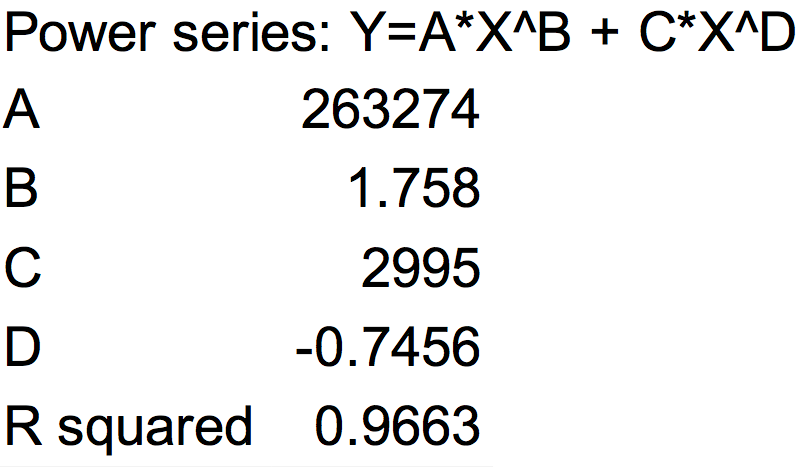Difference between revisions of "Part:BBa K2558001"
| (3 intermediate revisions by 2 users not shown) | |||
| Line 40: | Line 40: | ||
<br/>[2] Zeng, Weiqian, et al. "Rational design of an ultrasensitive quorum-sensing switch." Acs Synthetic Biology 6.8(2017). | <br/>[2] Zeng, Weiqian, et al. "Rational design of an ultrasensitive quorum-sensing switch." Acs Synthetic Biology 6.8(2017). | ||
| − | + | ||
===SJTU-BioX-Shanghai 2019's Characterization=== | ===SJTU-BioX-Shanghai 2019's Characterization=== | ||
This lux pR-HS promoter is put upstream of repL(BBa_K2996000) to form a composite part(BBa_K2996006) that will respond to AHL, together with a constant expression of LuxR. RepL is a P1 lytic replication element that can act on mini-F origin to promote plasmid replication. Thus we quantified the effect of lux pR-HS indirectly by measuring plasmid copy number through qPCR. We tried different AHL concentration from 0 to 10-6 M and measured copy number in the time course. Both absolute copy number and relevant copy number amplification compared to non-induction were calculated. The result confirmed the small leakage of lux pR-HS and it also showed obvious responding to AHL, with the maximum effect reached at a concentration of 10-7 M at 3.5h of induction. | This lux pR-HS promoter is put upstream of repL(BBa_K2996000) to form a composite part(BBa_K2996006) that will respond to AHL, together with a constant expression of LuxR. RepL is a P1 lytic replication element that can act on mini-F origin to promote plasmid replication. Thus we quantified the effect of lux pR-HS indirectly by measuring plasmid copy number through qPCR. We tried different AHL concentration from 0 to 10-6 M and measured copy number in the time course. Both absolute copy number and relevant copy number amplification compared to non-induction were calculated. The result confirmed the small leakage of lux pR-HS and it also showed obvious responding to AHL, with the maximum effect reached at a concentration of 10-7 M at 3.5h of induction. | ||
| − | + | ||
| − | < | + | <center>{{#tag:html|<img style="max-width: 50%" src="https://2019.igem.org/wiki/images/3/31/T--SJTU-BioX-Shanghai--copy1.jpg" alt="" />}}</center> |
| + | <center><b>Figure 1.</b> <i>Change of absolute copy number under different induction concentrations </i> </center> | ||
| + | <center>{{#tag:html|<img style="max-width: 50%" src="https://2019.igem.org/wiki/images/e/e8/T--SJTU-BioX-Shanghai--copy2.jpg" alt="" />}}</center> | ||
| + | <center><b>Figure 1.</b> <i>Change of relativecopy number under different induction concentrations </i> </center> | ||
