Difference between revisions of "Part:BBa K3593000"
(→Modeling) |
|||
| (17 intermediate revisions by 2 users not shown) | |||
| Line 5: | Line 5: | ||
Aptamer of alpha amatoxin found by iterative SELEX process. It forms special secondary structure and bind with amatoxin specifically, which is characterized by q PCR | Aptamer of alpha amatoxin found by iterative SELEX process. It forms special secondary structure and bind with amatoxin specifically, which is characterized by q PCR | ||
=Background= | =Background= | ||
| + | Amatoxins are chemicals present inside the genus Amanita and caused about 90% of mushroom poisoning. Being able to detect it before eating or in the field could possibly make a great decrease in people and animals who died because of poisonous mushrooms. Also being able to detect it in hospital can greatly help doctors in mushroom areas get correct information and do effective diagnosis to save the patient. | ||
| + | |||
| + | <div><ul> | ||
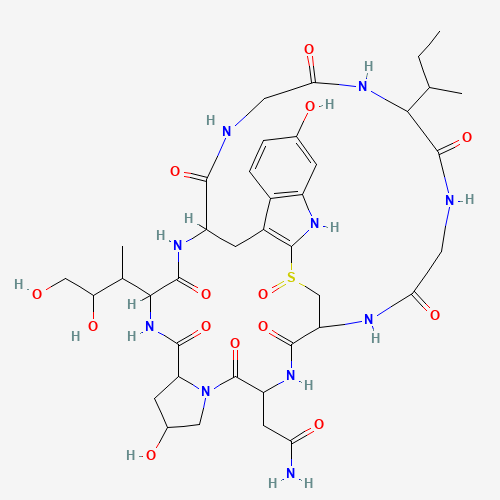
| + | <li style="display: inline-block;"> [[File:alpha-Amanitine_500.png|300px|thumb|none|Model-Fig-1. <span>ɑ</span>-Amanitin 2D structure]] </li> | ||
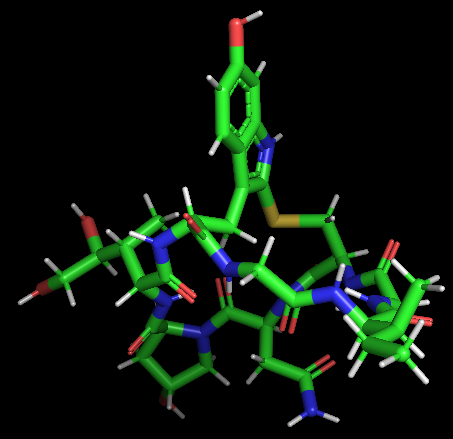
| + | <li style="display: inline-block;"> [[File:Amanitin-3D.png|300px|thumb|none|Model-Fig-2. <span>ɑ</span>-Amanitin 3D structure]] </li> | ||
| + | </ul></div> | ||
| + | |||
| + | <span>ɑ</span>-Amanitin is a highly modified bicyclic octapeptide consisting of an outer and an inner loop. The outer loop is formed by peptide bonds between a carboxyl terminus of an amino acid to the subsequent amino terminus of the next residue. The inner loop is closed by a tryptathionine linkage between 6-hydroxy-tryptophan and cysteine. In addition, | ||
