Difference between revisions of "Part:BBa K2541001"
| (42 intermediate revisions by 4 users not shown) | |||
| Line 4: | Line 4: | ||
<h5> | <h5> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
| − | A RNA-based thermosensor that can be used for temperature sensitive | + | A RNA-based thermosensor that can be used for temperature sensitive translational regulation which is based on the change of RNA sencondary structure. The heat-inducible RNA-based thermosensors can initiate translation of downstream genes at high temperatures. |
</p> | </p> | ||
</h5> | </h5> | ||
| Line 11: | Line 11: | ||
<h5> | <h5> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
| − | Heat-inducible RNA-based thermosensors are RNA genetic control systems that sense temperature changes. At low temperatures, the mRNA adopts a stem-loop conformation that masks the Shine–Dalgarno (SD) sequence within the 5’-untranslated region (5’-UTR) and, in this way, prevents ribosome binding and translation. At elevated temperatures, the RNA secondary structure melts locally, thereby giving the ribosomes access to the SD sequence to initiate translation (Figure 1). Whereas natural RNA-based thermosensors have a relatively complicated secondary structure with multiple stems, hairpin loops and bulges | + | Heat-inducible RNA-based thermosensors are RNA genetic control systems that sense temperature changes. At low temperatures, the mRNA adopts a stem-loop conformation that masks the Shine–Dalgarno (SD) sequence within the 5’-untranslated region (5’-UTR) and, in this way, prevents ribosome binding and translation. At elevated temperatures, the RNA secondary structure melts locally, thereby giving the ribosomes access to the SD sequence to initiate translation (Figure 1). Whereas natural RNA-based thermosensors have a relatively complicated secondary structure with multiple stems, hairpin loops and bulges which impeds application process. |
</p> | </p> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
| Line 230: | Line 230: | ||
</g> | </g> | ||
</svg> | </svg> | ||
| − | <p>Figure 1. Mechanism of heat inducible RNA-based thermosensors.</p> | + | <p>Figure 1. Mechanism of heat-inducible RNA-based thermosensors.</p> |
</div> | </div> | ||
<script type="text/javascript"> | <script type="text/javascript"> | ||
| Line 246: | Line 246: | ||
<h5> | <h5> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
| − | The | + | The heat-inducible RNA-based theromsensors are designed on the basis of the melting temperature of the minimum free energy structure, consisting of ASD (anti-SD) sequence, loop sequence and consensus SD sequence (5’-AAGGAG-3’). To optimize the thermosensors for the desired melting temperature, intensity and sensitivity, three structural parameters come into consideration: stem length, loop size and mismatches or bulges in the stem. |
</p> | </p> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
| − | Stem length is determined by ASD sequence because the SD sequence is conserved. Adding stem length can optimize heat inducible RNA-based thermosensors to more high temperature, while decreasing stem length has the opposite effect. The stem length is 4 base parings in K2541001. Loop size can moderate thermosensors melting temperature to a suitable temperature. In K2541001, the loop sequence is AAUAA. Finally, we get the sequence as the figure 2. | + | Stem length is determined by ASD sequence because the SD sequence is conserved. Adding stem length can optimize heat-inducible RNA-based thermosensors to more high temperature, while decreasing stem length has the opposite effect. The stem length is 4 base parings in K2541001. Loop size can moderate thermosensors melting temperature to a suitable temperature. In K2541001, the loop sequence is AAUAA. Finally, we get the sequence as the figure 2B. After designing, the theromsensor sequence is predicted by computational methods mFOLD to get its minimum free energy and secondary structure (figure 2). |
</p> | </p> | ||
</h5> | </h5> | ||
| − | [[File:K2541001 f2.png|center| | + | [[File:K2541001 f2 new.png|center|K2541001 f2 new]] |