| + | |||
===Protocol=== | ===Protocol=== | ||
| Line 81: | Line 85: | ||
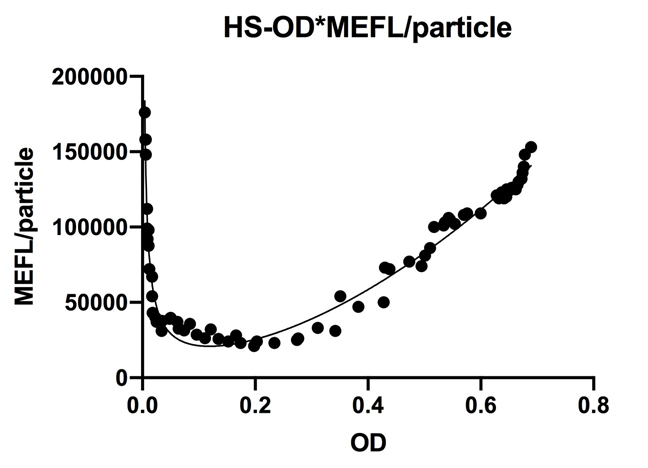
| − | <p>Fig.1 This figure shows luxpR-HS expression level induced by luxR-luxI box. The MEFL/particle changes as the OD goes up after dilution.</p> | + | <p>Fig.1 This figure shows luxpR-HS expression level induced by luxR-luxI box. The MEFL/particle changes as the OD goes up after dilution. Data points are shown in the figure for fitting analysis. Here gives the parameters of the fitting equation.</p> |
<p>At first the parental fluorescence and dilution effect keep fluorescence/OD600 at an extremely high level. Then the concentration of signal molecules remains low in the fresh culture while the bacteria are reproducing. In the next few hours the fluorescence/OD600 keeps going down. After the newly cultured bacteria grow to a higher concentration, luxpR-HS is activated and fluorescence/OD600 rises again as Fig.1 shows. Expression properties of luxpR-HS is predictable according to the fitting analysis results.</p> | <p>At first the parental fluorescence and dilution effect keep fluorescence/OD600 at an extremely high level. Then the concentration of signal molecules remains low in the fresh culture while the bacteria are reproducing. In the next few hours the fluorescence/OD600 keeps going down. After the newly cultured bacteria grow to a higher concentration, luxpR-HS is activated and fluorescence/OD600 rises again as Fig.1 shows. Expression properties of luxpR-HS is predictable according to the fitting analysis results.</p> | ||
Latest revision as of 03:58, 22 October 2019
lux pR-HS
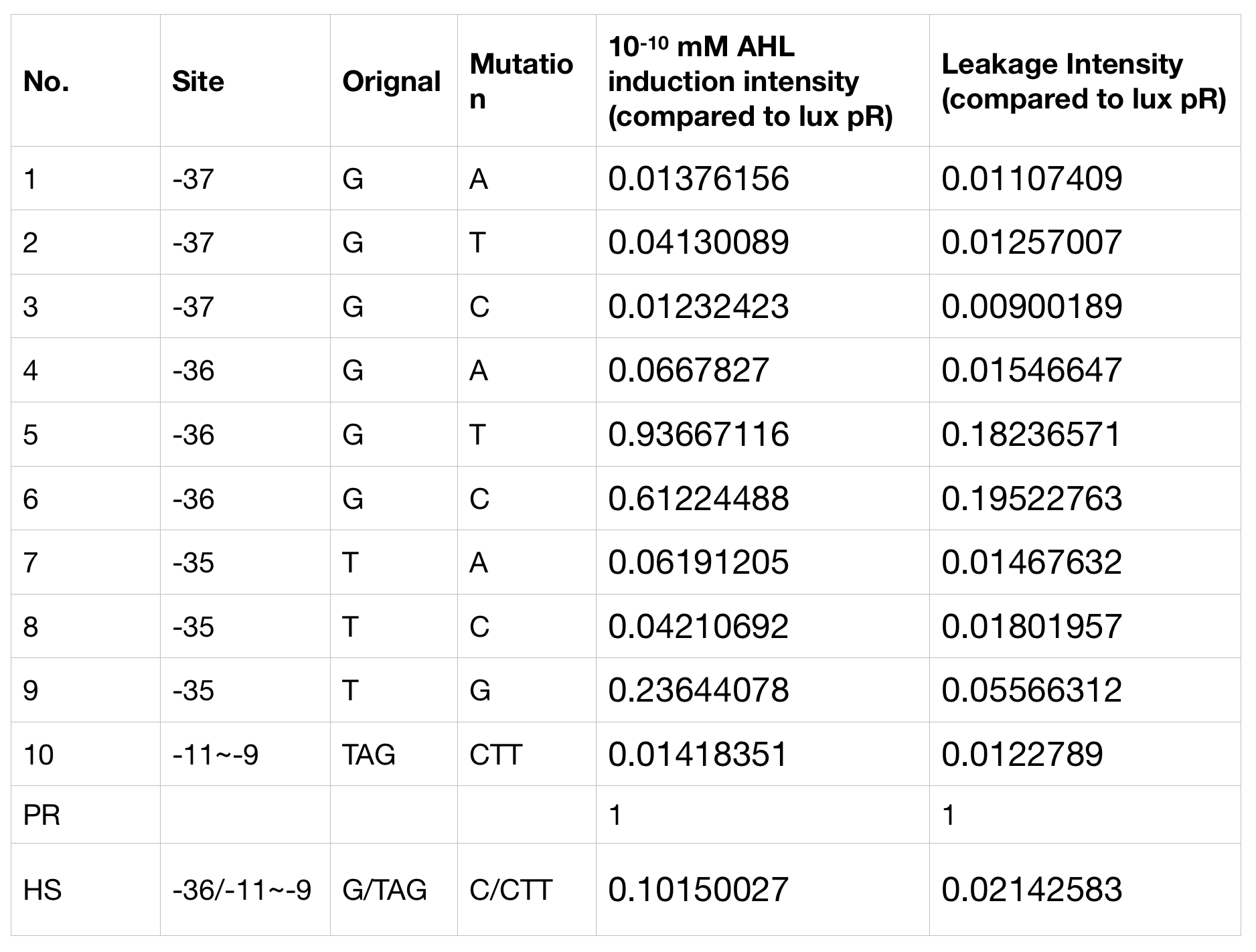
This part is derived from lux pR. They are similar except for one point mutation in the luxR binding site (G19T) and another three in the -10 region of the promotor (TAG44CTT). The function remains the same, the mutant lux pR promotor is still inducible by LuxR/AHL complex. However, they do differ in gene expression strength, leakage and other parameters.
Usage and Biology
With the hope to find an optimal promotor for our NEON system, we designed 9 mutations on sites -35, -36, -37 near the luxR binding site and tested another on -10 site that TUST 2017 reported to show decreased leakage. We conducted experiments to evaluate these lux pR mutants’ reaction to AHL stimulation and their leakage level. The test devices we designed include a constantly expressed luxR and a lux pR (or mutant) driven sfGFP (like BBa_K2558211 with original lux pR promotor, and BBa_K2558212 with lux pR-HS promotor).
Results