| + | <span>ɑ</span>-Amanitin has modified amino acid side chains (2S,3R,4R)-4,5-dihydroxy-isoleucine, trans-4-hydroxy-proline, which gives its high affinity for RNA polymerase II and III. The 2D and 3D structures are shown in Model-Fig-1 and Model-Fig-2 respectively. | ||
| + | |||
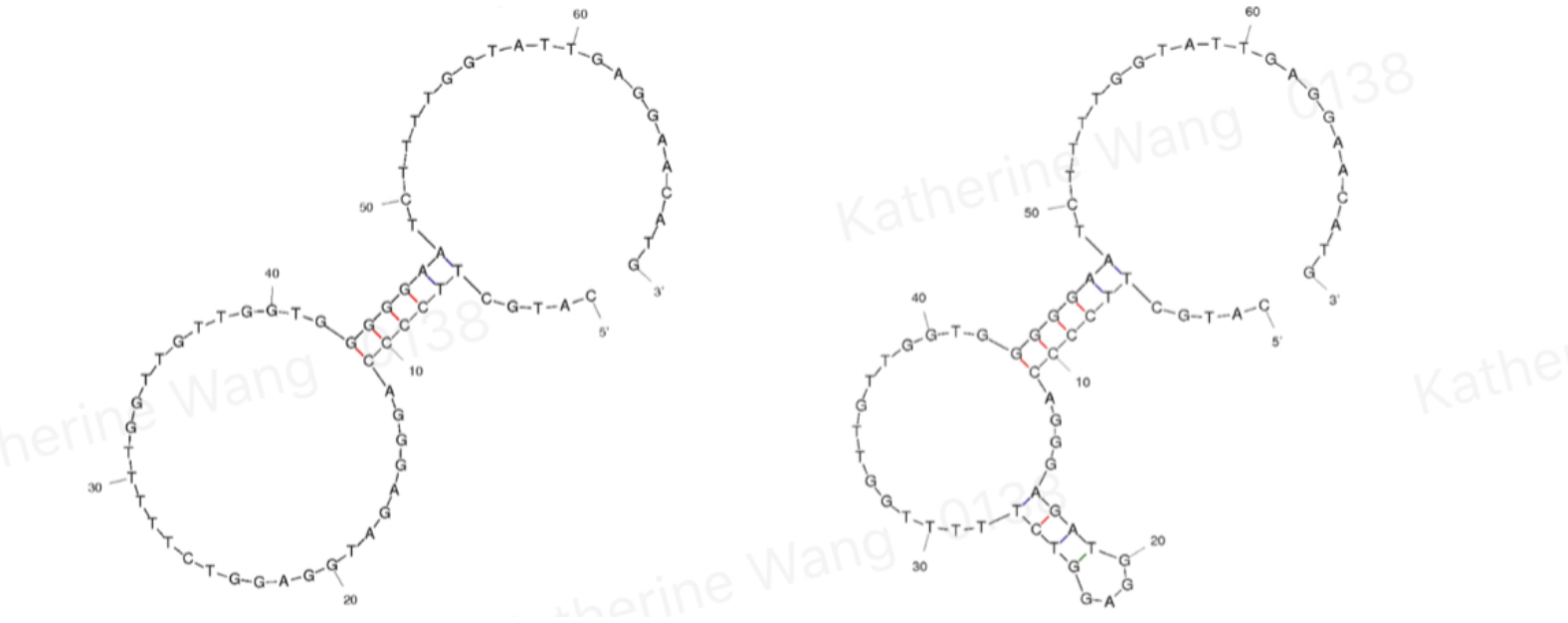
| + | Aptamers are oligonucleotides that form secondary structures, giving them the ability to bind targeted molecules, including ions or small molecules, and, in our case, amanitin. BBa_K3593000 is an aptamer of α-amanitin named Best 1. It comes from one previous literature[1]. Its binding is tested through qPCR. | ||
| + | [[File:T--GreatBay_SCIE--Fig.1 Structure of aptamer Best 1 and Best 2.png|600px|thumb|center]] | ||
| + | <p class="figure-description"><b><center>Fig.1 Structure of aptamer Best 1 and Best 2</center></b></p> | ||
| + | |||
=Sequence and features= | =Sequence and features= | ||
<partinfo>BBa_K3593000 SequenceAndFeatures</partinfo> | <partinfo>BBa_K3593000 SequenceAndFeatures</partinfo> | ||
=Characterisation of this part= | =Characterisation of this part= | ||
| − | == | + | ==Modeling== |
| + | (Section added by iGEM 2022 team IISER_Mohali) <br> | ||
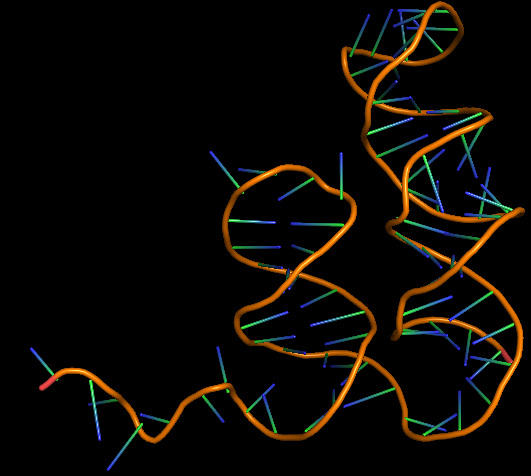
| + | <u>3D structure of the Aptamer</u><br> | ||
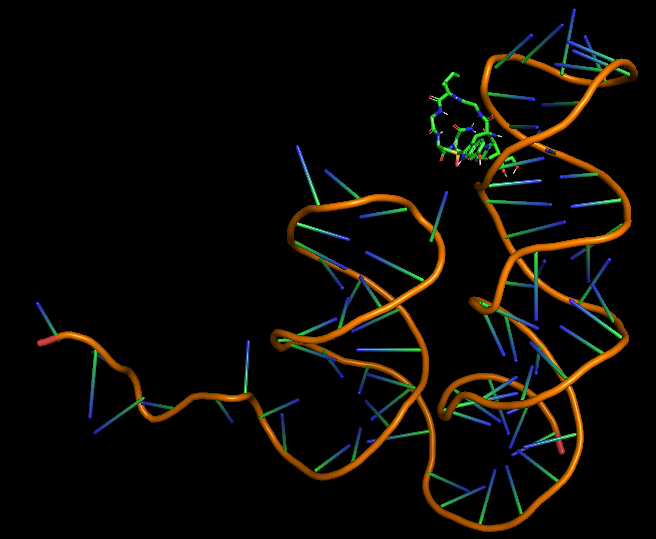
| + | [[File:Amanitin_aptamer3D.png|200px|thumb|center|Model-Fig-3. <span>ɑ</span>-Amanitin Aptamer most probable 3D structure]] | ||