| − | Figure 2. Design of | + | Figure 2. Design of K2541001. (A) The RNA secondary structure is predicted by mFOLD. (B) Thermosensor sequence, ASD sequence, loop sequence, the site of mismatch or bulge in the stem and ΔG are in the table. |
<h1>'''3.Characterization'''</h1> | <h1>'''3.Characterization'''</h1> | ||
| + | <h3>3.1 Measurement device</h3> | ||
<h5> | <h5> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
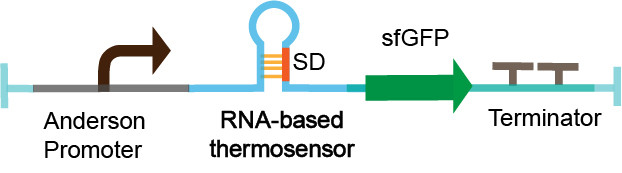
| − | The thermosensor sequence is constructed on the pSB1C3 vector by | + | The thermosensor sequence is constructed on the pSB1C3 vector by Golden Gate assembly. The measurement device is composed of Anderson promoter (BBa_J23104), thermosensor (BBa_K2541001), sfGFP_optimism (BBa_K2541400) and double terminator (BBa_B0010 and BBa_B0012). We select a constitutive Anderson promoter J23104 as an appropriate promoter by pre-experiment. The sfGFP_optimism has faster folding speed and higher fluorescence intensity. The double terminator can reduce leakage (Figure 3). We characterized RNA-based thermosensors in ''E.coli'' DH5a. |
| − | + | ||
</p> | </p> | ||
</h5> | </h5> | ||
| − | [[File:measurement device | + | [[File:measurement device 123.png|center|measurement device 123]] |
<html><center> | <html><center> | ||
Figure 3. The measurement device. | Figure 3. The measurement device. | ||
| Line 269: | Line 269: | ||
---- | ---- | ||
| − | + | <h3>3.2 Measurement results</h3> | |
<h5> | <h5> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
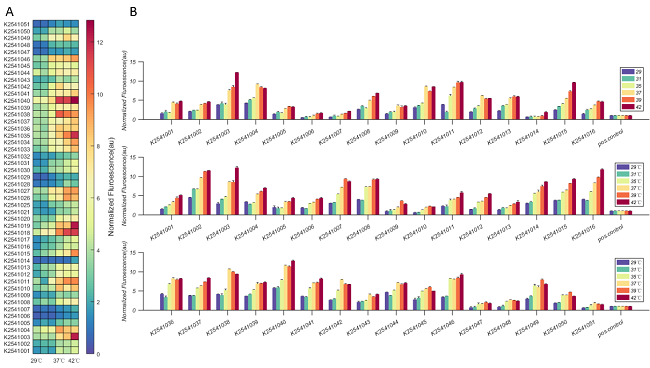
| − | + | K2541001, K2541029, K2541013 and K2541037 are four different heat-inducible RNA-based thermosensors. pos.control is positive control. The final normalized fluorescence was calculated as follows: normalized fluorescence = [(Fluorescence/Abs<sub>600</sub>)<sub>TS</sub> - (Fluorescence/Abs<sub>600</sub>)<sub>neg</sub>] / [(Fluorescence/Abs<sub>600</sub>)<sub>pos</sub> - (Fluorescence/Abs<sub>600</sub>)<sub>neg</sub>] ( TS = thermosensor, pos = positive control, and neg = BBa_J364007 ). As shown in figure 4, the fluorescence intensity of K2541001 increases with elevated temperature. | |
</p> | </p> | ||
</h5> | </h5> | ||
| − | [[File:K2541001 f4.png|center| | + | [[File:K2541001 f4.png|center|K2541001 f4]] |
| − | Figure 4. Characteristics of | + | Figure 4. Characteristics of K2541001. Each set of six bars represents the activity level of a different thermosensor. The bar colors purple, aquamarine, light green, orange, red and brown represent the temperatures 29, 31, 35, 37, 39 and 42°C, respectively. The height of the bars corresponds to the normalized fluorescence. |
---- | ---- | ||
| + | <h3>3.3 Thermodynamic analyses</h3> | ||
| + | <h5> | ||
| + | <P style="text-indent:2em;"> | ||
| + | This year, our project is to design a collection of RNA-based thermosensors with different melting temperatures, intensity and sensitivity. We used a combination of experimental measurements and computations of RNA secondary structures. We studied a set of measured synthetic RNA-based thermosensors, finding consistency among our experimental and computational analyses. | ||
| + | </p> | ||
| + | </h5> | ||
| + | [[File:K2514001 f5 c2.png|center|K2514001 f5 c2]] | ||
| + | Figure 5. Temperature response of the heat-inducible RNA-based thermosensor. (A) The red solid line represents the activity of the RNA-based thermosensor as a function of temperature. (B) Key quantitative features such as melting temperature, relative intensity, sensitivity and R-squared are in the table. | ||
| + | <h1>'''4. Collection of heat-inducible RNA-based thermosensors '''</h1> | ||
| + | [[File:RT figure6 新.png|center|RT figure6 新]] | ||
| + | Figure 6. Experimental measurements of the collection of heat-inducible RNA-based thermosensors show a variety of responses. (A) Rows represent activity levels of different thermosensors. (B) Each set of six bars represents the activity level of a different thermosensor. The bar colors purple, aquamarine, light green, orange, red and brown represent the temperatures 29, 31, 35, 37, 39 and 42°C, respectively. The height of the bars corresponds to the normalized fluorescence. | ||
| + | |||