We conducted the experiment first with 9 mutations on sites -35, -36, -37 near the luxR binding and another on -10 site that TUST 2017 reported to show decreased leakage. We transferred the plasmids with lux pR driven sfGFP into E. coli DH5α. In theory, AHL can induce gene expression by activating lux pR promotor. We measured the value of Fluorescence/OD without AHL stimulation by microplate reader to show the leakages of different lux pR mutants, as lux pR promotor is not supposed to be activated without AHL. Meanwhile, we also regard the value of Fluorescence/OD with 10^-10 M AHL stimulation to observe the sensitivity and expression intensity of different lux pR mutants. Experiment data is shown in the figures below. (Figure.1)
-
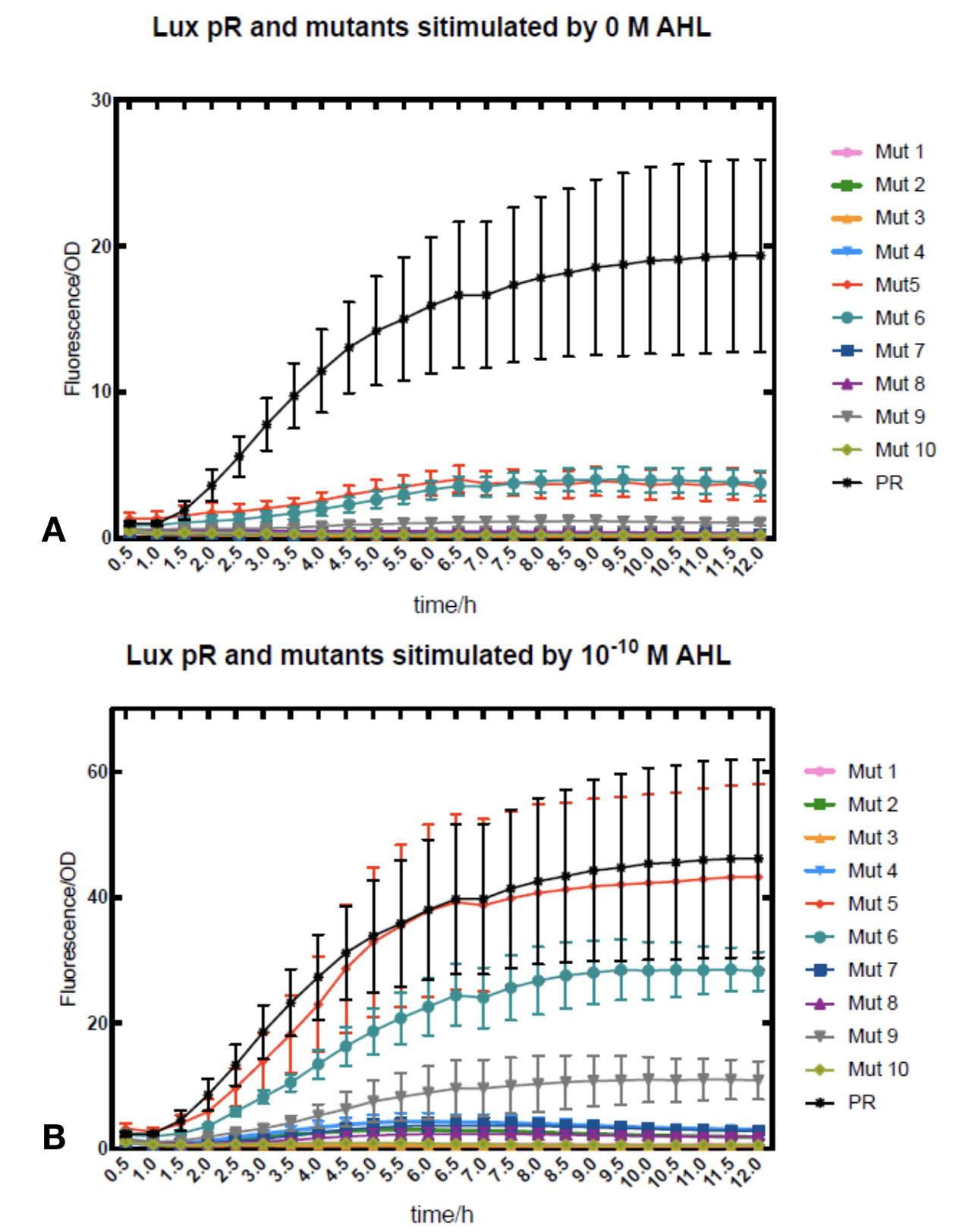 Figure.1. Leakage and 10^-10 mM AHL stimulation of lux pR promotor and its mutations. The fluorescence strengths and OD values are measured by microplate reader respectively at 510 nm and 600 nm wavelength. Plasmids with lux pR promotor and its mutants are transferred into E.coli DH5α. A The bacteria were treated without AHL inducing. High (Fluorescence/OD) ratio indicates strong leakage of promotor. Among all the strains, lux pR promotor has the strongest leakage and the largest error bar as well. B The bacteria were treated with 10-10 mM AHL induction. High (Fluorescence/OD) ratio indicates high expression intensity. Mutation 5 has almost the same expression intensity as the wild type lux pR promotor.
Figure.1. Leakage and 10^-10 mM AHL stimulation of lux pR promotor and its mutations. The fluorescence strengths and OD values are measured by microplate reader respectively at 510 nm and 600 nm wavelength. Plasmids with lux pR promotor and its mutants are transferred into E.coli DH5α. A The bacteria were treated without AHL inducing. High (Fluorescence/OD) ratio indicates strong leakage of promotor. Among all the strains, lux pR promotor has the strongest leakage and the largest error bar as well. B The bacteria were treated with 10-10 mM AHL induction. High (Fluorescence/OD) ratio indicates high expression intensity. Mutation 5 has almost the same expression intensity as the wild type lux pR promotor. -
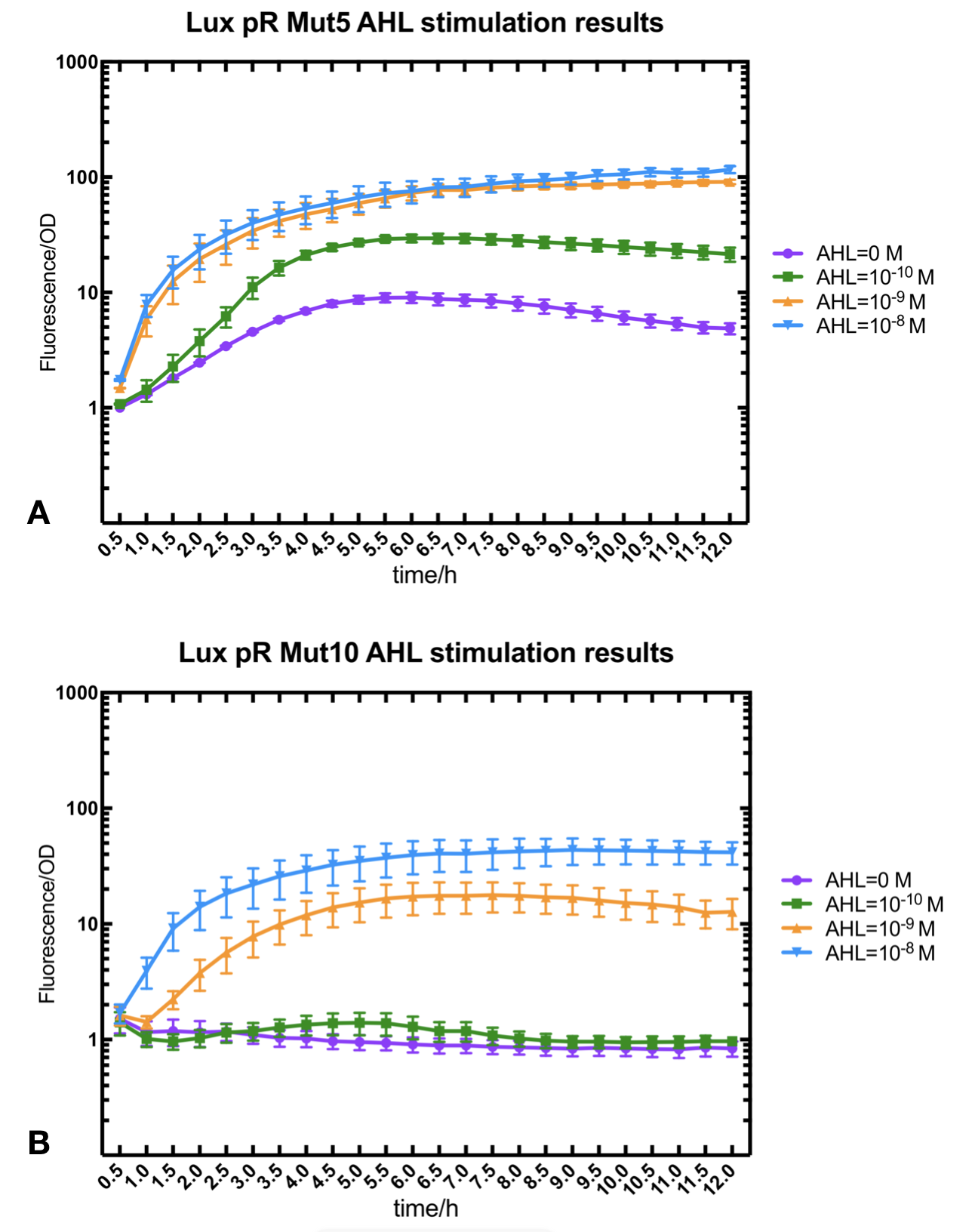 Figure.2. Leakage and AHL stimulation of of lux pR Mutant 5 and Mutant 10. The fluorescence strengths and OD values are measured by microplate reader respectively at 510 nm and 600 nm wavelength. A Plasmid with lux pR Mutant 5 is transferred into E.coli DH5α. The bacteria were treated without or with 10^-10 mM, 10^-9 mM and 10^-8 mM AHL inducing. B Plasmid with lux pR Mutant 10 is transferred into E.coli DH5α. The bacteria were treated without or with 10^-10 mM, 10^-9 mM and 10^-8 mM AHL inducing.
