Difference between revisions of "Part:BBa K3190501"
| (10 intermediate revisions by the same user not shown) | |||
| Line 3: | Line 3: | ||
<partinfo>BBa_K3190501 short</partinfo> | <partinfo>BBa_K3190501 short</partinfo> | ||
| − | Coding sequence of the mammalian apoptosis regulator Bax protein. The gene will be integrated into the genome of the fungal chassis Saccharomyces cerevisiae, acting as the key gene in our biosafety device. Overexpression of Bax is lethal in <i>S. cerevisiae</i>, leading to the release of cytochrome c from mitochondria and thus apoptosis. The following results are characterization of a part submitted by iGEM17_NAU-CHINA: <partinfo>BBa_K2365048</partinfo>. | + | Coding sequence of the mammalian apoptosis regulator Bax protein. The gene will be integrated into the genome of the fungal chassis <i>Saccharomyces cerevisiae</i>, acting as the key gene in our biosafety device. Overexpression of Bax is lethal in <i>S. cerevisiae</i>, leading to the release of cytochrome c from mitochondria and thus apoptosis. The following results are characterization of a part submitted by iGEM17_NAU-CHINA: <partinfo>BBa_K2365048</partinfo>. |
===Usage and Biology=== | ===Usage and Biology=== | ||
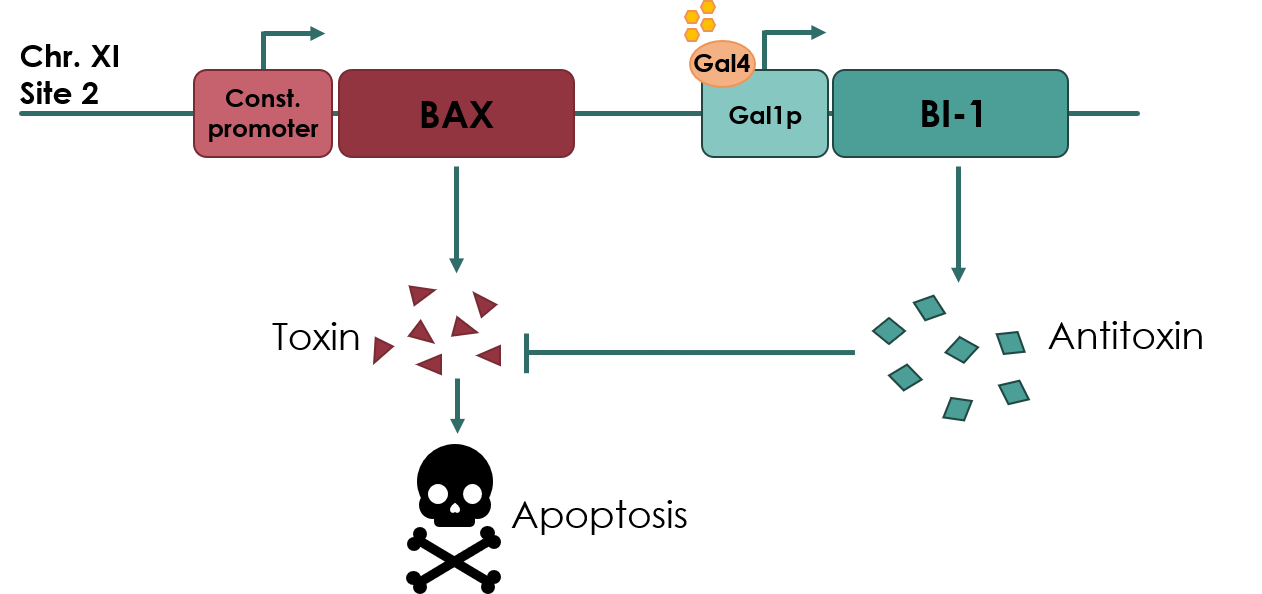
| − | For our biosafety device, the Bax protein will be under the expression of a constitutive promoter, pADH2 | + | For our biosafety device, the Bax protein will be under the expression of a constitutive promoter, pADH2. An anti-toxin, BAX Inhibitor-1 (BI-1) (<partinfo>BBa_K3190502</partinfo>) will be under the expression of an inducible promoter, pGAL1 (<partinfo>BBa_K3190050</partinfo>). As long as there is galactose present in the media, the anti-toxin will be expressed, inhibiting the apoptotic effects of Bax protein. If the cell escapes the media, the antitoxin will no longer be expressed, and Bax protein will cause apoptosis of the cell. |
[[File:UCopenhagen Killswitch.jpeg|600px]] | [[File:UCopenhagen Killswitch.jpeg|600px]] | ||
| − | <small> Figure | + | <small> <b> Figure 1: Overview of our toxin/anti-toxin biosafety device. </b> </small> |
| + | |||
<b> Designing the construct </b> | <b> Designing the construct </b> | ||
| Line 19: | Line 20: | ||
Using USER ligation, we assembled the BAX gene with pGAL1 on a plasmid backbone compatible with multiplex integration cassette. The backbone used contains a URA selection marker, and will integrate the construct in the yeast genome at chromosome 11, site 2. | Using USER ligation, we assembled the BAX gene with pGAL1 on a plasmid backbone compatible with multiplex integration cassette. The backbone used contains a URA selection marker, and will integrate the construct in the yeast genome at chromosome 11, site 2. | ||
| − | |||
| − | + | <b> Yeast transformation </b> | |
| + | |||
| + | Initially, our yeast was transformed with an episomal plasmids containing BAX under a galactose inducible promoter. For our control strain we used an empty vector. Following transformation, each culture were split into two. One half was plated on a plate containing raffinose selection media with 0.0 % galactose, and the other half on raffinose selection media containing 1.0 % galactose. In both strains a second empty vector was introduced, placing both strains under Uracil (U) and Tryptophan (W) selection. | ||
| + | |||
| + | As can be seen on Figure 3, no colonies were seen on the plate containing galactose, suggesting that the BAX protein was expressed in such a high concentration that all cells died. For our control strain an equal amount of cells were seen both with and without galactose. | ||
| + | |||
