Difference between revisions of "Part:BBa K2300003"
Ari edmonds (Talk | contribs) (→References) |
Ari edmonds (Talk | contribs) (→Part Verification) |
||
| Line 17: | Line 17: | ||
===Part Verification=== | ===Part Verification=== | ||
| + | |||
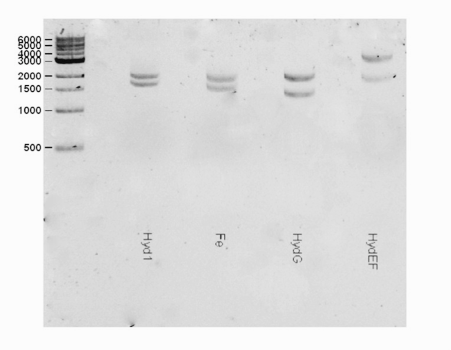
| + | <html><center><img src="https://static.igem.org/mediawiki/2016/b/bd/T--Macquarie_Australia--HYDShowGel.png" " width="50%" height="35%"></center> </html> | ||
| + | <b>Fig 1.</b> A gel consisting of EcorI/PstI for the hydEF part. The expected band for the hydEF (3611 bp) part was observed. | ||
===Protein information=== | ===Protein information=== | ||
Revision as of 08:32, 30 October 2017
HydEF (Updated 2017)
The original HydEF page (BBa_K1998012) had the wrong sequence so we have created an updated page. The HydEF protein contains two unique domains that are homologous to two distinct prokaryotic proteins, HydE and HydF, which are found exclusively in organisms containing [FeFe] hydrogenase (Mulder et al., 2010). This part is the first gene of the hydrogenase maturation enzyme complex.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12INCOMPATIBLE WITH RFC[12]Illegal NheI site found at 1849
Illegal NheI site found at 2059
Illegal NheI site found at 2503
Illegal NotI site found at 253 - 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Unknown
- 1000INCOMPATIBLE WITH RFC[1000]Illegal BsaI site found at 2684
Illegal BsaI.rc site found at 304
Illegal BsaI.rc site found at 416
Biology & Literature
HydEF in the majority of [FeFe] containing organisms exists as two separately transcribed proteins (HydE and HydF). However in several algal species it the two genes are fused (Böck et al., 2006). HydE (along with HydG) is a radical SAM (S-adenosyl methionine) enzyme which binds [4Fe-4S] clusters in the biosynthesis of the H-cluster (Mulder et al., 2010). HydF contains an N-terminal GTPase domain which is responsible for the P loop motif and FeS binding (Mulder et al., 2010). Both these genes have been shown to be essential for the maturation of HydA in E. coli (King et al., 2006). This is due to their role in the creation of the H-cluster.
Part Verification
Protein information
HydEF
Mass: 121.95 kDa
Sequence:
MAHSLSAHSRQAGDRKLGAGAASSRPSCPSRRIVRVAAHASASKATPDVPVDDLPPAHARAAVAAANRRARAMASAEAAAETLGDFLGLGKGGLSP
GATANLDREQVLGVLEAVWRRGDLNLERALYSHANAVTNKYCGGGVYYRGLVEFSNICQNDCSYCGIRNNQKEVWRYTMPVEEVVEVAKWALENGI
RNIMLQGGELKTEQRLAYLEACVRAIREETTQLDLEMRARAASTTTAEAAASAQADAEAKRGEPELGVVVSLSVGELPMEQYERLFRAGARRYLIRIET
SNPDLYAALHPEPMSWHARVECLRNLKKAGYMLGTGVMVGLPGQTLHDLAGDVMFFRDIKADMIGMGPFITQPGTPATDKWTALYPNANKNSHMK
SMFDLTTAMNALVRITMGNVNISATTALQAIIPTGREIALERGANVVMPILTPTQYRESYQLYEGKPCITDTAVQCRRCLDMRLHSVGKTSAAGVWGDPA
SFLHPIVGVPVPHDLSSPALAAAASADFHEVGAGPWNPIRLERLVEVPDRYPDPDNHGRKKAGAGKGGKAHDSHDDGDHDDHHHHHGAAPAGAAA
GKGTGAAAIGGGAGASRQRVAGAAAASARLCAGARRAGRVVASPLRPAAACRGVAVKAAAAAAGEDAGAGTSGVGSNIVTSPGIASTTAHGVPRINI
GVFGVMNAGKSTLVNALAQQEACIVDSTPGTTADVKTVLLELHALGPAKLLDTAGLDEVGGLGDKKRRKALNTLKECDVAVLVVDTDTAAAAIKSGRLA
EALEWESKVMEQAHKYNVSPVLLLNVKSRGLPEAQAASMLEAVAGMLDPSKQIPRMSLDLASTPLHERSTITSAFVKEGAVRSSRYGAPLPGCLPRW
SLGRNARLLMVIPMDAETPGGRLLRPQAQVMEEAIRHWATVLSVRLDLDAARGKLGPEACEMERQRFDGVIAMMERNDGPTLVVTDSQAIDVVHPW
TLDRSSGRPLVPITTFSIAMAYQQNGGRLDPFVEGLEALETLQDGDRVLISEACNHNRITSACNDIGMVQIPNKLEAALGGKKLQIEHAFGREFPELESG
GMDGLKLAIHCGGCMIDAQKMQQRMKDLHEAGVPVTNYGVFFSWAAWPDALRRALEPWGVEPPVGTPATPAAAPATAASGV
References
Böck, A., King, P.W., Blokesch, M. and Posewitz, M.C., 2006. Maturation of hydrogenases. Advances in Microbial Physiology, 51, pp.1-225.
King, P.W., Posewitz, M.C., Ghirardi, M.L. and Seibert, M., 2006. Functional studies of [FeFe] hydrogenase maturation in an Escherichia coli biosynthetic system. Journal of bacteriology, 188(6), pp.2163-2172.
Mulder, D.W., Shepard, E.M., Meuser, J.E., Joshi, N., King, P.W., Posewitz, M.C., Broderick, J.B. and Peters, J.W., 2011. Insights into [FeFe]-hydrogenase structure, mechanism, and maturation. Structure, 19(8), pp.1038-1052.
Posewitz, M.C., King, P.W., Smolinski, S.L., Zhang, L., Seibert, M. and Ghirardi, M.L., 2004. Discovery of two novel radical S-adenosylmethionine proteins required for the assembly of an active [Fe] hydrogenase. Journal of Biological Chemistry, 279(24), pp.25711-25720.
Vancouver