| + | <h1>'''5. Conclusion'''</h1> | ||
<h5> | <h5> | ||
<P style="text-indent:2em;"> | <P style="text-indent:2em;"> | ||
| − | + | Our data show that efficient RNA-based thermosensors with different melting temperatures, intensity and sensitivity can be built from a single small RNA stem-loop structure masking the SD sequence, thus providing useful SynRT toolkit for the regulation of gene expression. | |
</p> | </p> | ||
</h5> | </h5> | ||
| + | ==Verification from SHSBNU_China 2020== | ||
| + | In SHSBNU_China team's design, we use this thermosensor as a heat-responsive suicide switch. Locust plagues take place mainly in spring, and few in summer. Therefore, in spring, when the temperature is lower than 35 ℃, the mRNA adopts a stem-loop conformation that masks the Shine–Dalgarno (SD) sequence within the 5’-untranslated region (5’-UTR) and, in this way, prevents ribosome binding and translation [1]. In summer, when the temperature is higher than 35 ℃, the RNA secondary structure melts locally, thus the lysis gene in the downstream of the gene circuit would start transcription, resulting in the demise of our engineered bacteria. | ||
| − | + | ==Characterization== | |
| − | + | ===Plasmid construction=== | |
| + | In our experiments, we used this part: K2541001, along with the sfGFP (BBa_I746916) as a reporter and PhiX174E (BBa_K3594001), as our heat-responsive suicide switch. We constructed two plasmids in order to complete the thermal response suicide switch successfully: PSHS1_PSB1C3_BBa_J23104_K2541001_sfGFP and PSHS2_PSB1C3_BBa_J23104_K2541001_Phix174E. PSHS1 can sense high temperature and generate fluorescent, which is used to verify if the thermal response switch is effective. PSHS2 contains a cell lysis gene PhiX174E, and this plasmid is used to test if our heat-induced suicide system works well. | ||
| + | ===Testing Result=== | ||
| + | Firstly, we tested the function of BBa_K3594013. In order to test whether it works and produce fluorescence, we decided to use fluorescence microplate reader to measure the normalized fluorescence that the gene expresses in different temperature. | ||
| + | We transformed BBa_K3594013 to commercial E. coli DH5 alpha competent cells. Then, we cultured the single colonies with PSHS1 for overnight. At the next day, we diluted the culture by LB culture medium by 1:100, and continued to culture still the OD600 to 0.5-0.8. The bacteria were placed at 30℃, 33℃, 37℃, 42℃ with the negative control, positive control and blank control LB, and shaking at 220 rpm. We took samples at 0h, 4h, 8h and 20h respectively, used a microplate reader for spectrophotometer and fluorescence test, and recorded the data in OD600 and Fluorescence 485/510 (Excitation/Emission). The whole process is shown in Figure 3. We calculated the value of Normalized Fluorescence changes with time at different temperatures. | ||
| + | [[File:T--SHSBNU China--ways to calculate normalized fluorescence.jpeg|500px|thumb|center|]] | ||
| + | [[File:T--SHSBNU China--the process we fo to test the function of BBa K3594013.png|500px|thumb|center|the process we do to test the function of BBa K3594013]] | ||
| − | [[File: | + | the data from this experiment is shown in the following graph: |
| − | + | [[File:T--SHSBNU China--prediction and real fluorescence.jpeg|500px|thumb|center|prediction of fluorescence(left) and real normalized fluorescence value(right)]] | |
| + | As shown in the figure above, the value of Normalized Fluorescence in 42 ℃ sample increases most obviously, which proves that the heat-induced switch is effective at high temperatures. | ||
| − | + | ===Reference=== | |
| − | + | [1] Zihao Wang, Part: BBa_K2541001, iGEM18_Jilin_China, 2018-09-20 | |
| − | + | Available from: | |
| − | + | https://parts.igem.org/Part:BBa_K2541001 | |
| − | + | ||
| − | + | ||
| − | + | ||
Latest revision as of 04:40, 25 October 2020
Heat-inducible RNA-based thermosensor-1
A RNA-based thermosensor that can be used for temperature sensitive translational regulation which is based on the change of RNA sencondary structure. The heat-inducible RNA-based thermosensors can initiate translation of downstream genes at high temperatures.