| + | For more stable expression, we also interated pGAL1-BAX into the chromosome of <i>S. cerevisiae</i>, using U selection. Here, positive transformants were selected for by performing yeast colony PCR (Figure 1). This strain was used for all further experiments. | ||
| + | |||
| + | [[File:ovulaid29.jpg|300px]] | ||
| + | |||
| + | <small> <b> Figure 2: Yeast colony PCR</b> | The positive colony of yeast is confirmed by the expected band size of around 800 bp.</small> | ||
| + | |||
| + | [[File:PGAL1-BAX A.jpeg|300px]] [[File:PGAL1-BAX B.jpeg|300px]] | ||
| + | |||
| + | <small><b>Figure 3: Transformant plates of dual plasmid transformed <i>S. cerevisiae</i></b> | Both transformed with pGAL1-BAX (URA marker) and an empty vector (TRP marker). A: plate with no galactose. B: plate with 1 % galactose. </small> | ||
| + | |||
| + | |||
| + | To analyse the effect of BAX on yeast under different expression levels, we conducted a galactose induction assay using raff-U plates with five different galactose concentrations. | ||
| + | Cultures of yeast containing pGAL1-BAX or empty vector were grown O/N and then diluted to an OD<sub>600nm</sub> of 0.5. On plates with either 0%, 0.025%, 0.05%, 0.1% or 0.2% galactose, 10 µl of each culture were spotted in increasing dilutions (10<sup>-1</sup> to 10<sup>-4</sup>; Figure 4 and 5) and incubated for three days at 30 °C. The strain with the integrated pGAL1-BAX construct showed decreased growth compared to the control strain even when galactose was absent (Figure 5). This suggests that the galactose promoter is leaky and a low amount of BAX is produced at all times. | ||
| + | |||
| + | [[File:ovulaid31.png|600px]] | ||
| + | |||
| + | <small><b>Figure 4: Growth of pGAL1-BAX and the empty vector strain in the absence of galactose. </b>The cultures were spotted in the dilutions 10<sup>-1</sup> to 10<sup>-4</sup> of an OD<sub>600nm</sub> of 0.5 and incubated for three days at 30 °C. </small> | ||
| + | |||
| + | Moreover, a clear inverse correlation of CFU/ml and percentage of galactose in the media can be observed (Figure 5) when comparing the spots of the pGAL1-BAX strain at a dilution of 10<sup>-1</sup> at different galactose concentrations. This suggests that successful induction of BAX leads to apoptosis in our yeast. | ||
| + | |||
| + | [[File:ovulaid32.png|600px]] | ||
| − | < | + | <small><b>Figure 5: Growth comparison of pGAL1-BAX and control strain in the presence of varying galactose concentrations.</b> | Shown are the CFUs at an OD<sub>600nm</sub> of 0.05 after incubation at 30 °C for four days. The three yellow colonies seen at 0.2% galactose on the pGAL-BAX plate can be morphologically distinguished from the others, suggesting that they are contaminants.</small> |
| − | + | ||
| − | + | <br> | |
| − | + | <b> <font size="4">Quantitative galactose induction assay</font> </b> | |
| − | < | + | |
| − | < | + | To further analyze the effect of BAX on our yeast under different expression levels, we conducted a quantitative galactose induction assay. Here, an O/N culture of yeast containing pGAL1-BAX was diluted to an OD<sub>600nm</sub> of 0.5. Subsequently, 100 µl of the culture in dilutions of 10<sup>-3</sup> and 10<sup>-4</sup> were spread on plates with 0%, 0.05%, 0.1%, 0.3% and 1% galactose, respectively. Each plate was made in duplicate. After incubation for three days at 30 °C, the CFU/ml were calculated and compared to the control (Figure 6). |
| − | + | <b> [Discussion of below graph] </b> | |
| − | + | [[File:ovulaid30.png|600px]] | |