| + | The 3D folded structure of the aptamer was generated using Xiao Lab 3dRNA/DNA - An RNA and DNA tertiary structure prediction method (http://biophy.hust.edu.cn/new/3dRNA/create). The input was the DNA aptamer sequence, molecule type DNA, Predict 2nd Structure: Yes, 2nd Structure Prediction Method: RNA Fold, Procedure Best, # of Predictions 5. No changes were made in Advanced options. Model-Fig-3 shows the most stable folded 3D structure. | ||
| + | |||
| + | <u>Molecular Docking</u><br> | ||
| + | <span>ɑ</span>-Amanitin was docked with it's aptamer using AutoDock Version 4.2. The main steps involed in the docking process: | ||
| + | <ol> | ||
| + | <li>Adding polar hydrogen and kollman charges to the receptor and saving it as pdbqt</li> | ||
| + | <li>Defining torsion root and torsion angles for ligand and saving it as pdbqt</li> | ||
| + | <li>Making grid box (including whole of the protein) and defining grid parameters for receptor using autogrid</li> | ||
| + | <li>Docking using Genetic algorithm (ga runs: 50, population size:300, evals: 250,000; generations: 27000)</li> | ||
| + | <li>Output: Lamarckian GA</li> | ||
| + | <li>Run using autodock</li> | ||
| + | <li>Analysis of results using the dlg output file</li> | ||
| + | </ol> | ||
| + | <div><ul> | ||
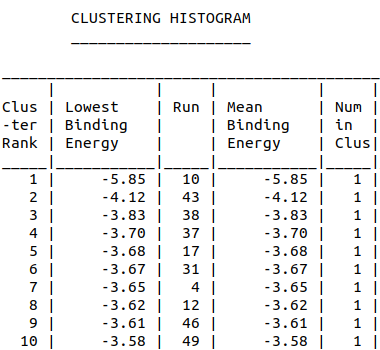
| + | <li style="display: inline-block;"> [[File:Amanitin_Dock-ClusteringHistogram.png|200px|thumb|none|Model-Table-1]] </li> | ||
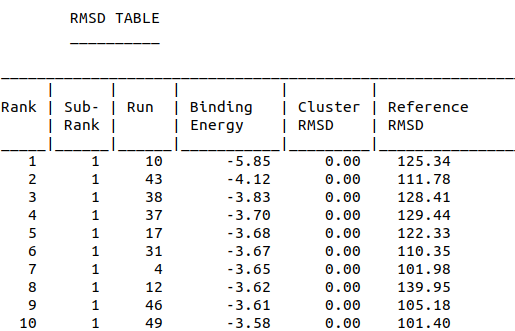
| + | <li style="display: inline-block;"> [[File:Amantinin_Dock-RMSDTable.png|200px|thumb|none|Model-Table-2]] </li> | ||
| + | <li style="display: inline-block;"> [[File:Amanatitin-Aptamer_Dock.png|200px|thumb|none|Model-Fig-4. Most probable docked configuration of<span>ɑ</span>-Amanitin and aptamer]] </li> | ||
| + | </ul></div> | ||
| + | |||
