Difference between revisions of "Part:BBa K3332028"
| Line 5: | Line 5: | ||
Aminoalkylphosphonate N-acetyltransferase with a 6*histidine-tag, which can degradate AMPA. Use BBa_K823004 to construct a new part that can catalyze the acyl transfer reaction between aminomethylphosphoric acid and acetyl-CoA, his tag is used for protein purification. | Aminoalkylphosphonate N-acetyltransferase with a 6*histidine-tag, which can degradate AMPA. Use BBa_K823004 to construct a new part that can catalyze the acyl transfer reaction between aminomethylphosphoric acid and acetyl-CoA, his tag is used for protein purification. | ||
| + | ===Biology=== | ||
| + | Under natural conditions, many microorganisms can use the glyphosate oxidoreductase to oxidize glyphosate to aminomethylphosphoric acid (AMPA). | ||
| + | Phn system is a gene cluster for organophosphorus transport and degradation in many microorganisms. The phnO gene encodes the acyltransferase which catalyzes the transfer of acyl to AMPA by acetyl-coa. Through this one-step acyl transfer reaction, AMPA can be converted into glyphosate analogue, which can then be degraded by C-P lyase. | ||
| + | ===Usage=== | ||
| + | In order to easily purify PhnO, we added a his-tag (6*His) at the C-terminal. we ligased the strong promoter (BBa_J23100) and the parts (RBS-phnO-Terminator) on the expression vector pSB1C3 by standard assembly. Then the ligation mixture was transformed into ''E. coli'' DH5α & ''E. coli'' BL21(DE3), and the correct recombinant one was confirmed by chloramphenicol, enzyme-cut identification and sequencing. | ||
| + | ===Characterization=== | ||
| + | 1. Agarose Gel Electrophoresis | ||
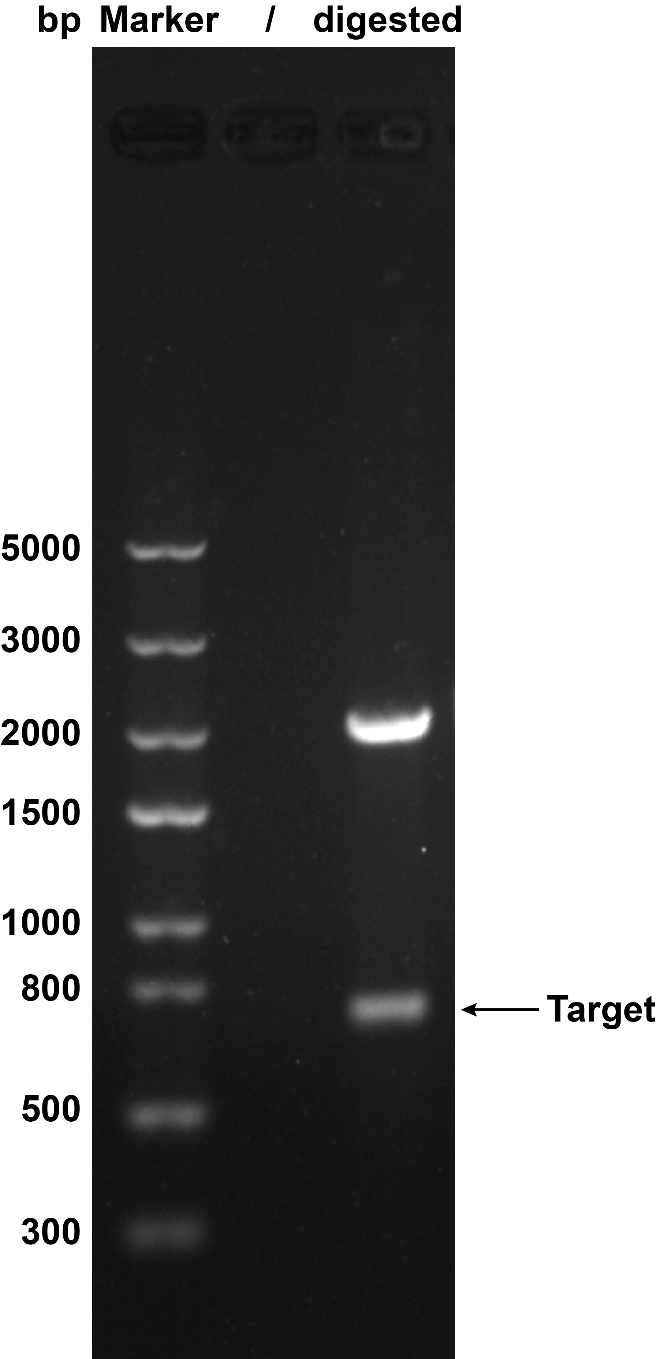
| + | When we were building this circuit, enzyme-cut identification was used to certify the plasmid was correct. We used the ''EcoR''I and ''Pst''I to cut the plasmid, then we got the target separate fragment-686bp. | ||
| + | <table><tr><th>[[File:T--XMU-China2020--BBa K3332028 1.png|thumb|300px|Fig.1 The result of plasmid cut with enzyme ''EcoR''I and ''Pst''I. Plasmid: pSB1C3.]]</th><th></table> | ||