| − | < | + | <small><b>Figure 6: Gradient induction of BAX in yeast using the inducible GAL1 promoter.</b> | If no errorbar is indicated, only one of the used duplets showed a quantifiable amount of colonies. </small> |
| − | + | ||
| − | + | As seen on figure 7, a clear reduction in growth was seen already at low galactose concentrations (0.05 %). In addition it appears BAX was expressed in sufficient concentrations to kill the yeast cells at a galactose concentrations of 0.3 %. Unfortunately our control strain was only plated on raff-U with 1 % galactose. However, no reduction in growth was seen at this concentration (compared to pGAL1-BAX grown in 0 % galactose media). | |
| − | + | In addition to a reduction in the number of colonies formed between the two strains, there was also a clear difference on the size of the colonies. Only very small colonies were formed upon induction of BAX, this that the cells were killed before a normal sized colony could be formed. To test this, the pGAL1-BAX plate was incubated for an additional day at 30 °C and subsequently left at room temperature. No additional growth was observed (Figure 7). | |
| − | + | [[File:ovulaid33.png|600px]] | |
| − | + | ||
| − | <b> | + | <small><b>Figure 7: Comparison of colony sizes of pGAL1-BAX and the control containing the empty vector grown on raff-U agar with galactose.</b> | Left: Colonies of the control strain (dilution 10<sup>-3</sup>) in the presence of 1% galactose after incubation for three days at 30 °C. Right: Colonies of the pGAL1-BAX strain (dilution 10<sup>-3</sup>) in the presence of 0.1% galactose after incubation for four days at 30 °C. </small> |
| − | + | ||
Latest revision as of 23:58, 21 October 2019
BAX: Bcl-2-associated X protein CDS
Coding sequence of the mammalian apoptosis regulator Bax protein. The gene will be integrated into the genome of the fungal chassis Saccharomyces cerevisiae, acting as the key gene in our biosafety device. Overexpression of Bax is lethal in S. cerevisiae, leading to the release of cytochrome c from mitochondria and thus apoptosis. The following results are characterization of a part submitted by iGEM17_NAU-CHINA: BBa_K2365048.
Usage and Biology
For our biosafety device, the Bax protein will be under the expression of a constitutive promoter, pADH2. An anti-toxin, BAX Inhibitor-1 (BI-1) (BBa_K3190502) will be under the expression of an inducible promoter, pGAL1 (BBa_K3190050). As long as there is galactose present in the media, the anti-toxin will be expressed, inhibiting the apoptotic effects of Bax protein. If the cell escapes the media, the antitoxin will no longer be expressed, and Bax protein will cause apoptosis of the cell.
Figure 1: Overview of our toxin/anti-toxin biosafety device.
Designing the construct
In order to characterize the Bax protein, we cloned it to inducible promoter pGAL1 (BBa_K3190050), as expression under a constitutive promoter would be expected to cause the cells to die.
Using USER ligation, we assembled the BAX gene with pGAL1 on a plasmid backbone compatible with multiplex integration cassette. The backbone used contains a URA selection marker, and will integrate the construct in the yeast genome at chromosome 11, site 2.
Yeast transformation
Initially, our yeast was transformed with an episomal plasmids containing BAX under a galactose inducible promoter. For our control strain we used an empty vector. Following transformation, each culture were split into two. One half was plated on a plate containing raffinose selection media with 0.0 % galactose, and the other half on raffinose selection media containing 1.0 % galactose. In both strains a second empty vector was introduced, placing both strains under Uracil (U) and Tryptophan (W) selection.
