Difference between revisions of "Part:BBa K638402"
| Line 5: | Line 5: | ||
This TorA leader sequence variant has had been successfully used to export GFP to the periplasm of E.coli as described [http://www.ncbi.nlm.nih.gov/pubmed/11123687 here]. | This TorA leader sequence variant has had been successfully used to export GFP to the periplasm of E.coli as described [http://www.ncbi.nlm.nih.gov/pubmed/11123687 here]. | ||
| + | |||
| + | <!-- --> | ||
| + | <span class='h3bb'>Sequence and Features</span> | ||
| + | <partinfo>BBa_K638402 SequenceAndFeatures</partinfo> | ||
===Usage and Biology=== | ===Usage and Biology=== | ||
| − | ===Improving efficiency of export=== | + | ===Improving efficiency of export (Literature Data)=== |
| − | [http://www.ncbi.nlm.nih.gov/pubmed/11123687 Thomas ''et al''] described use of the part by fusing it to GFP. | + | [http://www.ncbi.nlm.nih.gov/pubmed/11123687 Thomas ''et al''] described use of the part by fusing it to GFP. Efficiency of export is improved by removing induction from medium. |
| − | [[Image:TorA-GFP in periplasm.gif | thumb | | + | [[Image:TorA-GFP in periplasm.gif | thumb | 500px | Visualization of the periplasmic export of TorA–GFP by fluorescence microscopy. Wild-type MC4100AR cells (top) and the ΔtatC strain (bottom) containing the torA–GFP fusion plasmid pJDT1 were grown in the presence of arabinose, then resuspended in the absence of arabinose and incubated for 2.5 h. ''Image taken from [http://www.ncbi.nlm.nih.gov/pubmed/11123687 Thomas et al, 2001] ]] |
[[Image:TorA Export Fluorescence graph Thomas.gif | thumb | 300px|left | Values are given (in arbitrary units) for the fluorescence of cytoplasmic, periplasmic and membrane fractions (hatched, white and black bars respectively) of wild-type and two TAT export mutant strains (ΔtatB andΔtatC). Approx 50% of protein is exported in the WT strain.]] | [[Image:TorA Export Fluorescence graph Thomas.gif | thumb | 300px|left | Values are given (in arbitrary units) for the fluorescence of cytoplasmic, periplasmic and membrane fractions (hatched, white and black bars respectively) of wild-type and two TAT export mutant strains (ΔtatB andΔtatC). Approx 50% of protein is exported in the WT strain.]] | ||
| − | |||
| − | + | ||
| − | + | [http://www.ncbi.nlm.nih.gov/pubmed/12711311 Barrett ''et al''] characterised improved export by altering growth conditions and upregulating TAT export machinery , using the same TorA-GFP fusion. | |
| − | + | [[ | |
| + | |||
| + | As the below graph shows that the majority of GFP is membrane associated (and therefore does not fluoresce) - Barrett ''et al'' calculated that only ~4% of export tagged GFP reached the periplasm in an active form. They greatly improved export efficiency by upregulating production of | ||
Revision as of 14:44, 21 September 2011
TorA tag variant
This is an improved version of part BBa_K233307 designed to allow comparison with measurements of functionality in the literature, and to make it easier to synthesise.
This TorA leader sequence variant has had been successfully used to export GFP to the periplasm of E.coli as described [http://www.ncbi.nlm.nih.gov/pubmed/11123687 here].
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
Usage and Biology
Improving efficiency of export (Literature Data)
[http://www.ncbi.nlm.nih.gov/pubmed/11123687 Thomas et al] described use of the part by fusing it to GFP. Efficiency of export is improved by removing induction from medium.
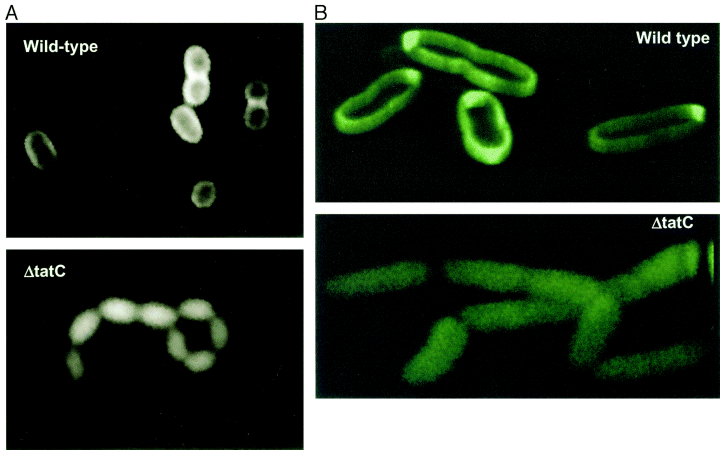
[http://www.ncbi.nlm.nih.gov/pubmed/12711311 Barrett et al] characterised improved export by altering growth conditions and upregulating TAT export machinery , using the same TorA-GFP fusion.
[[
As the below graph shows that the majority of GFP is membrane associated (and therefore does not fluoresce) - Barrett et al calculated that only ~4% of export tagged GFP reached the periplasm in an active form. They greatly improved export efficiency by upregulating production of
Producing BBa_k638402 from primer synthesis
We assembled the basic part by using 2 synthetic oligonucleotides (TorA-FWD and TorA-REV in the table and diagram below). These oligonucleotides have a 20bp overlap and can be used to generate the entire TorA tag by PCR.
| Name | Sequence | Tm |
|---|---|---|
| TorA FWD | ATGGCGAACAACGACTTATTTCAGGCTTCTCGGCGTCGCTTTCTGGCGCAGCTGGGCGGATTAACGGTGGCGGGT | 70.98°C |
| TorA REV | TGCGGCTTGTGCTGCCGTCGCTCTGCGAGGAGTCAACAGCGACGGGCCCAACATACCCGCCACCGTTAATCCGCC | 70.98°C |
Thermocycler profile: 10 cycles: Melt 95°C 10 sec / Anneal 65°C 30 sec / Extend 72°C 20 sec
Insertion of TorA tag into a construct
Cambridge 2011 created scar-free fusions of this export tag to our protein of interest by [http://www.cambridgeigem.org/RFC57.pdf Gibson Assembly]. Alternatively, Biobrick prefixes and suffixes could be added to this oligonucleotides (or added seperately) enabling the creation of in-frame fusions by assembly techniques such as BBF RFC 23 or 25.
The diagram below shows the 4 oligonucleotides needed to add a TorA tag by [http://www.cambridgeigem.org/RFC57.pdf Gibson Assembly]. TorA-FWD and TorA-REV generate the tag as described above. Gibson-primer-FWD and Gibson-primer-REV anneal to the template construct either side of where the TorA tag is to be placed. Gibson-primer-FWD has a tail which is the last 40bp of the TorA tag, and Gibson-primer-REV has a tail which is the reverse compliment of first 40bp of the TorA tag. These 40bp of overlap are what is required for [http://www.cambridgeigem.org/RFC57.pdf Gibson Assembly].