| + | Model-Table-1 shows the top 10 docked structures, while Model-Table-2 shows the RMSD scores for the top 10 docked structures. We see that the docked configuration corresponding to cluster 1, number 1 is the most probable one (least energy and highest RMSD score). This is shown in Model-Fig-4. | ||
| + | |||
==Experiment data== | ==Experiment data== | ||
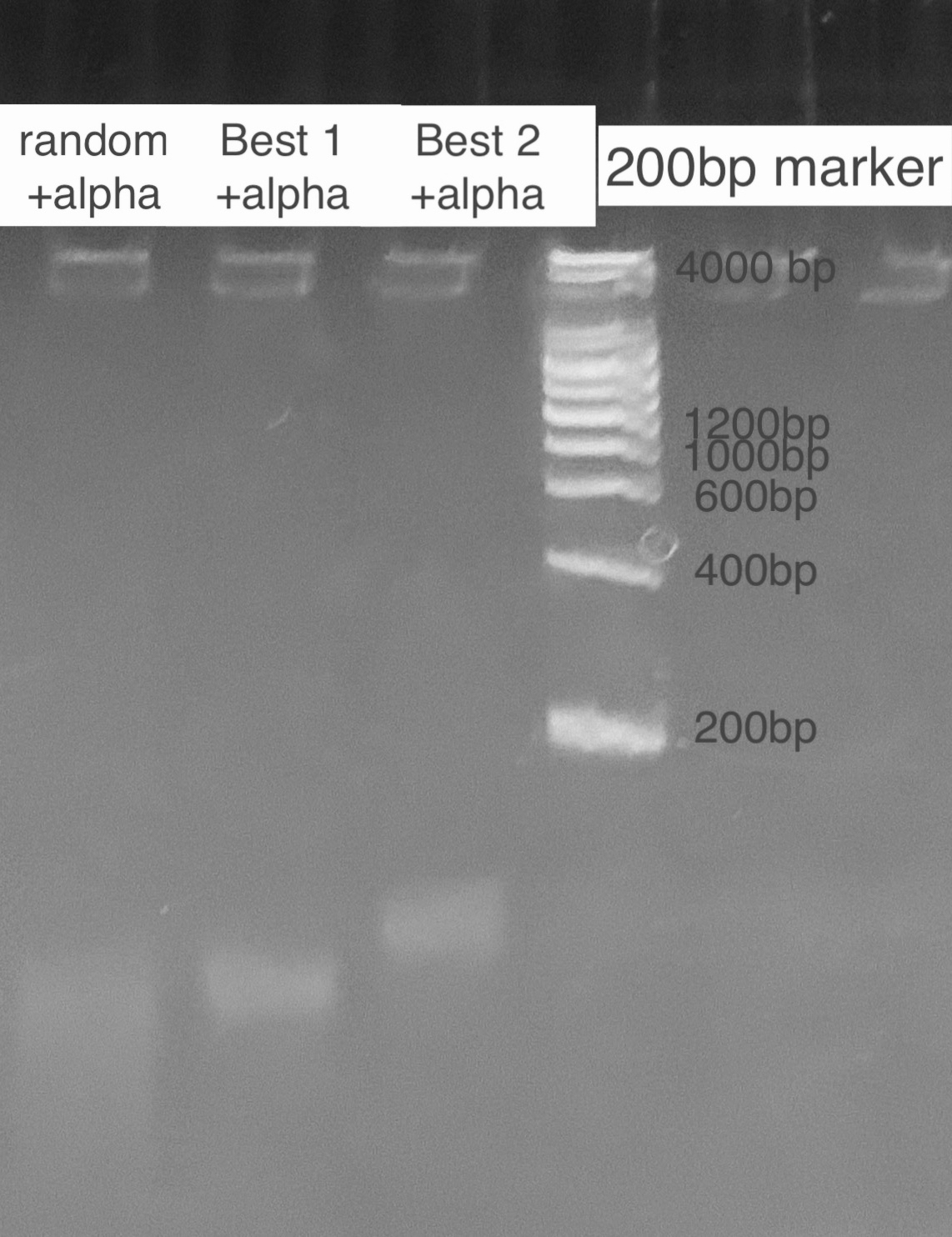
| − | = | + | According to the state of art in aptamer binding[3], we design an experiment based on the principle of Gel Shift(i.e lower rate of motion in electrophoresis when aptamer is bound to the toxin due to a conformational change). By mixing excess amatoxin determined by the calculation of dissociation constant and aptamer in certain reaction times, we expect to see 2 bands representing the bound and inbound aptamer respectively. For examination of specificity, we also mix a random ssDNA pool where we select aptamer from and compare the band with the correct aptamer. For a high resolution, we used the TBE PAGE gel for DNA, 12%. <br> |
| + | [[File:T--GreatBay_SCIE--Fig.2 Gel Shift of Best 1&Best 2.jpg|600px|thumb|center]] | ||
| + | <p class="figure-description"><b><center>Fig.2 Gel Shift of Best 1&Best 2</center></b></p> | ||
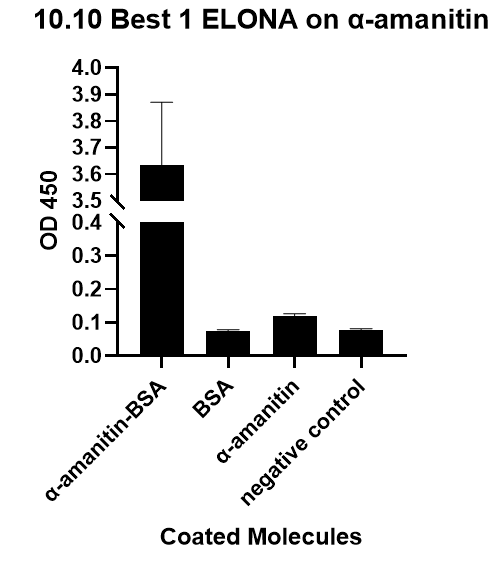
| + | We also verify its binding affinity via a method called ELONA(enzyme-linked oligonucleotide assay), a method | ||
| + | from another literature[2], with a similar principle to ELISA.<br> | ||
| + | [[File:T--GreatBay_SCIE--Fig.3 The ELONA on Best 1 aptamer.png|600px|thumb|center]] | ||
| + | <p class="figure-description"><b><center>Fig.3 The ELONA on Best 1 aptamer.</center></b></p> | ||
| − | < | + | =References= |