Figure.2. Leakage and AHL stimulation of of lux pR Mutant 5 and Mutant 10. The fluorescence strengths and OD values are measured by microplate reader respectively at 510 nm and 600 nm wavelength. A Plasmid with lux pR Mutant 5 is transferred into E.coli DH5α. The bacteria were treated without or with 10^-10 mM, 10^-9 mM and 10^-8 mM AHL inducing. B Plasmid with lux pR Mutant 10 is transferred into E.coli DH5α. The bacteria were treated without or with 10^-10 mM, 10^-9 mM and 10^-8 mM AHL inducing. -
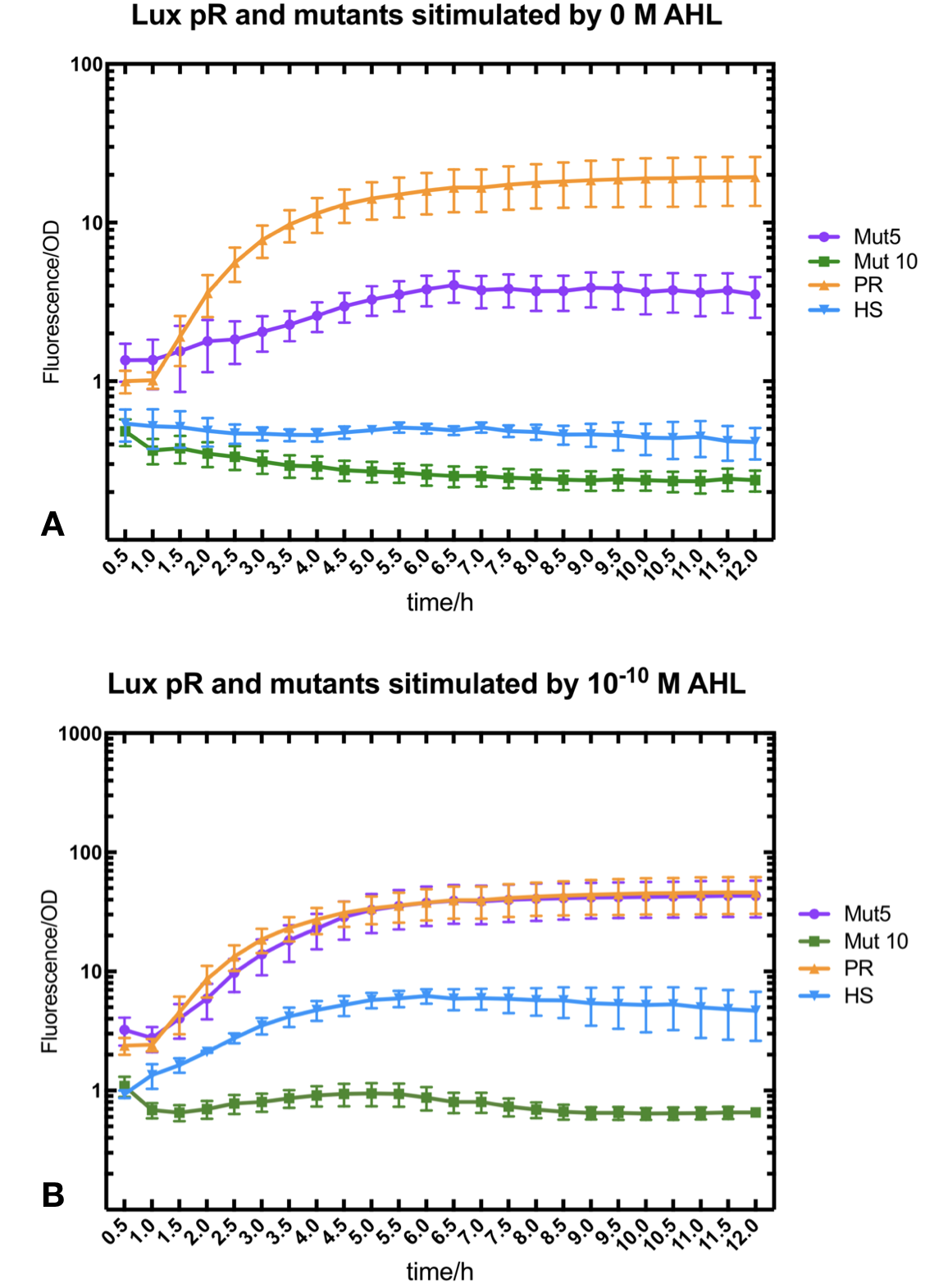 Figure.3. Leakage and 10^-10 mM AHL stimulation of of lux pR promotor WT, Mutant 5, Mutant 10 and HS. The fluorescence strengths and OD values are measured by microplate reader respectively at 510 nm and 600 nm wavelength. Plasmids with lux pR promotor WT, Mutant 5 and Mutant 10 are transferred into E.coli DH5α. A The bacteria were treated without AHL inducing. High (Fluorescence/OD) ratio indicates strong leakage of promotor. B The bacteria were treated with 10^-10 mM AHL induction. High (Fluorescence/OD) ratio indicates high expression intensity.
