Difference between revisions of "Part:BBa K592009:Experience"
MikeFerguson (Talk | contribs) (→User Reviews) |
MikeFerguson (Talk | contribs) (→User Reviews) |
||
| Line 19: | Line 19: | ||
<!-- End of the user review template --> | <!-- End of the user review template --> | ||
<!-- DON'T DELETE --><partinfo>BBa_K592009 EndReviews</partinfo> | <!-- DON'T DELETE --><partinfo>BBa_K592009 EndReviews</partinfo> | ||
| − | + | ||
{|width='80%' style='border:1px solid gray' | {|width='80%' style='border:1px solid gray' | ||
|- | |- | ||
| Line 36: | Line 36: | ||
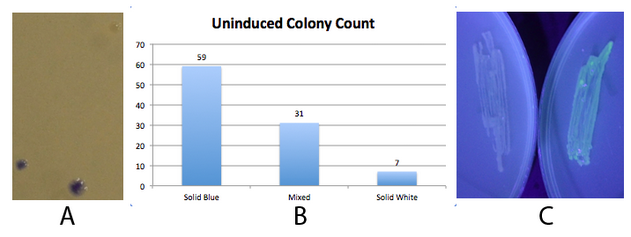
NEB 10-beta E. coli, which lack the native fim switch (fimS) and known fim recombinases, were co-transformed with K1077007(amilCP j23100 fim switch) or K1077003(GFP j23100 fim switch), and K1077002 (aTc inducible fimE, HSL inducible hbiF) and plated on to LB plates with or without inducer. A) Close up of 3 colonies on a plate containing K1077007 and K1077002 co transformants. Three distinct phenotypes were observed: Solid Blue colonies(bottom left), Mixed colonies that had distinct white and blue regions(bottom middle), and Solid White colonies(top right). Therefore, the uninduced fim transcriptor in the context of K1077002 is subject to leaky fimE and/or hbiF activity. B) Quantification of the phenotypes observed on the plate in A. C) When induced with 4.32µM aTc and grown overnight (left) , no GFP is produced as expected due to induced fimE turning the switch OFF. When induced with 1µM HSL and grown overnight (right), GFP is produced as expected due to induced hbiF turning the switch ON. | NEB 10-beta E. coli, which lack the native fim switch (fimS) and known fim recombinases, were co-transformed with K1077007(amilCP j23100 fim switch) or K1077003(GFP j23100 fim switch), and K1077002 (aTc inducible fimE, HSL inducible hbiF) and plated on to LB plates with or without inducer. A) Close up of 3 colonies on a plate containing K1077007 and K1077002 co transformants. Three distinct phenotypes were observed: Solid Blue colonies(bottom left), Mixed colonies that had distinct white and blue regions(bottom middle), and Solid White colonies(top right). Therefore, the uninduced fim transcriptor in the context of K1077002 is subject to leaky fimE and/or hbiF activity. B) Quantification of the phenotypes observed on the plate in A. C) When induced with 4.32µM aTc and grown overnight (left) , no GFP is produced as expected due to induced fimE turning the switch OFF. When induced with 1µM HSL and grown overnight (right), GFP is produced as expected due to induced hbiF turning the switch ON. | ||
| − | + | ||
{|width='80%' style='border:1px solid gray' | {|width='80%' style='border:1px solid gray' | ||
Revision as of 16:28, 29 September 2013
This experience page is provided so that any user may enter their experience using this part.
Please enter
how you used this part and how it worked out.
Applications of BBa_K592009
User Reviews
UNIQ0c913c96fbabcd0e-partinfo-00000000-QINU UNIQ0c913c96fbabcd0e-partinfo-00000001-QINU
|
•••••
iGEM Michigan 2013 |
The 2013 Michigan igem team successfully used this part to produce amilCP.
Fig 2. - An inducible fim transcriptor system changes states and produces protein output
NEB 10-beta E. coli, which lack the native fim switch (fimS) and known fim recombinases, were co-transformed with K1077007(amilCP j23100 fim switch) or K1077003(GFP j23100 fim switch), and K1077002 (aTc inducible fimE, HSL inducible hbiF) and plated on to LB plates with or without inducer. A) Close up of 3 colonies on a plate containing K1077007 and K1077002 co transformants. Three distinct phenotypes were observed: Solid Blue colonies(bottom left), Mixed colonies that had distinct white and blue regions(bottom middle), and Solid White colonies(top right). Therefore, the uninduced fim transcriptor in the context of K1077002 is subject to leaky fimE and/or hbiF activity. B) Quantification of the phenotypes observed on the plate in A. C) When induced with 4.32µM aTc and grown overnight (left) , no GFP is produced as expected due to induced fimE turning the switch OFF. When induced with 1µM HSL and grown overnight (right), GFP is produced as expected due to induced hbiF turning the switch ON.
|
•••••
Immudzen |
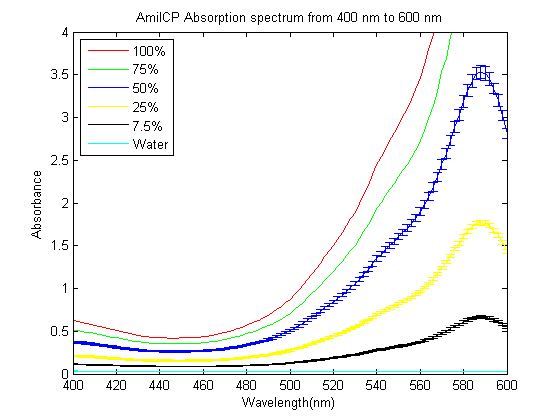
As part of the 2013 CU Boulder project we worked on separating RFP from AmilCP and we measured the spectrum of AmilCP from 400 nm to 600 nm. We also found that when running on an agarose gel RFP will run down on the gel while AmilCP runs up on the gel. |
|
•••••
iGEM 2013 Hong_Kong_HKUST Trevor Y.H. HO |
Error creating thumbnail: Invalid thumbnail parameters This part was employed in testing our Gibson Assembly protocol. In the experiment design, the mRFP coding DNA sequence in J04450 was replaced with that of amilCP. The resulting plate from transformation of the post-reaction mixture is shown. The color of the blue chromoprotein was clearly distinguished from that of mRFP, which aided in my evaluation of the performance of Gibson Assembly. |
|
BBa_K592009 iGEM Groningen 2012 |
Our team has managed to couple this biobrick part with our promoters: alsT, fnr, and sboA. The cloning was done in BBa_K818000 (plasmid backbone for B. subtilis, engineered by team Groningen 2012), to allow color expression in B. subtilis. We utilized a strong RBS BBa_B0034 for pigment expression in E. coli and B. subtilis. The purple/blue colour was strongly visible in E.coli without any induction, while the expression in B. subtilis was more subtle (B. subtilis colony looks slightly blue on the plate agar). After induction of the promoter before AmilCP, B. subtilis also turned clearly purple/blue. Please have a look at the page of BBa_K818400 for more information.
|
|
|
iGEM Team Uppsala University 2012 We have observed that expression of amilCP confers a noticeable fitness cost in E coli. This is surprising, given that such behaviour is not seen in other homologous chromoproteins and fluorescent proteins. We thus recommend using the new aeBlue (BBa_K864401) chromoprotein for blue color expression, which also has a more clear blue color.
|