| + | 1.Muszyńska K, Ostrowska D, Bartnicki F, et al. Selection and analysis of a DNA aptamer binding α-amanitin from Amanita phalloides. Acta Biochim Pol. 2017;64(3):401-406. <br> | ||
| + | 2.Han Q, Xia X, Jing L, et al. Selection and characterization of DNA aptamer specially targeting α-amanitin in wild mushrooms. SDRP J Food Sci Technol. 2018;3(6):497-508. <br> | ||
| + | 3.Muszyńska K, Ostrowska D, Bartnicki F, et al. Selection and analysis of a DNA aptamer binding α-amanitin from Amanita phalloides. Acta Biochim Pol. 2017;64(3):401-406. doi:10.18388/abp.2017_1615 <br> | ||
| + | 4. https://en.wikipedia.org/wiki/%CE%91-Amanitin#Chemical_structure <br> | ||
| + | 5. Zhao, Y., et al., Automated and fast building of three-dimensional RNA structures. Scientific Reports, 2012. 2: p. 734. <br> | ||
| + | 6. Morris, G. M., Huey, R., Lindstrom, W., Sanner, M. F., Belew, R. K., Goodsell, D. S. and Olson, A. J. (2009) Autodock4 and AutoDockTools4: automated docking with selective receptor flexiblity. J. Computational Chemistry 2009, 16: 2785-91. | ||
Latest revision as of 11:15, 12 October 2022
ssDNA, aptamer for α-amanitin(Best 1)
Aptamer of alpha amatoxin found by iterative SELEX process. It forms special secondary structure and bind with amatoxin specifically, which is characterized by q PCR
Background
Amatoxins are chemicals present inside the genus Amanita and caused about 90% of mushroom poisoning. Being able to detect it before eating or in the field could possibly make a great decrease in people and animals who died because of poisonous mushrooms. Also being able to detect it in hospital can greatly help doctors in mushroom areas get correct information and do effective diagnosis to save the patient.