Figure.3. Leakage and 10^-10 mM AHL stimulation of of lux pR promotor WT, Mutant 5, Mutant 10 and HS. The fluorescence strengths and OD values are measured by microplate reader respectively at 510 nm and 600 nm wavelength. Plasmids with lux pR promotor WT, Mutant 5 and Mutant 10 are transferred into E.coli DH5α. A The bacteria were treated without AHL inducing. High (Fluorescence/OD) ratio indicates strong leakage of promotor. B The bacteria were treated with 10^-10 mM AHL induction. High (Fluorescence/OD) ratio indicates high expression intensity.We repeated AHL stimulation test on Mutant 5 and Mutant 10 (Figure.2) and decided to combine the two mutations together to form a new promotor with high intensity and small leakage. We named it lux pR-HS. We tested lux pR-HS under the same conditions. We can see that the lux pR-HS has similar leakage to Mutant 10, while it has much higher expression intensity. (Figure.3) The statistics of relative induction and leakage intensity of the 10 Mutants and lux pR-HS are shown in Table.1
-
As for why some mutations on -35 to -37 sites would decrease the leakage of lux pR without severely influencing the expression level, we have some hypotheses and explanations. It is reported that -35 to -37 sites are last three bases of lux box which is upstream of the lux promotor and bound specifically by luxR, one of regulatory protein involved in lux expression system.[1] G-36T mutation has less influence than other mutation on sites -35 to -37, which is same to our result. Besides, G-36T is not involved in the two regions of nucleotides -52 to -50 and -39 to -37 which directly contacted with luxR protein. Therefore, this single substitution of nucleotide would decrease the leakage of lux promotor without influencing the expression level too much.[2]
Protocol
- one Transform the plasmids into E. coli DH5α.
- two Pick a single colony by a sterile tip from each of the LB plates for all the experimental and control groups. Add the colony into 5ml LB medium with ampicillin at 100 ng/µl. Incubate for 6-8 h at 37℃ in a shaker.
- three Measure OD600 of the culture medium with photometer. Dilute the culture medium until OD600 reaches 0.6.
- four Add 100 µl bacteria culture medium into a sterile 96-well plate. Add AHL to final concentrations of 0, 10^-10,10^-9,10^-8 M. Fresh LB medium serves as blank control. Positive control is colony constantly expressing sfGFP and negative control is colony without sfGFP expression. Place the 96-well plate into an automatic microplate reader. Incubate at 16℃ overnight and measure the fluorometric value at 510 nm and OD600 of each well every 30 minutes.
- five Each group should be repeated for at least 3 times.
Reference
[1] Antunes, L. C., et al. "A mutational analysis defines Vibrio fischeri LuxR binding sites." Journal of Bacteriology 190.13(2008):4392-4397.