| + | 2. SDS-PAGE | ||
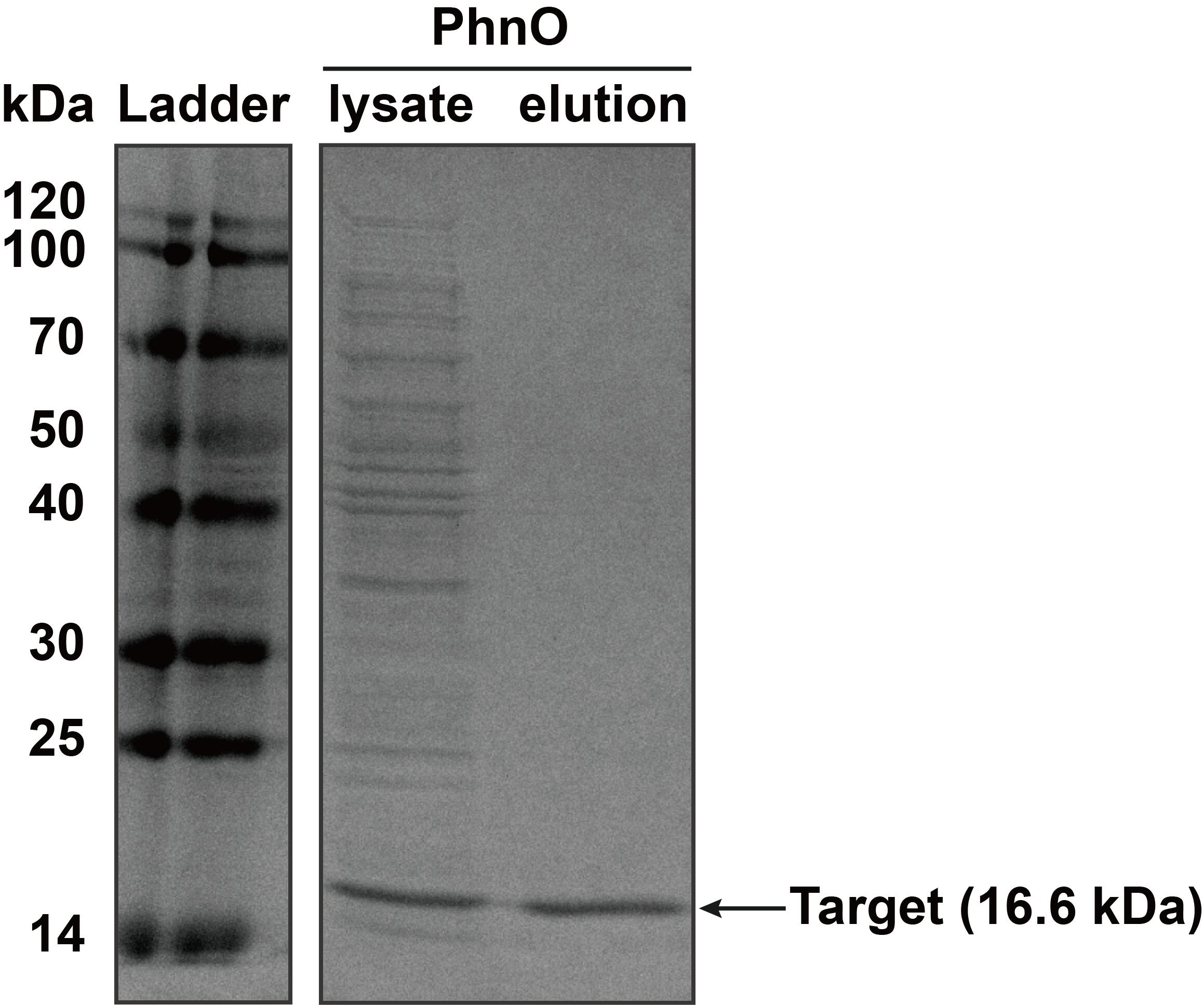
| + | The constructed plasmid was transformed into E. coli BL21 (DE3). Owing to the his-tag, the PhnO could be easily purified through AKTA with Ni-NTA. Compared with the lysate, the PhnO in the eluant can be found at around 16kDa. Both of them were electrophoresed on a sodium dodecyl sulfate (SDS)-12% (wt/vol) polyacrylamide gel, followed by Coomassie blue staining. | ||
| + | <table><tr><th>[[File:T--XMU-China2020--BBa K3332028 2.png|thumb|300px|Fig.2 SDS-PAGE analysis of protein in lysate of ''E. coli'' BL21 (DE3) cells and the eluant after AKTA by Coomassie blue staining. Target bands can be seen at about 16 kDa]]</th><th></table> | ||
| + | 3. Determination of enzyme kinetic constants | ||
| + | Velocities for the reaction of PhnO were determined using 4,4′-dithiodipyridine (DTDP) to continuously detect the formation of the product CoA at 324 nm (thiopyridone: ε= 19800 M-1·cm-1) at 25°C.(1)Enzyme activity was determined through enzyme-labeled instrument. | ||
| + | Km and kcat of different substrates were determined with a concentration gradient. | ||
| + | <table><tr><th>[[File:T--XMU-China2020--BBa K3332028 3A.png;T--XMU-China2020--BBa K3332028 3B.png|thumb|300px|Fig 3. The relationship of 1/Enzyme activity and 1/concentration of AcCoA and AMPA. Used to determine the enzyme kinetic constants.]]</th><th></table> | ||
| + | <table><tr><th>[[File:T--XMU-China2020--BBa K3332028 4.png|thumb|300px|Table 1. Enzyme kinetic constants with two substrates.]]</th><th></table> | ||
| − | |||
| − | |||
| − | |||
<!-- --> | <!-- --> | ||
Revision as of 03:57, 25 October 2020
phnO-his-tag
Aminoalkylphosphonate N-acetyltransferase with a 6*histidine-tag, which can degradate AMPA. Use BBa_K823004 to construct a new part that can catalyze the acyl transfer reaction between aminomethylphosphoric acid and acetyl-CoA, his tag is used for protein purification.
Biology