1. Usage and Biology
Heat-inducible RNA-based thermosensors are RNA genetic control systems that sense temperature changes. At low temperatures, the mRNA adopts a stem-loop conformation that masks the Shine–Dalgarno (SD) sequence within the 5’-untranslated region (5’-UTR) and, in this way, prevents ribosome binding and translation. At elevated temperatures, the RNA secondary structure melts locally, thereby giving the ribosomes access to the SD sequence to initiate translation (Figure 1). Whereas natural RNA-based thermosensors have a relatively complicated secondary structure with multiple stems, hairpin loops and bulges which impeds application process.
Our team designed synthetic heat-inducible RNA-based thermosensors that are considerably simpler than naturally occurring thermosensors and can be exploited as convenient on/off switches of gene expression.
Figure 1. Mechanism of heat-inducible RNA-based thermosensors.
2. Design
The heat-inducible RNA-based theromsensors are designed on the basis of the melting temperature of the minimum free energy structure, consisting of ASD (anti-SD) sequence, loop sequence and consensus SD sequence (5’-AAGGAG-3’). To optimize the thermosensors for the desired melting temperature, intensity and sensitivity, three structural parameters come into consideration: stem length, loop size and mismatches or bulges in the stem.
Stem length is determined by ASD sequence because the SD sequence is conserved. Adding stem length can optimize heat-inducible RNA-based thermosensors to more high temperature, while decreasing stem length has the opposite effect. The stem length is 4 base parings in K2541001. Loop size can moderate thermosensors melting temperature to a suitable temperature. In K2541001, the loop sequence is AAUAA. Finally, we get the sequence as the figure 2B. After designing, the theromsensor sequence is predicted by computational methods mFOLD to get its minimum free energy and secondary structure (figure 2).
Figure 2. Design of K2541001. (A) The RNA secondary structure is predicted by mFOLD. (B) Thermosensor sequence, ASD sequence, loop sequence, the site of mismatch or bulge in the stem and ΔG are in the table.
3.Characterization
3.1 Measurement device
The thermosensor sequence is constructed on the pSB1C3 vector by Golden Gate assembly. The measurement device is composed of Anderson promoter (BBa_J23104), thermosensor (BBa_K2541001), sfGFP_optimism (BBa_K2541400) and double terminator (BBa_B0010 and BBa_B0012). We select a constitutive Anderson promoter J23104 as an appropriate promoter by pre-experiment. The sfGFP_optimism has faster folding speed and higher fluorescence intensity. The double terminator can reduce leakage (Figure 3). We characterized RNA-based thermosensors in E.coli DH5a.
3.2 Measurement results
K2541001, K2541029, K2541013 and K2541037 are four different heat-inducible RNA-based thermosensors. pos.control is positive control. The final normalized fluorescence was calculated as follows: normalized fluorescence = [(Fluorescence/Abs600)TS - (Fluorescence/Abs600)neg] / [(Fluorescence/Abs600)pos - (Fluorescence/Abs600)neg] ( TS = thermosensor, pos = positive control, and neg = BBa_J364007 ). As shown in figure 4, the fluorescence intensity of K2541001 increases with elevated temperature.