[2] Zeng, Weiqian, et al. "Rational design of an ultrasensitive quorum-sensing switch." Acs Synthetic Biology 6.8(2017).
SJTU-BioX-Shanghai 2019's Characterization
This lux pR-HS promoter is put upstream of repL(BBa_K2996000) to form a composite part(BBa_K2996006) that will respond to AHL, together with a constant expression of LuxR. RepL is a P1 lytic replication element that can act on mini-F origin to promote plasmid replication. Thus we quantified the effect of lux pR-HS indirectly by measuring plasmid copy number through qPCR. We tried different AHL concentration from 0 to 10-6 M and measured copy number in the time course. Both absolute copy number and relevant copy number amplification compared to non-induction were calculated. The result confirmed the small leakage of lux pR-HS and it also showed obvious responding to AHL, with the maximum effect reached at a concentration of 10-7 M at 3.5h of induction.
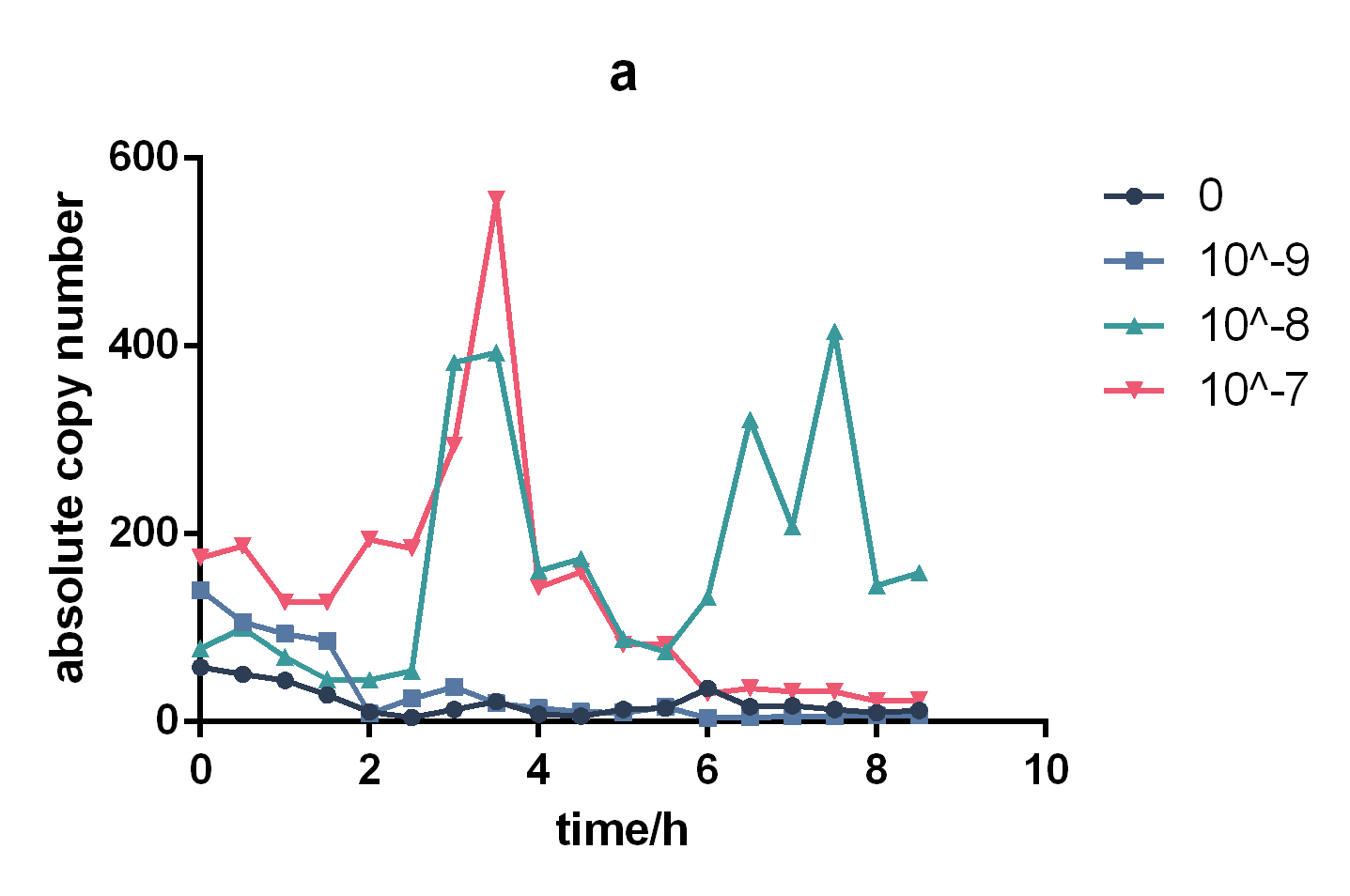
Figure 1. Change of absolute copy number under different induction concentrations 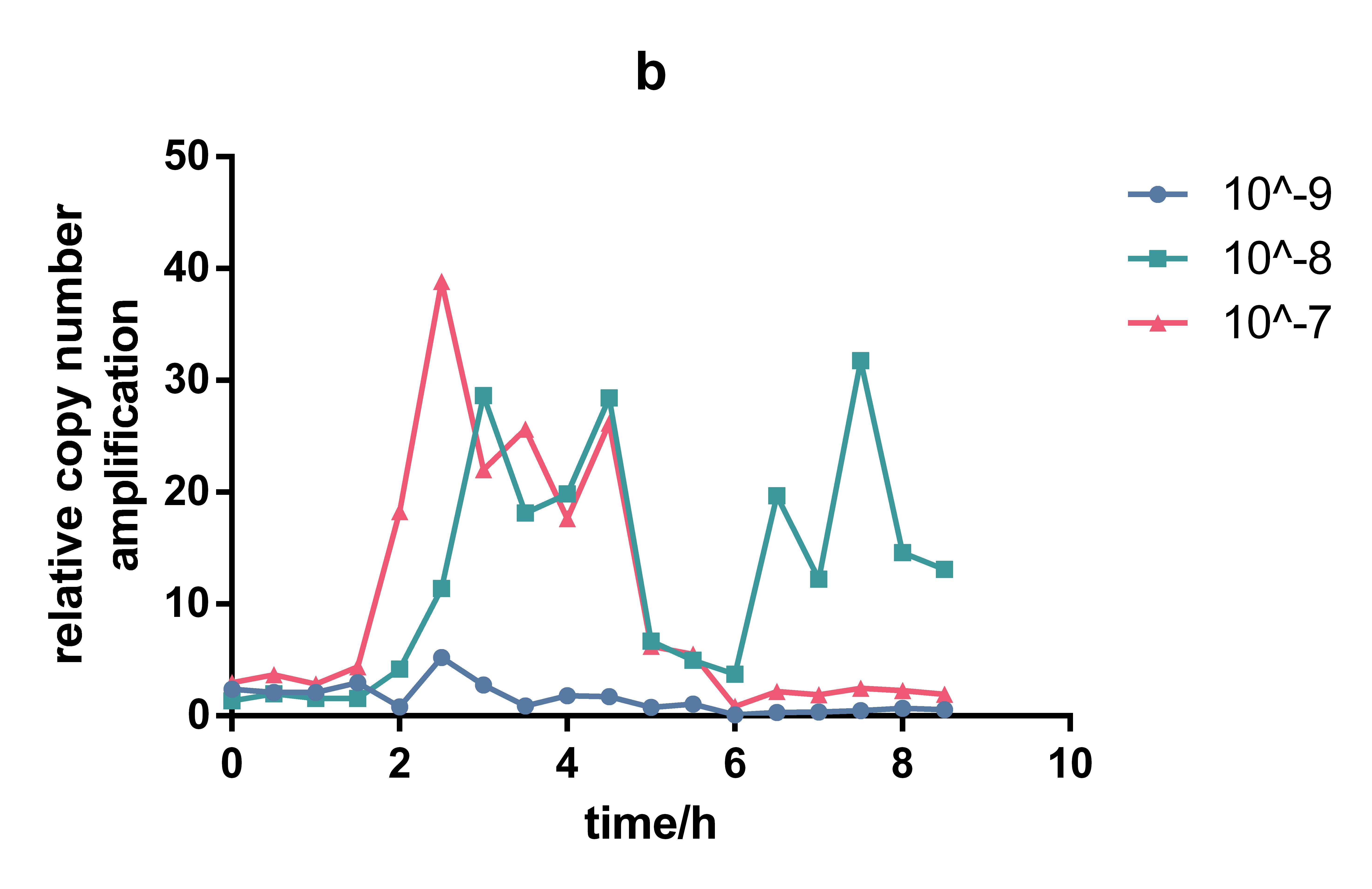
Figure 1. Change of relativecopy number under different induction concentrations
Protocol
AHL induction and preparation for copy number measurement. (1)Transfer pLux-trig(with luxpR-HS driven repL) and pLux-cas(with luxpR-HS driven Cas1/2 and constantly expressed LuxR) into E.coli BL21(DE3) (2)Inoculate 1% overnight bacterial culture in Erlenmeyer flask. (3)Incubate for 2h until early exponential stage (OD600 is 0.2-0.3). (4)Add AHL to final concentrations of 10-9, 10-8, 10-7, 10-6 M. (5)Take 500μL of the culture every 30min. (6)Centrifuge at 4000rpm for 5min immediately after taking out. (7)Discard the supernatant and wash with 500μL PBS. (8)Heat at 100℃ for 10min. (9)Store at -20℃ for at least 20min. (10)Centrifuge at 6500rpm for 1min. (11)Remove the supernatant into a clean 1.5mL EP tube. (12)Store at -20℃. (13)Add 3ul as qPCR template and run qPCR.
Fudan 2019’s characterization and improvement:
Background:
In our project, we hope that this promoter can trigger the regulation of the downstream tetR circuit properly without leakage and subsequent deactivation. Since we use the production of its upstream regulating genes luxI and luxR as its signal in our final circuit, our measurement continued this design and measure the curve basing on OD value. Data collected in unit of the concentration of bacteria will directly reflect if the system works properly.
Experiment design and measurement:
To test this part’s function, we transferred DH10B bacteria with plasmids carrying the promoter with another LuxI-LuxR ( BBa_K3245002 ) plasmid at the same time. LuxI-LuxR is a upstream regulation box which produces signal that can activate luxpR in QS system. And we added GFP after luxpR as the reporter of its expression level. By measuring fluorescence/OD600, the expressing level of luxpR can be known and the data are comparable with our other works.