Under natural conditions, many microorganisms can use the glyphosate oxidoreductase to oxidize glyphosate to aminomethylphosphoric acid (AMPA). Phn system is a gene cluster for organophosphorus transport and degradation in many microorganisms. The phnO gene encodes the acyltransferase which catalyzes the transfer of acyl to AMPA by acetyl-coa. Through this one-step acyl transfer reaction, AMPA can be converted into glyphosate analogue, which can then be degraded by C-P lyase.
Usage
In order to easily purify PhnO, we added a his-tag (6*His) at the C-terminal. we ligased the strong promoter (BBa_J23100) and the parts (RBS-phnO-Terminator) on the expression vector pSB1C3 by standard assembly. Then the ligation mixture was transformed into E. coli DH5α & E. coli BL21(DE3), and the correct recombinant one was confirmed by chloramphenicol, enzyme-cut identification and sequencing.
Characterization
1. Agarose Gel Electrophoresis When we were building this circuit, enzyme-cut identification was used to certify the plasmid was correct. We used the EcoRI and PstI to cut the plasmid, then we got the target separate fragment-686bp.
2. SDS-PAGE The constructed plasmid was transformed into E. coli BL21 (DE3). Owing to the his-tag, the PhnO could be easily purified through AKTA with Ni-NTA. Compared with the lysate, the PhnO in the eluant can be found at around 16kDa. Both of them were electrophoresed on a sodium dodecyl sulfate (SDS)-12% (wt/vol) polyacrylamide gel, followed by Coomassie blue staining.
3. Determination of enzyme kinetic constants Velocities for the reaction of PhnO were determined using 4,4′-dithiodipyridine (DTDP) to continuously detect the formation of the product CoA at 324 nm (thiopyridone: ε= 19800 M-1·cm-1) at 25°C.(1)Enzyme activity was determined through enzyme-labeled instrument. Km and kcat of different substrates were determined with a concentration gradient.
File:T--XMU-China2020--BBa K3332028 3A.png;T--XMU-China2020--BBa K3332028 3B.png Fig 3. The relationship of 1/Enzyme activity and 1/concentration of AcCoA and AMPA. Used to determine the enzyme kinetic constants. |
|---|
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BamHI site found at 159
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]