As can be seen on Figure 3, no colonies were seen on the plate containing galactose, suggesting that the BAX protein was expressed in such a high concentration that all cells died. For our control strain an equal amount of cells were seen both with and without galactose.
For more stable expression, we also interated pGAL1-BAX into the chromosome of S. cerevisiae, using U selection. Here, positive transformants were selected for by performing yeast colony PCR (Figure 1). This strain was used for all further experiments.
Figure 2: Yeast colony PCR | The positive colony of yeast is confirmed by the expected band size of around 800 bp.
Figure 3: Transformant plates of dual plasmid transformed S. cerevisiae | Both transformed with pGAL1-BAX (URA marker) and an empty vector (TRP marker). A: plate with no galactose. B: plate with 1 % galactose.
To analyse the effect of BAX on yeast under different expression levels, we conducted a galactose induction assay using raff-U plates with five different galactose concentrations.
Cultures of yeast containing pGAL1-BAX or empty vector were grown O/N and then diluted to an OD600nm of 0.5. On plates with either 0%, 0.025%, 0.05%, 0.1% or 0.2% galactose, 10 µl of each culture were spotted in increasing dilutions (10-1 to 10-4; Figure 4 and 5) and incubated for three days at 30 °C. The strain with the integrated pGAL1-BAX construct showed decreased growth compared to the control strain even when galactose was absent (Figure 5). This suggests that the galactose promoter is leaky and a low amount of BAX is produced at all times.
Figure 4: Growth of pGAL1-BAX and the empty vector strain in the absence of galactose. The cultures were spotted in the dilutions 10-1 to 10-4 of an OD600nm of 0.5 and incubated for three days at 30 °C.
Moreover, a clear inverse correlation of CFU/ml and percentage of galactose in the media can be observed (Figure 5) when comparing the spots of the pGAL1-BAX strain at a dilution of 10-1 at different galactose concentrations. This suggests that successful induction of BAX leads to apoptosis in our yeast.
Figure 5: Growth comparison of pGAL1-BAX and control strain in the presence of varying galactose concentrations. | Shown are the CFUs at an OD600nm of 0.05 after incubation at 30 °C for four days. The three yellow colonies seen at 0.2% galactose on the pGAL-BAX plate can be morphologically distinguished from the others, suggesting that they are contaminants.
Quantitative galactose induction assay
To further analyze the effect of BAX on our yeast under different expression levels, we conducted a quantitative galactose induction assay. Here, an O/N culture of yeast containing pGAL1-BAX was diluted to an OD600nm of 0.5. Subsequently, 100 µl of the culture in dilutions of 10-3 and 10-4 were spread on plates with 0%, 0.05%, 0.1%, 0.3% and 1% galactose, respectively. Each plate was made in duplicate. After incubation for three days at 30 °C, the CFU/ml were calculated and compared to the control (Figure 6).
[Discussion of below graph]
Figure 6: Gradient induction of BAX in yeast using the inducible GAL1 promoter. | If no errorbar is indicated, only one of the used duplets showed a quantifiable amount of colonies.
As seen on figure 7, a clear reduction in growth was seen already at low galactose concentrations (0.05 %). In addition it appears BAX was expressed in sufficient concentrations to kill the yeast cells at a galactose concentrations of 0.3 %. Unfortunately our control strain was only plated on raff-U with 1 % galactose. However, no reduction in growth was seen at this concentration (compared to pGAL1-BAX grown in 0 % galactose media). In addition to a reduction in the number of colonies formed between the two strains, there was also a clear difference on the size of the colonies. Only very small colonies were formed upon induction of BAX, this that the cells were killed before a normal sized colony could be formed. To test this, the pGAL1-BAX plate was incubated for an additional day at 30 °C and subsequently left at room temperature. No additional growth was observed (Figure 7).
Figure 7: Comparison of colony sizes of pGAL1-BAX and the control containing the empty vector grown on raff-U agar with galactose. | Left: Colonies of the control strain (dilution 10-3) in the presence of 1% galactose after incubation for three days at 30 °C. Right: Colonies of the pGAL1-BAX strain (dilution 10-3) in the presence of 0.1% galactose after incubation for four days at 30 °C.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21INCOMPATIBLE WITH RFC[21]Illegal BamHI site found at 452
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]