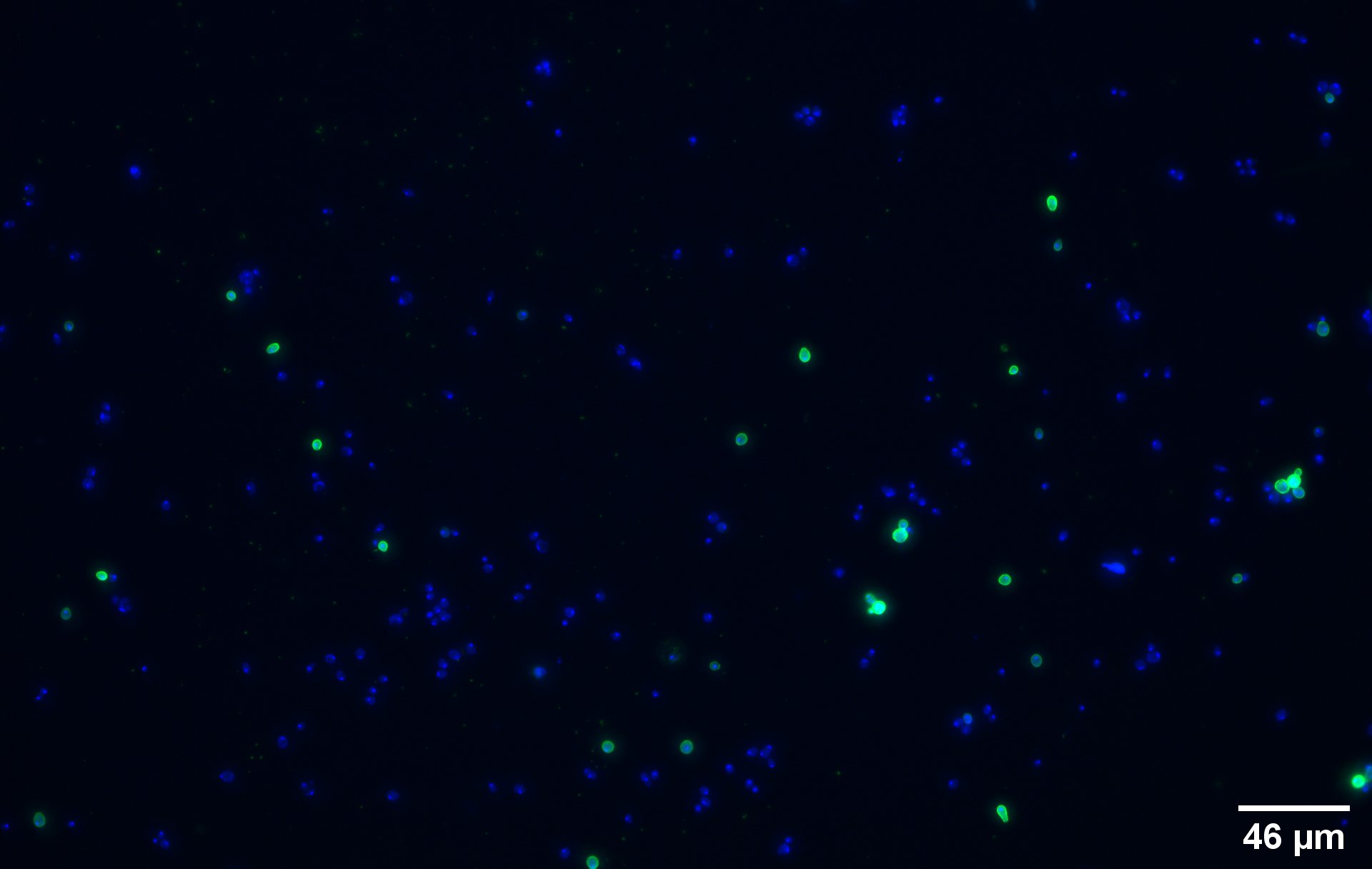
| − | [[File:BBa_K4035003-immuno_image_1.jpg|400px|thumb|right|'''Figure 2a''' : Immunostaining of the yeast transformed with the pCTcon2-CUP1-(GGGGS) | + | [[File:BBa_K4035003-immuno_image_1.jpg|400px|thumb|right|'''Figure 2a''' : Immunostaining of the yeast transformed with the pCTcon2-CUP1-(GGGGS)4-CUP1-V5 plasmid]] |
To show that the fusion protein is expressed at the membrane of the cell we performed an Immunostaining. The cells are incubated with a primary mouse anti-V5 antibody as well as with a secondary goat anti-mouse couple with Alexa Fluor Plus, a green fluorophore. The same strains for control have been used namely, plasmid backbone induced ('''Figure 2c''') and wild type yeast ('''Figure 2d'''). | To show that the fusion protein is expressed at the membrane of the cell we performed an Immunostaining. The cells are incubated with a primary mouse anti-V5 antibody as well as with a secondary goat anti-mouse couple with Alexa Fluor Plus, a green fluorophore. The same strains for control have been used namely, plasmid backbone induced ('''Figure 2c''') and wild type yeast ('''Figure 2d'''). | ||
Revision as of 13:36, 20 October 2021
Dimerization of the copper metallothionein 1 : CUP1-(GGGGS)4-CUP1
This protein is made of two copies of the yeast copper metallotionein protein, CUP1 (BBa_M45090), linked together by a flexible linker made of four times the GGGGS amino acid sequence.
