Difference between revisions of "Part:BBa K3078004"
| Line 19: | Line 19: | ||
<h1>'''2. Characterization'''</h1> | <h1>'''2. Characterization'''</h1> | ||
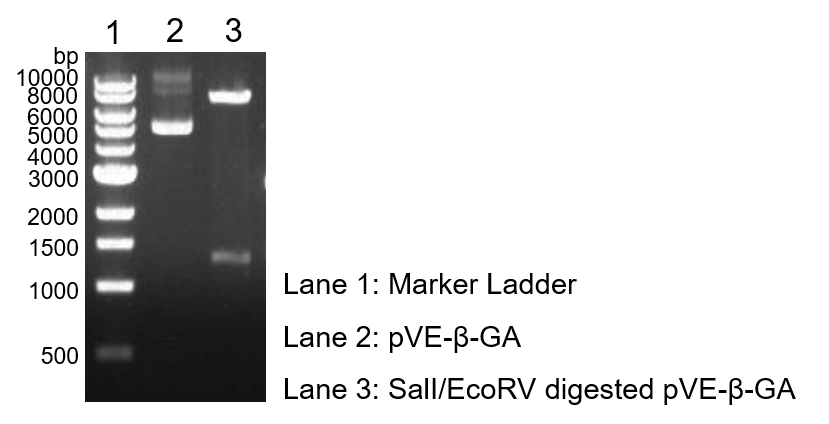
| − | <h4>'''2.1 Validation of | + | <h4>'''2.1 Validation of β-1,3-glucanase construction'''</h4> |
<h5> | <h5> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
| Line 33: | Line 33: | ||
| − | <h4>'''2.2 Expression of | + | <h4>'''2.2 Expression of β-1,3-glucanase '''</h4> |
<h5> | <h5> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
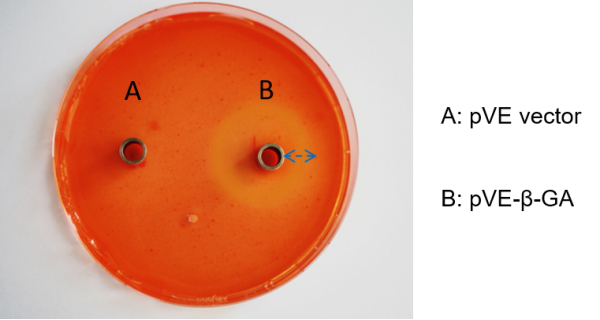
| − | To assess the β-1,3-glucanase expression of our construct, Congo Red experiment was used. Congo Red has a strong red chromogenic reaction with β-1,3-glucan. In contrast, when β-1, 3-glucan is decomposed into reducing monosaccharides by β-1,3-glucanase, the hydrolyzed region forms a pale yellow transparent hydrolytic circle. Compared with control, there was a larger size of transparent hydrolytic circle caused by β-1,3-glucanase expressed in | + | To assess the β-1,3-glucanase expression of our construct, Congo Red experiment was used. Congo Red has a strong red chromogenic reaction with β-1,3-glucan. In contrast, when β-1, 3-glucan is decomposed into reducing monosaccharides by β-1,3-glucanase, the hydrolyzed region forms a pale yellow transparent hydrolytic circle. Compared with control, there was a larger size of transparent hydrolytic circle caused by β-1,3-glucanase expressed in E. coli with pVE-β-GA, indicating the expression of β-1,3-glucanase (Figure 2). |
</p> | </p> | ||
</h5> | </h5> | ||
[[File:B132.png|center|B132]] | [[File:B132.png|center|B132]] | ||
<center style="text-align:left;"> | <center style="text-align:left;"> | ||
| − | Figure 2. Expression of β-1,3-glucanase. Add 10 mg/mL Congo Red solution to LB medium containing β-1,3-glucan substrate (0.1 g/100 mL) at a ratio of 1:100. 100 μL supernatant obtained by centrifugation after ultrasonic crushing of | + | Figure 2. Expression of β-1,3-glucanase. Add 10 mg/mL Congo Red solution to LB medium containing β-1,3-glucan substrate (0.1 g/100 mL) at a ratio of 1:100. 100 μL supernatant obtained by centrifugation after ultrasonic crushing of E. coli with pVE-β-GA is added to the Oxford cup, and the pVE empty vector bacteria supernatant is used as the control. Stand at 37 ℃ for 24 hours. |
</center> | </center> | ||
| Line 48: | Line 48: | ||
<h5> | <h5> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
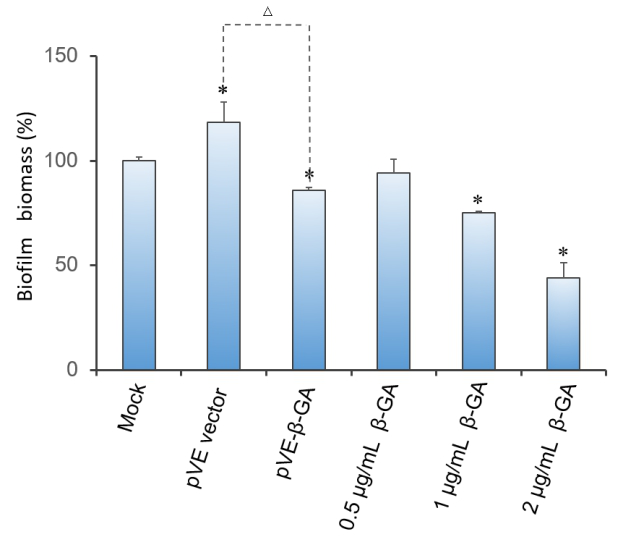
| − | Crystal violet (CV) reduction method, which is commonly used for quantitative analysis of biofilm, was used to evaluate the antibiofilm activity of β-1,3-glucanase. Under the condition of using bacteria with pVE5523 vector(pVE vector) to exclude the influence of bacterial substances on the staining results, as Figure | + | Crystal violet (CV) reduction method, which is commonly used for quantitative analysis of biofilm, was used to evaluate the antibiofilm activity of β-1,3-glucanase. Under the condition of using bacteria with pVE5523 vector(pVE vector) to exclude the influence of bacterial substances on the staining results, as Figure 10 shows, β-1,3-glucanase produced by our engineered bacteria has the effect of degrading biofilm. The supernatant of E. coli with pVE-β-GA diluted by one time is estimated to reach the effect of 0.5 μg/mL~2 μg/mL samples of standard β-1,3-glucanase. The result demonstrated that β-1,3-glucanase had disruption effect on mature biofilm with concentration-dependent manner (Figure 3). |
</p> | </p> | ||
</h5> | </h5> | ||
[[File:B133.png|center|B133]] | [[File:B133.png|center|B133]] | ||
<center style="text-align:left;"> | <center style="text-align:left;"> | ||