Result:
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
It is observed that high expression intensity is accompanied by high level of leakage in lux pR promotors. Wild type lux pR represented with black strand in the figures has the highest level of both leakage and intensity. The next three are Mutant 5 (G-36T), Mutant 6 (G-36C), Mutant 9 (T-35G). Mutant 5 has almost the same intensity as wild type, but the leakage is significantly smaller. Mutant 6 and Mutant 9 have a little bit less leakage and medium intensity. We also did the experiment with 10^-9 M and 10^-8 M AHL, all of the promotors show high expression levels and therefore unable to compare. (Data not shown)
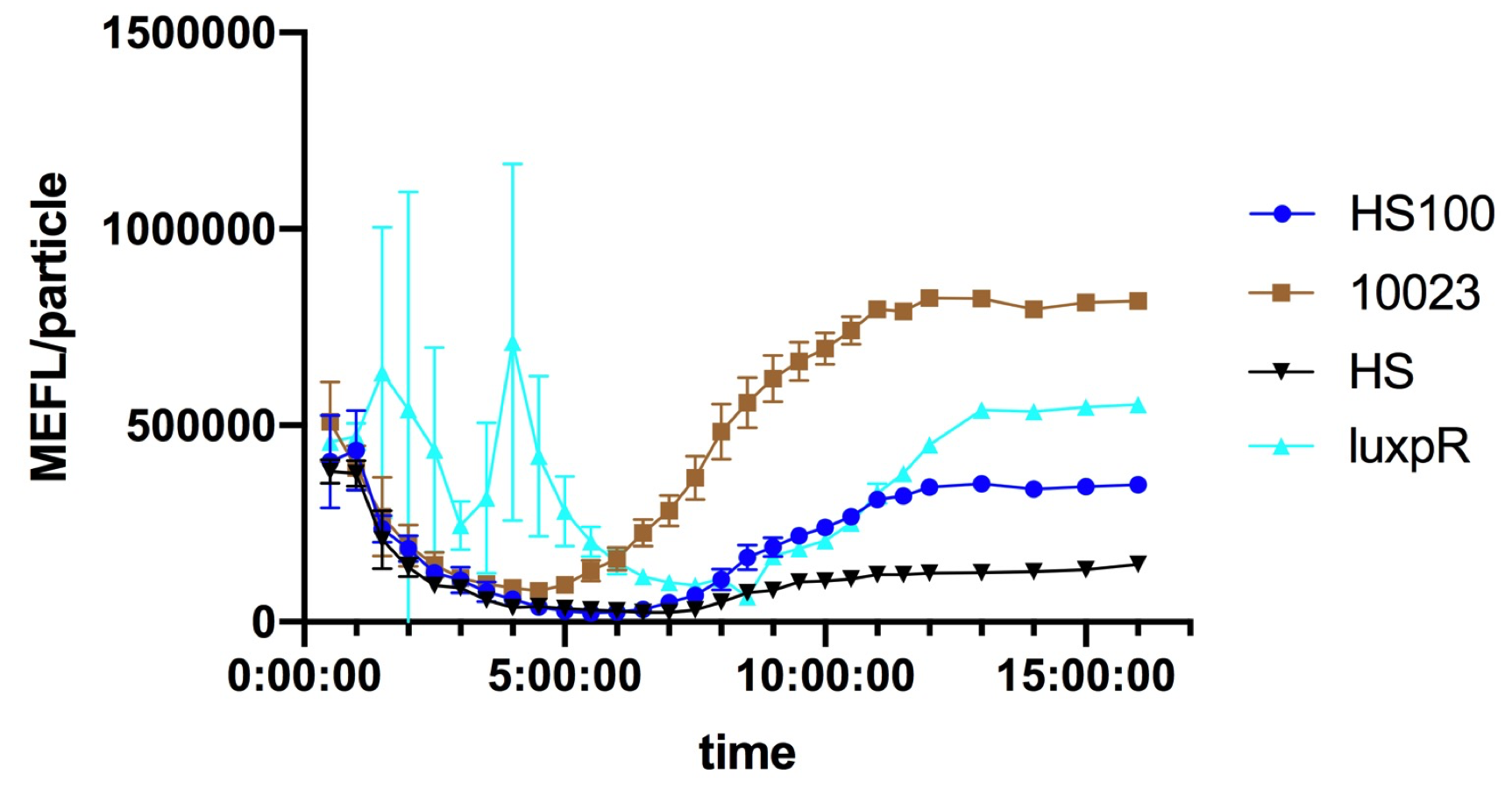
Fig.1 This figure shows luxpR-HS expression level induced by luxR-luxI box. The MEFL/particle changes as the OD goes up after dilution. Data points are shown in the figure for fitting analysis. Here gives the parameters of the fitting equation.
At first the parental fluorescence and dilution effect keep fluorescence/OD600 at an extremely high level. Then the concentration of signal molecules remains low in the fresh culture while the bacteria are reproducing. In the next few hours the fluorescence/OD600 keeps going down. After the newly cultured bacteria grow to a higher concentration, luxpR-HS is activated and fluorescence/OD600 rises again as Fig.1 shows. Expression properties of luxpR-HS is predictable according to the fitting analysis results.
