Difference between revisions of "Part:BBa K3182104"
| Line 44: | Line 44: | ||
<h1>Antimicrobial Agent</h1> | <h1>Antimicrobial Agent</h1> | ||
| − | + | Bacteriophage endolysins digest the peptidoglycan wall of bacteria. The LysK is an endolysin targeting gram+ bacteria. It has two catalytical domains known as CHAP (cysteine-and histidine dependent amidohydrolase/peptidase) and a central amidase-2 domain together with a cell binding domain. The CHAP domain which is the truncated part expressed in this year’s LiU iGEM (more specifically CHAP C1-C162) team still has lytic activity against staphylococcus. The C1 to C162 amino acids are required for full endopeptidase activity | |
<br> | <br> | ||
<br> | <br> | ||
Revision as of 13:39, 6 October 2019
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BamHI site found at 580
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
Introduction
pT7-CBDcipA-LysK
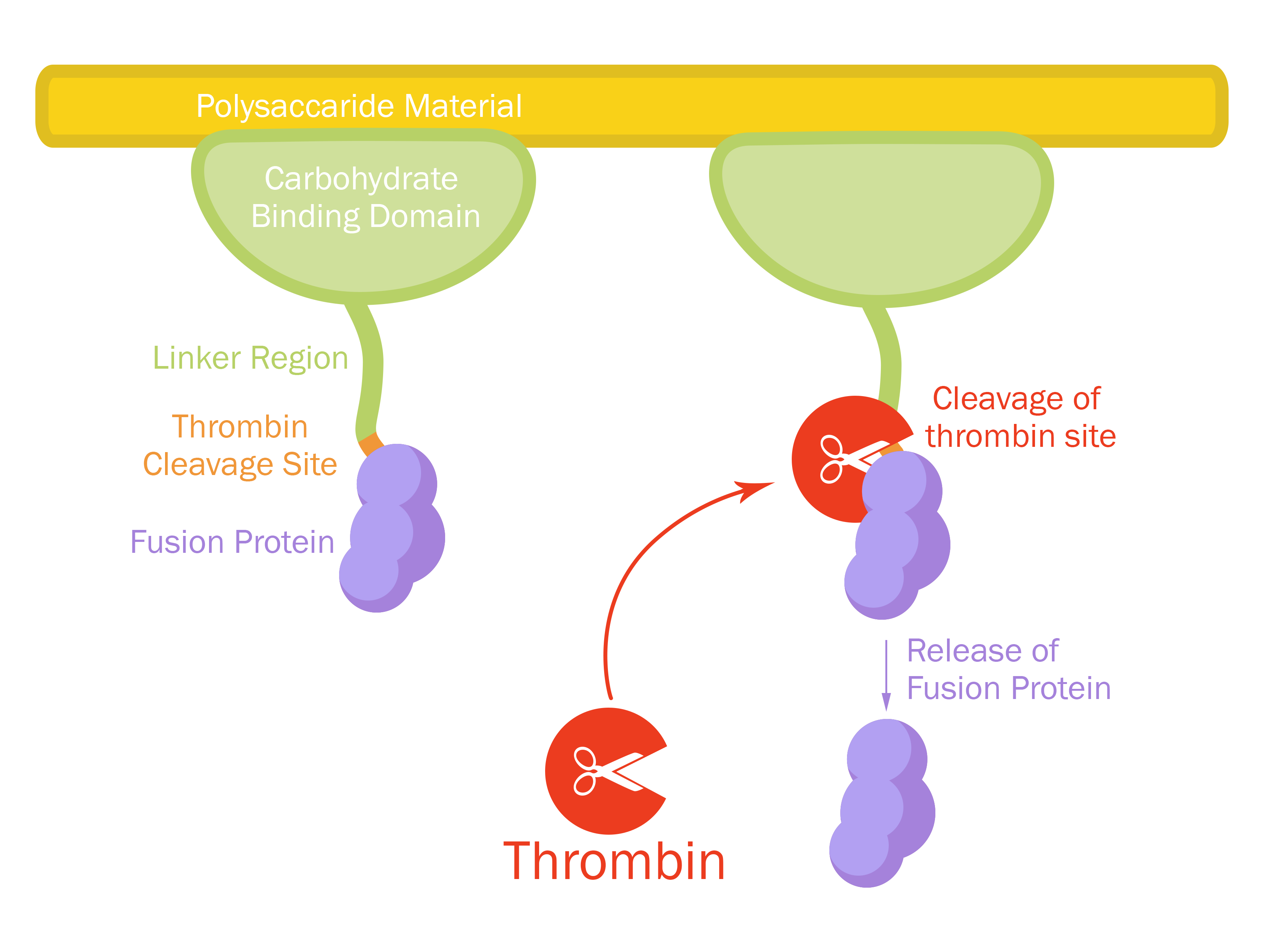
This part consists of a carbohydrate binding domain (CBD) from Clostridium thermocellum (C. thermocellum) cellulose scaffolding protein (CipA) and is a central part Clostridium thermocellum's cellusome. The CBD-fusion were fused using a flexible GS-linker (-GGGGSGGGGS-). A thrombin cleavage site (-LVPRGS-) was added to the end of the linker and its breakage will leave a glycine and serine attached to the N-terminal of the fusion protein.