Usage and Biology
Copper metallotionein CUP1 (BBa_M45090) is a protein responsible for copper binding protein in the yeast Saccharomyces cerevisiae. In order to increase the copper retrieval efficiency, two copies of CUP1 were linked together and expressed at the outter surface of S. cerevisiae (BBa_K4035010). The GGGGS linker is a common flexible linker.
Sequence and Features
- 10INCOMPATIBLE WITH RFC[10]Illegal PstI site found at 90
Illegal PstI site found at 408 - 12INCOMPATIBLE WITH RFC[12]Illegal PstI site found at 90
Illegal PstI site found at 408 - 21INCOMPATIBLE WITH RFC[21]Illegal BamHI site found at 235
- 23INCOMPATIBLE WITH RFC[23]Illegal PstI site found at 90
Illegal PstI site found at 408 - 25INCOMPATIBLE WITH RFC[25]Illegal PstI site found at 90
Illegal PstI site found at 408 - 1000INCOMPATIBLE WITH RFC[1000]Illegal SapI.rc site found at 84
Characterization
Expression of the protein in the recombinant yeast
Western Blot Analysis
The protein expression characterization has been accomplished by two experiments, the first being a Western Blot analysis. After having transformed the EBY100 yeast with our newly formed plasmid pCTcon2V5-CUP1-(GGGGS)4-CUP1-V5 we tested its protein expression. For control we also tested the wild type yeast (untransformed) as well as a transformed yeast with the plasmid backbone (without insert) in two different media, one containing galactose and allowing for expression and the other, lacking galactose and thus blocking expression of the protein. As the plasmid contains a Gal1 promoter, the system can only be expressed in the presence of galactose.
Figure 1 : The first two lines, EBY100 and pCTcon2V5 without galactose which are, respectively, wild type yeast and uninduced transformed yeast with the backbone plasmid, serve as negative controls, and show no presence of the V5 tag, as we expected. The third line, pCTcon2V5 + galactose is the induced transformed yeast with the backbone plasmid and shows expression of the V5 tag which proves that our system is expressed in the transformed yeast when induced with galactose. The fourth line is our transformed yeast with the recombinant plasmid pCTcon2-CUP1-(GGGGS)4-CUP1-V5 and also shows expression of the system.
We can see that we have two bands on the gel, one is approximately 30 kDa which is the size of the fusion protein Aga2-CUP1-(GGGGS)4-CUP1-V5 and the other is slightly smaller. The smaller band could be a truncated version of the protein since we identified a second in-frame start codon in the DNA sequence. Unfortunately the truncated version seems to be the most expressed one, corresponding to the signal intensity.
The presence of our CUP1 dimer in our yeast transformants is thus shown.
Immunostaining
To show that the fusion protein is expressed at the membrane of the cell we performed an Immunostaining. The cells are incubated with a primary mouse anti-V5 antibody as well as with a secondary goat anti-mouse couple with Alexa Fluor Plus, a green fluorophore. The same strains for control have been used namely, plasmid backbone induced (Figure 2c) and wild type yeast (Figure 2d).
The nuclei have been stainined with DAPI and are depicted in blue on the figures. The green disks are representing the the recombinant yeast cells expressing the CUP1 dimer at their surface (Figure 2a).
Due to the more intense circle we can clearly see that our system is expressed on the membrane of the protein (this is better seen on Figure 2b which the higher magnification picture of yeast transformed with a single copy of CUP1, part BBa_K4035001). As these are non permeabilized cells, the antibodies bind only the extracellular proteins.
We can also remark that not all the cells are expressing the system. This is because the expression is not 100% efficient.
On the negative control, the little green signal we see is background noise or unspecific antibody binding.

Growth and Survival Characterization
Growth curves of the different yeast strains
In order to check if our expression system would affect the growth of our micro-organism, we performed multiple growth curves with different parameters.
Colony forming assays of the different yeast strains
A second experiment was done to check if the expression system would affect the growth of the yeast cells, a colony forming assay. That experiment consisted of counting the colonies formed by the different yeast strains, respectively wild type, backbone uninduced, backbone induced and transformant, on agar plates.
Copper absorption assays
To finally test our system we imagined an experiment to measure how muchcopper our transformed yeast could absorb.