Difference between revisions of "Part:BBa K3182000"
| Line 17: | Line 17: | ||
This part can be used to track purification, measure CBD binding ability and report cleavage at the thrombin site. | This part can be used to track purification, measure CBD binding ability and report cleavage at the thrombin site. | ||
| − | <br><br><br><br><br><br> | + | <br><br><br><br><br><br><br> |
<h2>CBDcipA crystal structure</h2> | <h2>CBDcipA crystal structure</h2> | ||
Revision as of 16:05, 6 September 2019
Contents
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BamHI site found at 580
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
Introduction
pT7-CBDcipA-AsPink
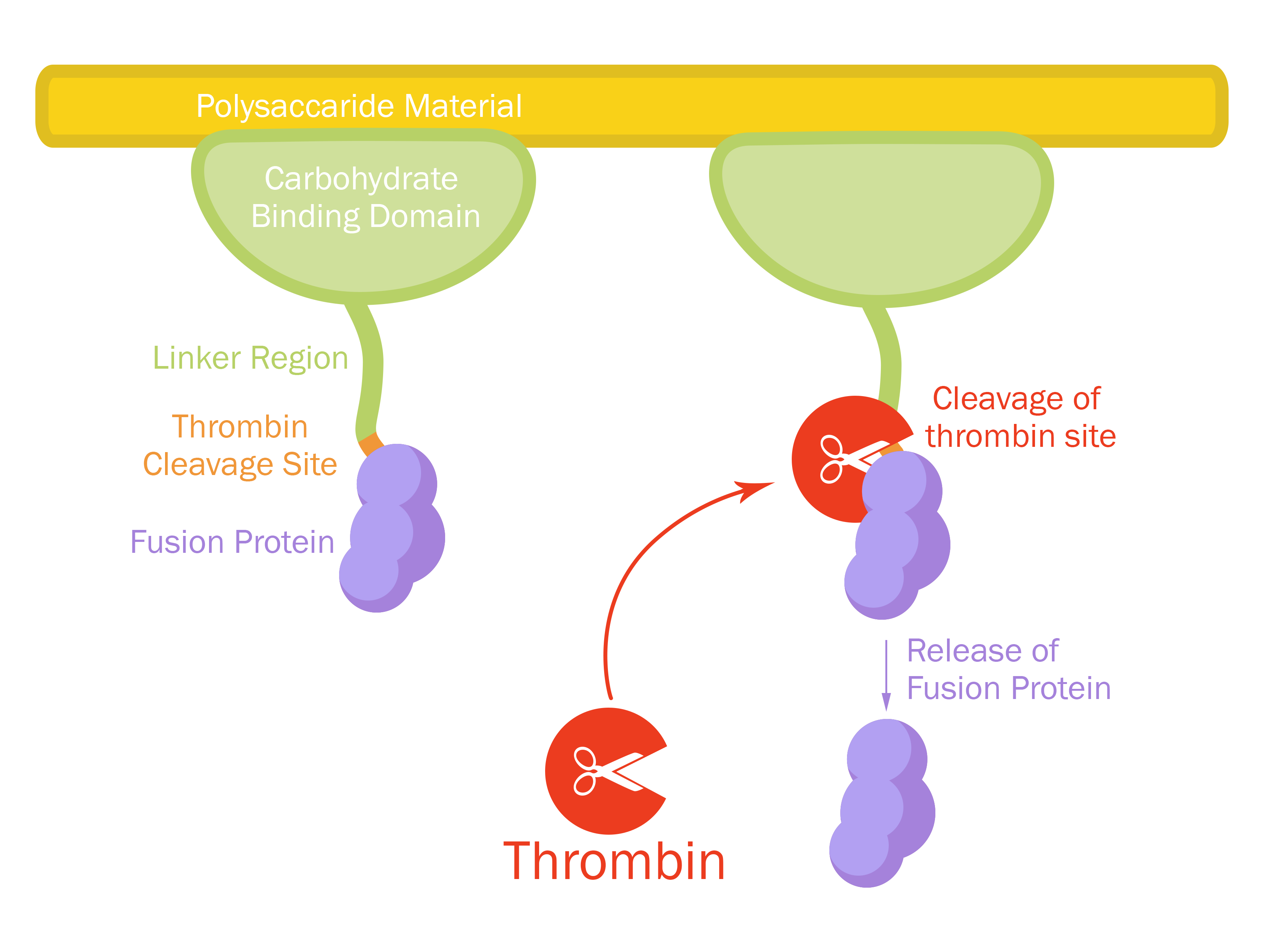
This part consists of a cellulose binding domain (CBD) from Clostridium thermocellum (C. thermocellum) cellulose scaffolding protein (CipA) and is a central part Clostridium thermocellum's cellusome. The CBD was fused to sfGFP in this part to easily track the binding capacities and to test our release mechanism. The CBD-sfGFP were fused using a flexible GS-linker (-GGGGSGGGGS-). A thrombin cleavage site (-LVPRGS-) was added to the end of the linker and its breakage will leave a glycine and serine attached to the N-terminal of the sfGFP fusion protein.