| − | Figure 3. Degradation effect of β-1,3-glucanase on biofilm. Biofilm formed in RPMI 1640 medium for 48 hrs. Mature biofilm was treated with RPMI 1640 medium, bacteria supernatant of pVE vector or pVE-β-GA | + | Figure 3. Degradation effect of β-1,3-glucanase on biofilm. Biofilm formed in RPMI 1640 medium for 48 hrs. Mature biofilm was treated with RPMI 1640 medium, bacteria supernatant of pVE vector or pVE-β-GA or standard β-1,3-glucanase in different concentrations (0.5, 1 and 2 μg/mL) for another 24 hrs. Values obtained are given as the percentage of biofilm. The experiment was performed three times in triplicate. *, P < 0.05 from mock control using Student’s t test. △, P < 0.05. |
</center> | </center> | ||
Revision as of 23:16, 21 October 2019
β-1,3-glucanase
β-1,3-glucanase protein coding region. β-1,3-glucanase can degrade biofilm.
1. Usage and Biology
β-1,3-glucan is one of the primary components in C. albicans biofilm EPS, which is important for Candida biofilm formation and resistance to stresses. The enzyme β-1,3-glucanase, form Cellulosimicrobium cellulans, can degrade β-1,3-glucan. Therefore, this year, we decided use β-1,3-glucanase to disrupt the Candida biofilm matrix and increase the effect of the antimicrobial drug.
2. Characterization
2.1 Validation of β-1,3-glucanase construction
To verify the construction of pVE-β-1,3-glucanase (pVE-β-GA) which we generated, the digestion by SalI/EcoRV was performed by a standard protocol followed by agarose gel electrophoresis (Figure 1).
Figure 1. Digestion and electrophoresis of pVE-β-GA.
2.2 Expression of β-1,3-glucanase
To assess the β-1,3-glucanase expression of our construct, Congo Red experiment was used. Congo Red has a strong red chromogenic reaction with β-1,3-glucan. In contrast, when β-1, 3-glucan is decomposed into reducing monosaccharides by β-1,3-glucanase, the hydrolyzed region forms a pale yellow transparent hydrolytic circle. Compared with control, there was a larger size of transparent hydrolytic circle caused by β-1,3-glucanase expressed in E. coli with pVE-β-GA, indicating the expression of β-1,3-glucanase (Figure 2).
Figure 2. Expression of β-1,3-glucanase. Add 10 mg/mL Congo Red solution to LB medium containing β-1,3-glucan substrate (0.1 g/100 mL) at a ratio of 1:100. 100 μL supernatant obtained by centrifugation after ultrasonic crushing of E. coli with pVE-β-GA is added to the Oxford cup, and the pVE empty vector bacteria supernatant is used as the control. Stand at 37 ℃ for 24 hours.
2.3 Degradation effect of β-1,3-glucanase on biofilm
Crystal violet (CV) reduction method, which is commonly used for quantitative analysis of biofilm, was used to evaluate the antibiofilm activity of β-1,3-glucanase. Under the condition of using bacteria with pVE5523 vector(pVE vector) to exclude the influence of bacterial substances on the staining results, as Figure 10 shows, β-1,3-glucanase produced by our engineered bacteria has the effect of degrading biofilm. The supernatant of E. coli with pVE-β-GA diluted by one time is estimated to reach the effect of 0.5 μg/mL~2 μg/mL samples of standard β-1,3-glucanase. The result demonstrated that β-1,3-glucanase had disruption effect on mature biofilm with concentration-dependent manner (Figure 3).
Figure 3. Degradation effect of β-1,3-glucanase on biofilm. Biofilm formed in RPMI 1640 medium for 48 hrs. Mature biofilm was treated with RPMI 1640 medium, bacteria supernatant of pVE vector or pVE-β-GA or standard β-1,3-glucanase in different concentrations (0.5, 1 and 2 μg/mL) for another 24 hrs. Values obtained are given as the percentage of biofilm. The experiment was performed three times in triplicate. *, P < 0.05 from mock control using Student’s t test. △, P < 0.05.
3. Conclusion
Our engineered bacteria successfully characterized β-1,3-glucanase,moreover, it was effective in performing the function of degrading C. albicans biofilm.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 274
Illegal NgoMIV site found at 483
Illegal NgoMIV site found at 622 - 1000COMPATIBLE WITH RFC[1000]