Figure 4. Characteristics of K2541001. Each set of six bars represents the activity level of a different thermosensor. The bar colors purple, aquamarine, light green, orange, red and brown represent the temperatures 29, 31, 35, 37, 39 and 42°C, respectively. The height of the bars corresponds to the normalized fluorescence.
3.3 Thermodynamic analyses
This year, our project is to design a collection of RNA-based thermosensors with different melting temperatures, intensity and sensitivity. We used a combination of experimental measurements and computations of RNA secondary structures. We studied a set of measured synthetic RNA-based thermosensors, finding consistency among our experimental and computational analyses.
Figure 5. Temperature response of the heat-inducible RNA-based thermosensor. (A) The red solid line represents the activity of the RNA-based thermosensor as a function of temperature. (B) Key quantitative features such as melting temperature, relative intensity, sensitivity and R-squared are in the table.
4. Collection of heat-inducible RNA-based thermosensors
Figure 6. Experimental measurements of the collection of heat-inducible RNA-based thermosensors show a variety of responses. (A) Rows represent activity levels of different thermosensors. (B) Each set of six bars represents the activity level of a different thermosensor. The bar colors purple, aquamarine, light green, orange, red and brown represent the temperatures 29, 31, 35, 37, 39 and 42°C, respectively. The height of the bars corresponds to the normalized fluorescence.
5. Conclusion
Our data show that efficient RNA-based thermosensors with different melting temperatures, intensity and sensitivity can be built from a single small RNA stem-loop structure masking the SD sequence, thus providing useful SynRT toolkit for the regulation of gene expression.
Verification from SHSBNU_China 2020
In SHSBNU_China team's design, we use this thermosensor as a heat-responsive suicide switch. Locust plagues take place mainly in spring, and few in summer. Therefore, in spring, when the temperature is lower than 35 ℃, the mRNA adopts a stem-loop conformation that masks the Shine–Dalgarno (SD) sequence within the 5’-untranslated region (5’-UTR) and, in this way, prevents ribosome binding and translation [1]. In summer, when the temperature is higher than 35 ℃, the RNA secondary structure melts locally, thus the lysis gene in the downstream of the gene circuit would start transcription, resulting in the demise of our engineered bacteria.
Characterization
Plasmid construction
In our experiments, we used this part: K2541001, along with the sfGFP (BBa_I746916) as a reporter and PhiX174E (BBa_K3594001), as our heat-responsive suicide switch. We constructed two plasmids in order to complete the thermal response suicide switch successfully: PSHS1_PSB1C3_BBa_J23104_K2541001_sfGFP and PSHS2_PSB1C3_BBa_J23104_K2541001_Phix174E. PSHS1 can sense high temperature and generate fluorescent, which is used to verify if the thermal response switch is effective. PSHS2 contains a cell lysis gene PhiX174E, and this plasmid is used to test if our heat-induced suicide system works well.
Testing Result
Firstly, we tested the function of BBa_K3594013. In order to test whether it works and produce fluorescence, we decided to use fluorescence microplate reader to measure the normalized fluorescence that the gene expresses in different temperature. We transformed BBa_K3594013 to commercial E. coli DH5 alpha competent cells. Then, we cultured the single colonies with PSHS1 for overnight. At the next day, we diluted the culture by LB culture medium by 1:100, and continued to culture still the OD600 to 0.5-0.8. The bacteria were placed at 30℃, 33℃, 37℃, 42℃ with the negative control, positive control and blank control LB, and shaking at 220 rpm. We took samples at 0h, 4h, 8h and 20h respectively, used a microplate reader for spectrophotometer and fluorescence test, and recorded the data in OD600 and Fluorescence 485/510 (Excitation/Emission). The whole process is shown in Figure 3. We calculated the value of Normalized Fluorescence changes with time at different temperatures.
the data from this experiment is shown in the following graph:
As shown in the figure above, the value of Normalized Fluorescence in 42 ℃ sample increases most obviously, which proves that the heat-induced switch is effective at high temperatures.
Reference
[1] Zihao Wang, Part: BBa_K2541001, iGEM18_Jilin_China, 2018-09-20 Available from: https://parts.igem.org/Part:BBa_K2541001
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]