Improvement:
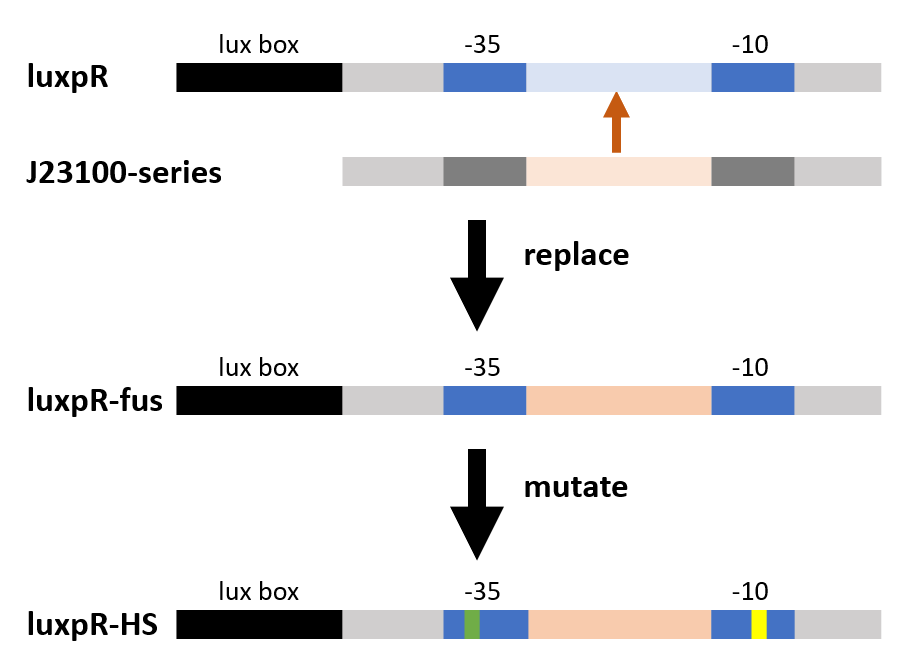
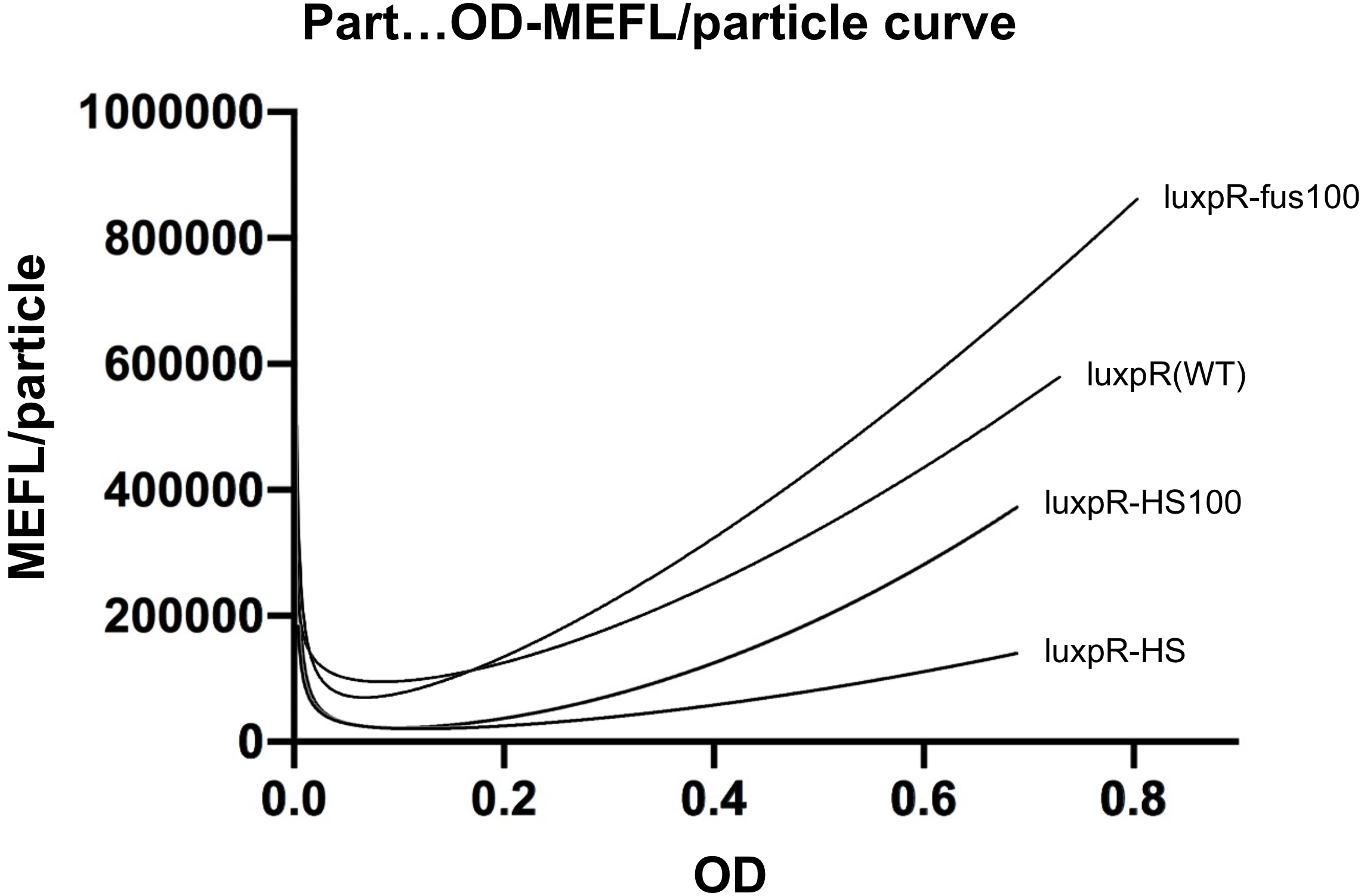
We substituted the -35 to -10 region of the original promoter, luxpR-HS with the one from constitutive promoter family members (BBa_J23100 :https://parts.igem.org/Part:BBa_J23100), according to the knowledge of lux box[1] and the method of constructing hybrid promoters[2]. Finally we picked luxpR-HS100 as our improved part and deployed it in our circuit.
Design:
Fig.2 Hybrid promoter diagram. As the figure shows, we substituted the -35 to -10 region of the original promoter luxpR with the one of J23100, a strong constitutive promoter. This region is rarely concerned as it’s rather conservative in promoters with the same function, but it has crucial structural effect on σ factor binding and other events in transcription regulation. Fortunately the change proved to be effective on adjusting the behavior of the regulatory promoter.
Result:
Fig. 3 Microplate reader data of quorum sensing promoters ( time as X-axis ). It shows the overall status of the promoters. Both luxpR-HS100 and luxpR-HS show low leakage at the low OD600 period, at the platform HS100 shows higher expression level than HS.
Fig. 4 Microplate reader data of quorum sensing promoters ( OD600 as X-axis ). The downward curve before OD600 <0.1 is due to the parental fluorescence and the dilution effect after proliferation. It is shown that luxpR-HS100 has a higher expression level and keeps low leakage similar to luxpR-HS.
Protocol:
We cultured the bacteria mentioned above overnight. As it reached a high concentration, luxpR-HS became activated. Then we diluted the culture to 1/5000 and measured its fluorescence and OD600 every 30 minutes.
1. Inoculate the selected single colony with 4 kinds of luxpR promoters above into 3 ml LB medium with 50 mg/ml kanamycin and 100 mg/ml ampicillin at 37℃.
2. Dilute the overnight culture to 1/5000 in 15ml fresh LB medium with 50 mg/ml kanamycin and 100mg/ml ampicillin. Each group with two parallel copies.
3. Incubate all culture above at 37℃.
4. Pipette 200 μl each as sample every 30 minutes and measure their fluorescence and OD600.
Reference:
[1] Antunes, L. C., et al. "A mutational analysis defines Vibrio fischeri LuxR binding sites." Journal of Bacteriology 190.13(2008):4392-4397.
[2] de Boer HA, Comstock LJ, Vasser M. The tac promoter: a functional hybrid derived from the trp and lac promoters. Proc Natl Acad Sci U S A. 1983;80(1):21–25. doi:10.1073/pnas.80.1.21
Sequence and Features