ɑ-Amanitin is a highly modified bicyclic octapeptide consisting of an outer and an inner loop. The outer loop is formed by peptide bonds between a carboxyl terminus of an amino acid to the subsequent amino terminus of the next residue. The inner loop is closed by a tryptathionine linkage between 6-hydroxy-tryptophan and cysteine. In addition, ɑ-Amanitin has modified amino acid side chains (2S,3R,4R)-4,5-dihydroxy-isoleucine, trans-4-hydroxy-proline, which gives its high affinity for RNA polymerase II and III. The 2D and 3D structures are shown in Model-Fig-1 and Model-Fig-2 respectively.
Aptamers are oligonucleotides that form secondary structures, giving them the ability to bind targeted molecules, including ions or small molecules, and, in our case, amanitin. BBa_K3593000 is an aptamer of α-amanitin named Best 1. It comes from one previous literature[1]. Its binding is tested through qPCR.
Sequence and features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
Characterisation of this part
Modeling
(Section added by iGEM 2022 team IISER_Mohali)
3D structure of the Aptamer
The 3D folded structure of the aptamer was generated using Xiao Lab 3dRNA/DNA - An RNA and DNA tertiary structure prediction method (http://biophy.hust.edu.cn/new/3dRNA/create). The input was the DNA aptamer sequence, molecule type DNA, Predict 2nd Structure: Yes, 2nd Structure Prediction Method: RNA Fold, Procedure Best, # of Predictions 5. No changes were made in Advanced options. Model-Fig-3 shows the most stable folded 3D structure.
Molecular Docking
ɑ-Amanitin was docked with it's aptamer using AutoDock Version 4.2. The main steps involed in the docking process:
- Adding polar hydrogen and kollman charges to the receptor and saving it as pdbqt
- Defining torsion root and torsion angles for ligand and saving it as pdbqt
- Making grid box (including whole of the protein) and defining grid parameters for receptor using autogrid
- Docking using Genetic algorithm (ga runs: 50, population size:300, evals: 250,000; generations: 27000)
- Output: Lamarckian GA
- Run using autodock
- Analysis of results using the dlg output file
Model-Table-1 shows the top 10 docked structures, while Model-Table-2 shows the RMSD scores for the top 10 docked structures. We see that the docked configuration corresponding to cluster 1, number 1 is the most probable one (least energy and highest RMSD score). This is shown in Model-Fig-4.
Experiment data
According to the state of art in aptamer binding[3], we design an experiment based on the principle of Gel Shift(i.e lower rate of motion in electrophoresis when aptamer is bound to the toxin due to a conformational change). By mixing excess amatoxin determined by the calculation of dissociation constant and aptamer in certain reaction times, we expect to see 2 bands representing the bound and inbound aptamer respectively. For examination of specificity, we also mix a random ssDNA pool where we select aptamer from and compare the band with the correct aptamer. For a high resolution, we used the TBE PAGE gel for DNA, 12%.
We also verify its binding affinity via a method called ELONA(enzyme-linked oligonucleotide assay), a method
from another literature[2], with a similar principle to ELISA.
References
1.Muszyńska K, Ostrowska D, Bartnicki F, et al. Selection and analysis of a DNA aptamer binding α-amanitin from Amanita phalloides. Acta Biochim Pol. 2017;64(3):401-406.
2.Han Q, Xia X, Jing L, et al. Selection and characterization of DNA aptamer specially targeting α-amanitin in wild mushrooms. SDRP J Food Sci Technol. 2018;3(6):497-508.
3.Muszyńska K, Ostrowska D, Bartnicki F, et al. Selection and analysis of a DNA aptamer binding α-amanitin from Amanita phalloides. Acta Biochim Pol. 2017;64(3):401-406. doi:10.18388/abp.2017_1615
4. https://en.wikipedia.org/wiki/%CE%91-Amanitin#Chemical_structure
5. Zhao, Y., et al., Automated and fast building of three-dimensional RNA structures. Scientific Reports, 2012. 2: p. 734.
6. Morris, G. M., Huey, R., Lindstrom, W., Sanner, M. F., Belew, R. K., Goodsell, D. S. and Olson, A. J. (2009) Autodock4 and AutoDockTools4: automated docking with selective receptor flexiblity. J. Computational Chemistry 2009, 16: 2785-91.