Assembly compabilities
An internal BamHI recognition sequence (RS) has been added to enable changeable fusion proteins. BamHI was chosen because its RS codes for glycine and serine, fitting it to the end of the thrombin site. It is also cost-effective enzyme and is unaffected by methylated DNA.
This part can be used to track purification, measure CBD binding ability and report cleavage at the thrombin site.
CBDcipA crystal structure
Important molecular faces
CBDcipA is composed of a nine-stranded beta sandwich with a jelly roll topology and binds a calcium ion. It further contains conserved residues exposed on the surface which map into two clear surfaces on each side of the molecule. One of faces mainly contains planar strips of aromatic and polar residues which may be the cellulose binding part. Further aspect are unknown and unique with this CBD such as the other conserved residues which are contained in a groove.
The choice of cellulose binding domain
iGEM Linköping 2019 choose CBDcipA due to many other iGEM teams exploring the possibilities of this domain. Our basic design was influenced by iGEM14 Imperial, iGEM15 Edinburgh and iGEM18 Ecuador. Purification and where to place the fusion protein (N- or C-terminal) was determined by studying the former projects. CBDcipA also originates from a thermophilic bacteria which further increases the domains applications.
Expression system
The part has a very strong expression with a T7-RNA-polymerase promotor (BBa_I719005) as well as a 5'-UTR (BBa_K1758100) region which has been shown to further increase expression in E. coli (BBa_K1758106), ([http://www.ncbi.nlm.nih.gov/pubmed/2676996 Olins et al. 1989]), ([http://www.ncbi.nlm.nih.gov/pubmed/23927491 Takahashi et al. 2013]). Both this part and the part were sfGFP was changed for AsPink (BBa_K3182000) showed great expression.

Usage and Biology
AsPink bound to cellulose bandage
Epiprotect (microbial cellulose bandage, S2Medical) was incubated in AsPink lysate from BL21 (DE3) Gold for 30 min. Epiprotect was later washed with 70% ethanol three times. To the left is AsPink incubated Epiprotect and to the right is plain Epiprotect.

Thrombin Cleavage
In figure 9 bacterial lysate of sonicated E. coli BL21 was incubated with cellulose (Whatman CF-11 Fibrous Medium Cellulose Powder) in 1h on an end-to-end rotator at R.T. The cellulose was saturated with AsPink lysate. The samples were afterwards centriuged at 3000 g in 1 min and the supernatant was removed. In all samples cleavage buffer (20 mM Tris-HCl pH 8.4, 150 mM NaCl and 2.5 mM CaCl₂) was added and three different dilutions of thrombin (Novagen, #69671) was added. The dilutions were 0,02, 9,94 and 0,08 units (U). One unit refers to the amount of thrombin needed to cleave 1 mg of protein for 16h at R.T in a 200 µl reaction containing the cleavage buffer. The samples were put on an end-to-end rotator over-night (ca 16h) and left at R.T. The control with only cleavage buffer has a transparent supernatant and a pink pellet of cellulose, the most diluted (0,02 U) has a slightly pinker supernatant and less pink pellet however the 0,04 U and 0,08 U dilutions has a pinker supernatant and paler pellet with 0,08 U dilution being the sample with most cleaved AsPink due to the more intense supernatant.