Protease site and use
The thrombin site was added to enable the ability to release the fusion protein down into skin wounds. Because of our integrated human practice we learned that infection span much deeper into wounds that we thought. Simply attaching the CBD-fusion protein to a carbohydrate material wouldn't make the fusion protein reach far into the wound. The thrombin site was also chosen because of thrombins endogenous occurrence in humans.
Assembly compabilities
An internal BamHI recognition sequence (RS) has been added to enable changeable fusion proteins. BamHI was chosen because its RS codes for glycine and serine, fitting it to the end of the thrombin site. It is also a cost-effective enzyme and is unaffected by methylated DNA.
CBDcipA crystal structure
Important molecular faces
CBDcipA is composed of a nine-stranded beta sandwich with a jelly roll topology and binds a calcium ion. It further contains conserved residues exposed on the surface which map into two clear surfaces on each side of the molecule. One of faces mainly contains planar strips of aromatic and polar residues which may be the carbohydrate binding part. Further aspect are unknown and unique with this CBD such as the other conserved residues which are contained in a groove.
Carbohydrate binding domain specificity
Since the CBD is from the cellusome of C. thermocellum some researches call it a cellulose binding domain. However, iGEM19 Linköping noticed that this domain could also bind to different sources of polysaccaride materials. This serves as a domain for iGEM19 Linköpings modular bandage, where the polysaccaride material can be changed for anything and not exclusively cellulose.
The choice of carbohydrate binding domain
iGEM Linköping 2019 choose CBDcipA due to many other iGEM teams exploring the possibilities of this domain. Our basic design was influenced by [http://2014.igem.org/Team:Imperial iGEM14 Imperial], [http://2015.igem.org/Team:edinburgh iGEM15 Edinburgh] and [http://2018.igem.org/Team:ecuador iGEM18 Ecuador]. Purification and where to place the fusion protein (N- or C-terminal) was determined by studying the former projects. CBDcipA also originates from a thermophilic bacteria which further increases the domains applications.
Expression system
The part has a strong expression with a T7-RNA-polymerase promotor (BBa_I719005) as well as a 5'-UTR (BBa_K1758100) region which has been shown to further increase expression in E. coli (BBa_K1758106), ([http://www.ncbi.nlm.nih.gov/pubmed/2676996 Olins et al. 1989]), ([http://www.ncbi.nlm.nih.gov/pubmed/23927491 Takahashi et al. 2013]).

Antimicrobial Agent
Bacteriophage endolysins digest the peptidoglycan wall of bacteria. The LysK is an endolysin targeting gram+ bacteria. It has two catalytical domains known as CHAP (cysteine-and histidine dependent amidohydrolase/peptidase) and a central amidase-2 domain together with a cell binding domain. The CHAP domain which is the truncated part expressed in this year’s LiU iGEM (more specifically CHAP C1-C162) team still has lytic activity against staphylococcus. The C1 to C162 amino acids are required for full endopeptidase activity
The endolysin is designed to battle the Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, Enterobacter spp. family of pathogens (ESKAPE). ESKAPE is a family(ies) of bacteria which has multiple understrains that has evolved resistance to the most commonly used antibiotics.
Antimicrobial activity of CHAP
Sequence and Features




